- Title
-
Zebrafish Tbx16 regulates intermediate mesoderm cell fate by attenuating Fgf activity
- Authors
- Warga, R.M., Mueller, R.L., Ho, R.K., and Kane, D.A.
- Source
- Full text @ Dev. Biol.
|
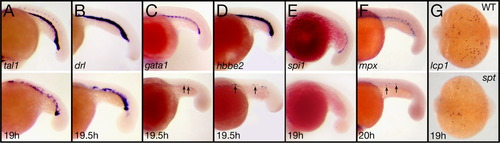
spt mutants have less of all types of blood. (A–F) Expression in the intermediate cell mass at 19–20 h of: (A) tal1 and (B) drl in all blood cells; (C) gata1 and (D) hbbe2 in red blood cells; (E) spi1 in white blood cells; and (F) mpx in neutrophils. (G) lcp expression at 19 h in macrophages derived from the head blood island. Embryos are shown from a (A–F) left side view, or (G) dorsal view with anterior to the left. Arrows designate single cells. EXPRESSION / LABELING:
PHENOTYPE:
|
|
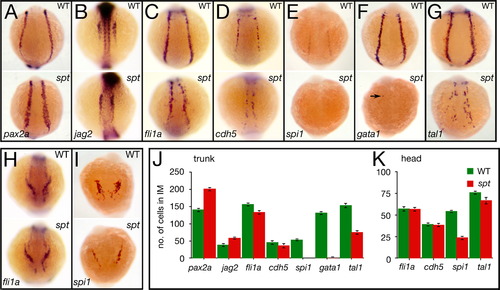
spt mutants possess more nephric precursors, but have fewer endothelial and hematopoietic precursors. (A–G) Expression in the trunk at 14.5 h of: (A) pax2a and (B) jag2, in nephric cells; (C) fli1a and (D) cdh5, in endothelial cells; and (E) spi1, in white, (F) gata1, in red, and (G) tal1, in all blood cells (arrow designates a single cell). (H, I) Expression in the head at 14.5 h of: (H) fli1a, in endothelial cells; and (I) spi1, in macrophages. (J, K) Quantification of intermediate mesoderm precursors for: (J) trunk, and (K) head. Graphs show the average number of cells per marker including standard error; 9 embryos for each category were counted. Embryos are shown from a (A–G) posterior view, or (H, I) anterior view with dorsal to the top. EXPRESSION / LABELING:
PHENOTYPE:
|
|
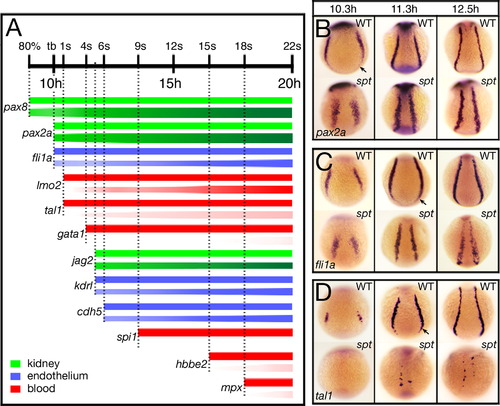
Spatiotemporal gene expression is altered in the intermediate mesoderm of spt mutants. (A) Temporal expression profiles of nephric (green), endothelial (blue) and hematopoietic (red) genes expressed in the trunk intermediate mesoderm. For each gene, the bar above symbolizes wild-type expression and the bar below mutant expression. Lightness or darkness of color for mutant denotes less or more cells. Vertical dotted lines denote onset of expression in wildtype. Examples of expression are shown in adjacent panels B–D and in Fig. 1, Fig. 2 and Fig. 5 and Supplementary Fig. 2. (B–D) Spatial pattern of trunk intermediate mesoderm genes: (B) pax2a, in nephric progenitors; (C) fli1a, in endothelial progenitors; and (D) tal1, in hematopoietic progenitors. Embryos are shown from a posterior view with dorsal to the top. Arrow designates portion missing in the mutant. EXPRESSION / LABELING:
