- Title
-
Fox proteins are modular competency factors for facial cartilage and tooth specification
- Authors
- Xu, P., Balczerski, B., Ciozda, A., Louie, K., Oralova, V., Huysseune, A., Crump, J.G.
- Source
- Full text @ Development
|
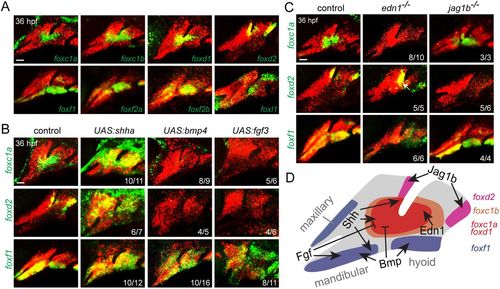
Combinatorial signaling establishes distinct Fox expression domains. (A) In situ hybridization at 36 hpf shows expression of Fox genes (green) relative to dlx2a+ CNCs (red) of the first two arches. (B) Expression of foxc1a, foxd2 and foxf1 (green) relative to dlx2a+ CNCs (red) upon misexpression of Shha, Bmp4 or Fgf3. Embryos doubly transgenic for hsp70l:Gal4 and UAS:shha, UAS:bmp4 or UAS:fgf3 were subjected to heat shock induction from 20 to 24 hpf and then fixed at 36 hpf for analysis. (C) Expression of foxc1a, foxd2 and foxf1 (green) relative to dlx2a+ CNCs (red) in edn1 or jag1b mutants versus sibling controls. Numbers denote proportions of embryos with displayed patterns. Scale bars: 25 μm. (D) Summary of positive and negative inputs on Fox expression in the first two arches. |
|
Loss of upper facial cartilage in Fox-C mutants. (A) Ventral views of dissected facial skeletons, with cartilage in blue and bones and teeth in red. Schematics show affected elements in color. Ch, ceratohyal; Hm, hyomandibular; M, Meckel's; Pq, palatoquadrate; Sy, symplectic. (B) Lateral views of intact heads stained with Alcian Blue (cartilage) and Alizarin Red (bones and teeth). Shown below are unilateral dissections of skeletal elements of the first and second arches (red boxed region). Accompanying schematics show dose-dependent reductions of the Pq (magenta), Sy (green) and Hm (blue) cartilages. foxc1a−/− and foxc1a−/−; foxc1b−/− mutants also display cardiac edema and an overall smaller head. (C) Quantification of the relative length of Sy compared with Ch. ***P<0.001 versus wild-type sibling control using Student's t-test. n.s., not significant versus control (P=0.35). Error bars represent s.e.m. (D) Dissections of neurocranial cartilages show loss of the trabecular cartilages (Tr, arrow) in foxc1a−/−; foxc1b−/− mutants. Numbers denote proportions of embryos with displayed phenotypes. Ep, ethmoid plate. Scale bars: 25 μm. |
|
Loss of midline cartilage and pharyngeal teeth in Fox-F mutants. (A) Ventral views of dissected facial skeletons, with cartilage in blue and bones and teeth in red. Schematics show affected elements in color. Bb, basibranchials; Bh, basihyal; Ch, ceratohyal; M, Meckel's; Ptp, pterygoid process. (B) Lateral views of wild-type sibling control and foxf1; foxf2a; foxf2b triple mutant at 5 dpf. Mutants show anterior truncation of the head (arrow). (C) Dissections of neurocranial cartilages show loss of the trabecular cartilages (Tr, arrow) in foxf1; foxf2a; foxf2b triple mutants. Ep, ethmoid plate. (D) Sagittal sections through the tooth-forming seventh arch of a wild-type sibling control and foxf1; foxf2a; foxf2b triple mutant at 4 dpf. All teeth (3V1, 4V2 and 5V1) are missing in mutants. c, chorda; cb5, ceratobranchial 5; ov, otic vesicle. (E,F) Whole-mount in situ hybridizations show loss of dlx2b (E) and fgf3 (F) in the developing tooth regions (arrows) of foxf1; foxf2a; foxf2b triple mutants. Numbers denote proportions of embryos with displayed phenotypes or expression patterns. Scale bars: 25 μm. EXPRESSION / LABELING:
PHENOTYPE:
|
|
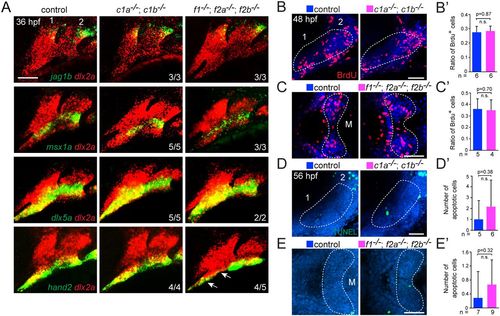
Dorsoventral gene expression, proliferation and apoptosis in Fox mutants. (A) Fluorescence in situ hybridizations of the first two arches (numbered) show expression of jag1b (dorsal), msx1a and dlx5a (intermediate), or hand2 (ventral) in green relative to dlx2a+ arch CNCs in red. Expression is largely unaffected in foxc1a; foxc1b double mutants (c1a−/−; c1b−/−) and foxf1; foxf2a; foxf2b triple mutants (f1−/−; f2a−/−; f2b−/−) compared with sibling controls, with the exception of reduced hand2 in the ventral first arch of foxf1; foxf2a; foxf2b triple mutants (arrows). (B-C′) Analysis of proliferation. Confocal sections show BrdU+ cells (red) compared with all nuclei (Hoechst, blue) in lateral views of the first two arches (numbered) of foxc1a−/−; foxc1b−/− embryos and ventral views of the first arch Meckel's cartilage domains (M) of foxf1−/−; foxf2a−/−; foxf2b−/− embryos, relative to sibling controls. Dashed lines represent the regions quantified in the accompanying graphs. (D-E′) Analysis of apoptosis. Maximum intensity projections show the number of TUNEL+ cells (green) in lateral views of the first two arches (numbered) of foxc1a−/−; foxc1b−/− embryos and ventral views of the first arch M cartilage domains of foxf1−/−; foxf2a−/−; foxf2b−/− embryos, relative to sibling controls. Dashed lines represent the regions quantified in the accompanying graphs. Error bars represent s.e.m. Scale bars: 50 μm. n.s., not significant. |
|
Fox genes are required for chondrogenic gene expression downstream of Sox9a. (A) Fluorescence in situ hybridizations show that barx1+ pre-cartilage condensations of the first two arches are unaltered in foxc1a; foxc1b double mutants (c1a−/−; c1b−/−) and foxf1; foxf2a; foxf2b triple mutants (f1−/−; f2a−/−; f2b−/−) compared with sibling controls. (B,C) Schematic of the cartilaginous facial skeleton shows regions examined by fluorescence in situ hybridization. Maximum intensity projections for foxc1a; foxc1b mutants at 56 hpf (B) and representative sections for foxf1; foxf2a; foxf2b mutants at 72 hpf (C) show expression of the chondrogenic genes col2a1a, matn1, matn4 or acana (green) relative to sox9a (red). Arrows indicate regions in which sox9a+ cells have reduced chondrogenic gene expression relative to sibling controls. Numbers denote proportions of embryos with displayed patterns. Scale bars: 50 μm. EXPRESSION / LABELING:
PHENOTYPE:
|
|
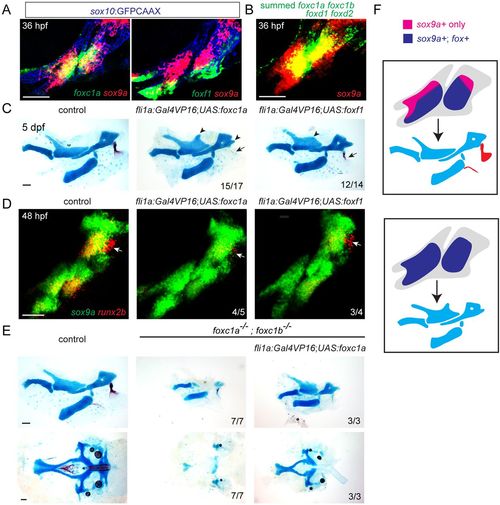