|
|
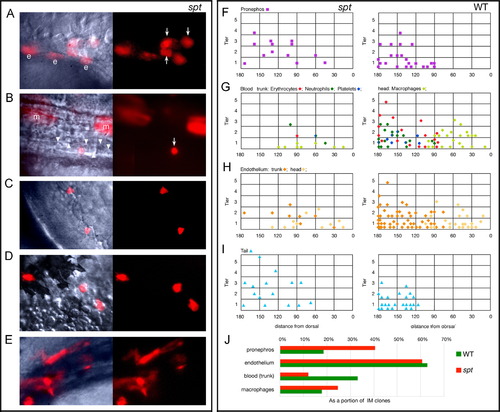
Trunk intermediate mesoderm and tail derivatives originate from more dorsal and animal locations in the spt mutant. (A–E) High magnification bright field and UV views of labeled cells in spt mutants at 24–36 h: (A) Pronephric cells (arrows); (B) an erythrocyte (arrow), merged view shows neighboring unlabeled erythrocytes (arrowheads); (C) neutrophils; (D) macrophages; and (E) endothelial cells. All cells resembled their wild-type counterpart. Other labeled cells include hindgut endoderm (e) and muscle cells (m). (F–J) Location of clones at 6 h that gave rise to: (F) Pronephros; (G) Blood: trunk (erythrocytes, neutrophils and platelets) versus head (macrophages); (H) Endothelium: trunk versus head; and (I) Tail (caudal fin mesenchyme and tail muscle). For conventional presentation, clones are projected onto the left side even if they were on the right. Axes: y, distance from the blastoderm margin (zero) measured in cell tiers and x, distance from dorsal (zero) measured in degrees radian. For trunk blood, the color of the symbol indicates whether the clone gave rise only to erythrocytes (red), or also to neutrophils (green), platelets (blue) or both (green/blue). (J) The percent of clones that gave rise to a specific derivative. The entire data set for intermediate mesoderm included 130 wild-type clones, and 33 mutant clones. |
|
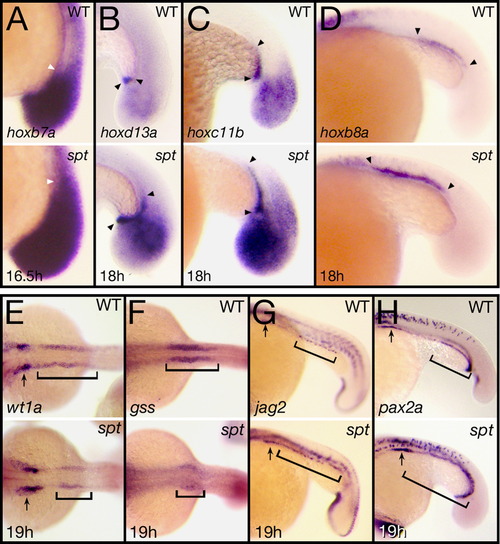
spt mutants possess more posterior mesoderm. (A) Expression at 16.5 h of hoxb6b, arrowhead indicates the extent of anterior staining within the tailbud mesoderm. (B–D) Expression at 18 h of: (B) hoxd13a, (C) hoxc11b, and (D) hoxb8a, arrowheads indicate extent of staining in the cloaca (B) or in the pronephros (C and D). (E–I) Expression at 19 h of: (E) wt1a and (F) gss, marking anterior segments and (G) jag2 and (H) pax2a, marking posterior segments. Arrows indicate label in the most anterior nephric segment and brackets indicate regions that are altered. Embryos are shown from a (A–D and G, H) left side view, or (E, F) dorsal view with anterior to the left. EXPRESSION / LABELING:
|
|
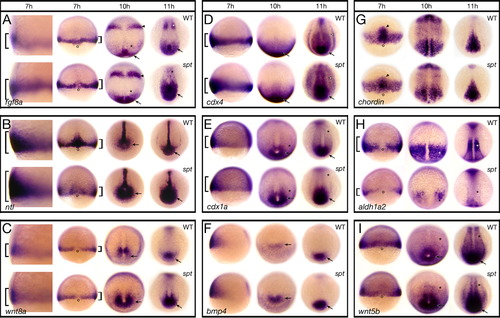