Fox CNC misexpression induces ectopic cartilage and inhibits dermal bone. (A) Confocal sections show expression of foxc1a or foxf1 (green) relative to sox9a (red) in sox10:GFPCAAX+ CNCs of the first two arches (anti-GFP, blue). (B) Maximum intensity projection shows fluorescence in situ hybridization using pooled probes against foxc1a, foxc1b, foxd1 and foxd2 (green) relative to sox9a (red). (C) Unilateral dissections of the first and second arch skeletons stained for cartilage (Alcian Blue) and bone (Alizarin Red). CNC misexpression of Foxc1a (fli1a:Gal4VP16; UAS:foxc1a) or Foxf1 (fli1a:Gal4VP16; UAS:foxf1) results in ectopic cartilage in the upper face (arrowheads), and loss of the opercle bone (arrows). (D) Double fluorescence in situ hybridizations of the first two arches show reduction of the osteoblast gene runx2b (red) in the future opercle bone (arrow) but no change in sox9a (green) upon misexpression of Foxc1a or Foxf1. (E) Dissections of the first and second arch skeletons and neurocranial cartilages. Misexpression of Foxc1a in CNCs (fli1a:Gal4VP16; UAS:foxc1a) rescues cartilage development in the upper face (top) and neurocranium (bottom). Scale bars: 50 μm. (F) Model of how arch-wide Fox misexpression expands the territory in which Sox9a induces cartilage (blue) at the expense of dermal bone (red). |
|
Arch expression of Fox genes (A) Transcripts Per Million Reads (TPM) values for Fox genes in FACS-purified arch CNCs at 20, 28, and 36 hpf. (B) Fluorescent in situ hybridizations at 30 hpf show similar expression of foxc1a, foxc1b, foxd1, and foxd2 (green) in dlx2a+ CNCs (red) of the first two arches. (C) Fluorescent in situ hybridizations at 36 hpf show expression of foxf1 in the ventral domains (arrows) of the gill-bearing branchial arches (numbered 3-6), but only minimal expression of foxc1a and foxd2. (D) Double fluorescent in situ hybridizations at 36 hpf show relative expression of foxc1a (green) to foxc1b (red) and foxd1 (red), and foxf1 (red) to foxf2a (green), in Sox10:GFPCAAX+ CNCs (white) of the first two arches (dotted lines). foxc1b is more broadly expressed than foxc1a and foxd1. foxf1 and foxf2a are expressed in largely similar domains. (E-F) Fluorescent in situ hybridizations show expression of foxc1a, foxc1b, foxd2, and foxf1 (green) relative to sox9a+ chondrocytes (red) at 48 hpf. By this stage, Fox gene expression is generally excluded from early cartilage, including around the distal tip of Meckel’s (M), although expression of foxc1a and foxc1b is observed in nascent sox9a+ chondrocytes of the hyoid joint (Hj, arrows). We also observe expression of foxc1b and foxd2 in intermediate and dorsal domains (arrows), respectively, of the posterior arches, and weak expression of foxc1a in the seventh arch (arrow). (G) In the developing neurocranium, foxc1b co-localizes with sox9a, while foxf1 is seen in cells surrounding the trabeculae (Tr) and ethmoid plate (Ep). (H) Confocal section shows expression of foxf1 (green) in the dental mesenchyme adjacent to the dlx2b+ dental epithelium (red) and the sox10:GFPCAAX+ ceratobranchial cartilage 5 (cb5, labeled in white by anti-GFP antibody). Scale bars = 25 μm. EXPRESSION / LABELING:
|
|
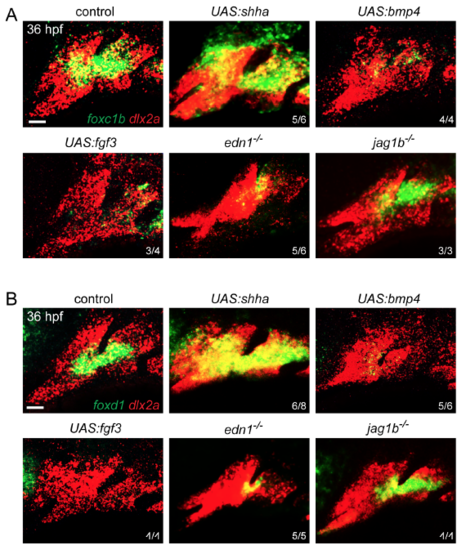
Regulation of foxc1b and foxd1 arch expression Expression of foxc1b (A) and foxd1 (B) in green, relative to dlx2a+ CNCs (red) of the first two arches. Similar to foxc1a in Figure 1, foxc1b and foxd1 expression is expanded by Shha misexpression, inhibited by Bmp4 and Fgf3 misexpression, and reduced in edn1 but not jag1b mutants. Embryos doubly transgenic for hsp70l:Gal4 and UAS:shha, UAS:bmp4, or UAS:fgf3 were subjected to a heatshock from 20-24 hpf to induce ligand expression throughout embryos. Numbers indicate proportion of animals showing the displayed patterns. Scale bars = 25 μm. |