Components of the Fgf and Wnt pathway are more highly expressed in spt mutants. Expression of (A) fgf8a; (B) ntl; (C) wnt8a; (D) cdx4; (E) cdx1a; (F) bmp4; (G) chordin; (H) aldh1a2; and (I) wnt5b at: 7 h, 10 h and 11 h. Embryos at 7 h are shown in (A–C) a high magnification left side view and a low magnification dorsal view, (D–F) a left side view, and (G–I) a dorsal view. Older embryos are all shown in a dorsal posterior view. Designations (7 h): brackets, more or less expression; open circles, dorsal side; arrowhead, prechordal plate; (later stages): arrow, more expression in the posterior region and tailbud; asterisk, less expression in the paraxial mesoderm; arrowhead, more expression in the future midbrain-hindbrain boundary; open arrowhead, more expression in the intermediate mesoderm. EXPRESSION / LABELING:
|
|
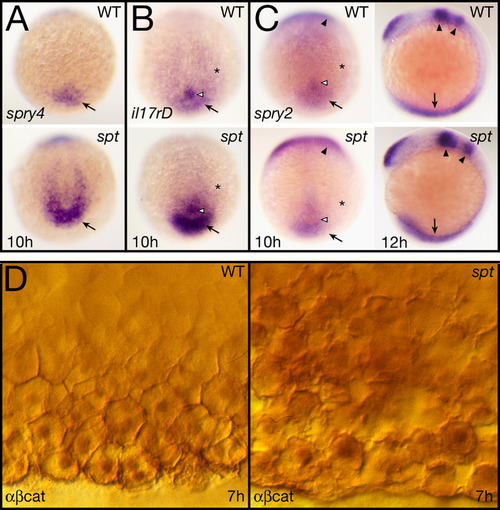
Fgf and Wnt signaling activity are elevated in spt mutants. (A–C) Expression at 10 and 12 h of: (A) spry4, (B) il17rd, and (C) spry2. All 10 h embryos are shown in a dorsal posterior view, the 12 h embryos in (C) are shown in left side view. Designations: arrow, tailbud (10 h) or posterior mesoderm (12 h); open arrowhead, Kupfer′s vesicle; asterisk, paraxial mesoderm; and arrowheads, midbrain-hindbrain boundary region and rhombomere 4. (D) Expression at 7 h of β-catenin protein seen at high magnification in ventral marginal cells. Cells in the mutant (right), are not as cohesive with one another as in wildtype and make abnormal intercellular contacts and gaps. They also appear to have higher cytoplasmic and nuclear staining. EXPRESSION / LABELING:
|
|
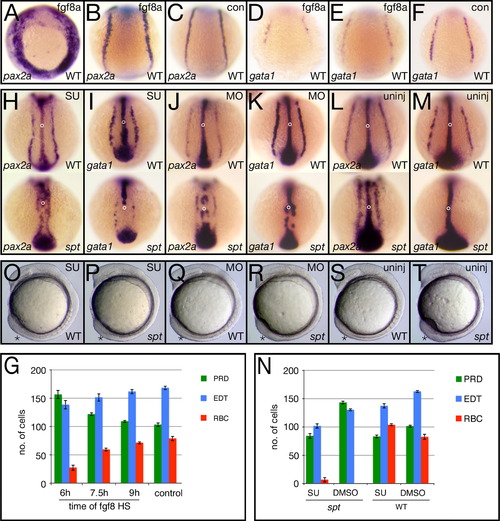
Altering Fgf8a signaling activity changes the balance of intermediate mesoderm-derived fates. (A–G) Overexpressing fgf8a by heat-shock effects individual intermediate mesoderm fates differently. (A–F) in situ analysis at 12 h after heat shock of the transgene at 6 h showing different examples of the phenotypes. Control siblings (con) not carrying the transgene, were also heat-shocked. pax2a expression identifies pronephric precursors (PRD), gata1 expression identifies red blood precursors (BLD) and (not shown) fli 1a expression identifies endothelial precursors (EDT). (G) Quantification of precursor cells at 12 h after heat shock at 6, 7.5 or 9 h. Graphs show the average number of cells per fate and the standard error. (H–T) Inhibiting Fgf8a activity effects individual intermediate mesoderm fates in an opposite manner to overexpression. (H–M) in situ analysis at 12 h of embryos exposed to Su5402 for 1 h at 6 h (H, I), injected at the 2 to 8-cell stage with the fgf8a morpholino (K, L) and their uninjected siblings (L, M). Embryos were also probed with ntl to monitor the effect on morphogenesis (open circle designates the notochord). Note that the tailbud marked by ntl expression is also smaller in all the SU5402 treated and fgf8a morphant embryos. (N) Quantification of precursor cells at 12 h after exposure to SU5402 for 1 h at 6 hours. (O–T) Live embryos depicting the tailbud phenotype after 1 h exposure to Su5402 at 6 h (O, P), or following injection of the fgf8a morpholino (Q, R) and their uninjected siblings (S, T). Embryos are shown from a (A–F and H–M) dorsal posterior view, or (O–T) left side view; asterisk designates the tailbud. EXPRESSION / LABELING:
|
|
wildtype and spt mutant hematopoietic, hemangioblast and endothelial gene expression in the intermediate cell mass. (A) drl a hematopoietic marker; (B) tal1 and (C) lmo2 hemangioblast markers; and (D) kdrl, (E) fli1a and (F) cdh5 endothelial markers. Based on the pattern of expression at this time, tal1 is mostly restricted to blood cells whereas lmo2 is mostly restricted to endothelial cells (compare A,B and C,D). Embryos are shown from a (A–D) dorsal view, and (E–F) left side view; rostral is to the left. |
|
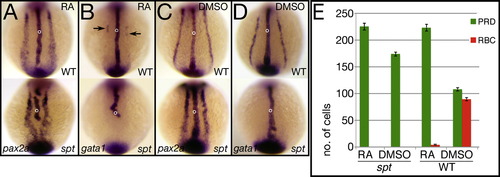
Transient exposure to RA gives results similar to overexpression of Fgf8a. (A–E) Exposure at 6 h to 1 μM RA for 1 h. (A–D) in situ analysis at 12 h. Control siblings were exposed to just the carrier (5% DMSO). pax2a expression identifies pronephric precursors (PRD), and gata1 expression identifies red blood precursors (BLD). Embryos were also probed with ntl (open circle). (E) Quantification of precursor cells at 12 h. Graphs show the average number of cells per fate and the standard error. Embryos are shown from a posterior view. |
|
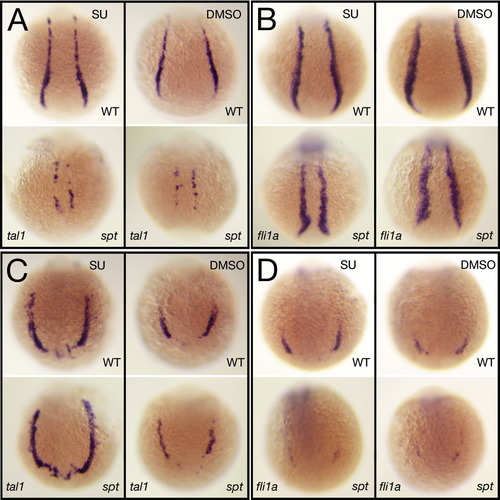
Blood and endothelium are affected by inhibiting Fgf8a activity. (A–D): in situ analysis at 12 h after 1 h exposure to Su5402 at 6 h. Control siblings were exposed to just the carrier (5% DMSO). Similar results were obtained using the fgf8a morpholino (data not shown). tal1 expression identifies hematopoietic precursors and fli1a expression identifies endothelial precursors in the trunk (A, B) and in the head (C, D). Embryos are shown from a (A,B) posterior view and (C,D) anterior view, dorsal to the top. |
Reprinted from Developmental Biology, 383(1), Warga, R.M., Mueller, R.L., Ho, R.K., and Kane, D.A., Zebrafish Tbx16 regulates intermediate mesoderm cell fate by attenuating Fgf activity, 75-89, Copyright (2013) with permission from Elsevier. Full text @ Dev. Biol.