|
Fox mutant alleles (A) Schematics of Fox alleles generated for this study. The predicted protein products are shown in black with the Forkhead DNA-binding domains in magenta. For the targeted DNA sequences, series of red dashes indicate deletions, and green letters indicate insertions. For the foxl1sa1842 generated by the Sanger Center, the nonsense mutation (t > a) is indicated in green. Sites where the predicted proteins are truncated by mutations are indicated with arrows. (B-C) Unilateral dissections of the skeletons of the first two arches stained with Alcian Blue (cartilage) and Alizarin Red (bone) at 5 dpf (B) or 6 dpf (D). No defects were seen in at least 20 larvae examined for each genotype. Scale bars = 25 μm. |
|
Fate mapping of the Fox-C expression domain Images show the first two arches at 36 hpf (A) and the resultant skeleton at 6 dpf (B). sox10:kikGR+ arch CNCs (green) were photoconverted to red fluorescence with UV light using the ROI function on a Zeiss LSM800 confocal microscope. Photoconversion in a similar domain to where foxc1a and foxd1 are expressed resulted in labeling of the palatoquadrate (Pq), symplectic (Sy), and hyosymplectic (Hm) cartilages. Scale bars = 25 μm. |
|
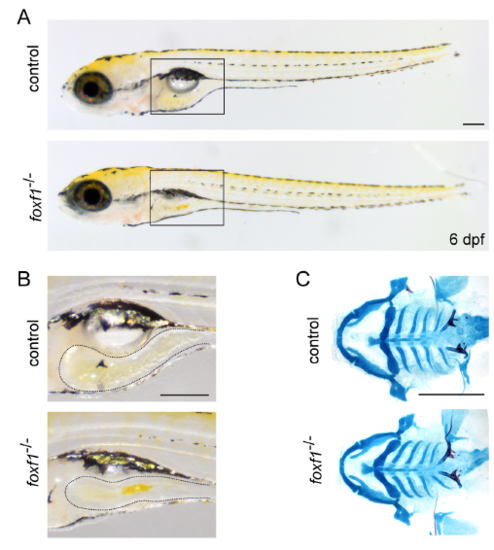
Phenotype of foxf1 single mutants (A) Brightfield images show morphology of control and foxf1 single mutants in lateral view at 6 dpf. Swim bladders do not properly inflate in all foxf1 mutants. (B) Enlarged images of the boxed regions show reduced intestine (dotted areas) in foxf1 mutants compared to controls. (C) Ventral views of dissected facial skeletons stained by Alcian Blue (cartilage) and Alizarin Red (bones and teeth). No defects are observeded in foxf1 mutants. Scale bars = 100 μm. PHENOTYPE:
|
|
Expression of foxc1a in sox9a mutants and chondrocyte defects in Fox-C mutants (A) Expression of foxc1a in the first two arches is unchanged in sox9a mutants at 42hpf. (B) Confocal imaging of chondrocytes labeled by col2a1:mCherry-NTR (red) and osteoblasts labeled by sp7:GFP (green) at 72 hpf. In foxc1a; foxc1b mutants, col2a1:mCherry-NTR expression is weaker and/or reduced in the palatoquadrate (Pq), symplectic (Sy), and hyosymplectic (Hm) domains. sp7:GFP+ osteoblasts of the developing opercular bone (Op, arrows) are less affected. Numbers indicate proportion of animals showing the displayed patterns. Scale bars = 100 μm. EXPRESSION / LABELING:
PHENOTYPE:
|
|
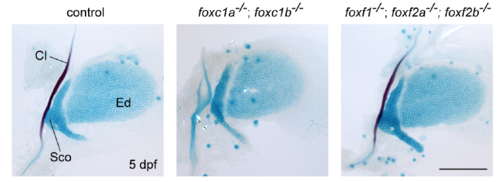
Normal pectoral fins in Fox-C and Fox-F mutants Images show pectoral fin skeletons stained with Alcian Blue (cartilage) and Alizarin Red (bone) at 5 dpf. No defects were seen in either foxc1a; foxc1b or foxf1; foxf2a; foxf2b mutants. The lack of mineralization of the Cl bone in foxc1a; foxc1b mutants is likely an indirect consequence of cardiac edema. Cl, cleithrum; Ed, endoskeletal disc; Sco, scapulocoracoid. Scale bar = 100 μm. |