- Title
-
Gremlin 2 regulates distinct roles of BMP and Endothelin 1 signaling in dorsoventral patterning of the facial skeleton
- Authors
- Zuniga, E., Rippen, M., Alexander, C., Schilling, T.F., and Crump, J.G.
- Source
- Full text @ Development
|
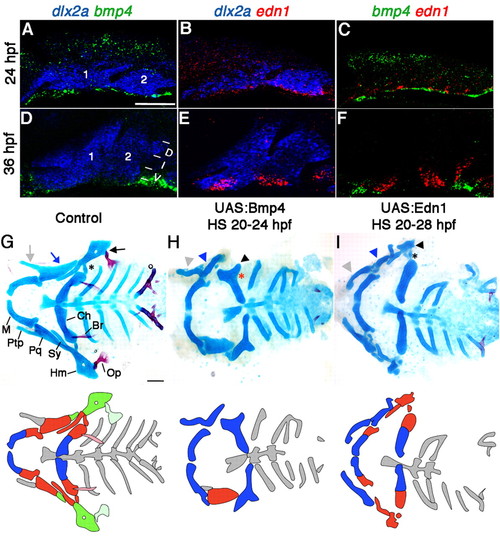
Facial skeletal defects upon Bmp4 or Edn1 misexpression. (A-F) Confocal projections of in situ hybridization for dlx2a (blue), bmp4 (green) and edn1 (red) at 24 hpf (A-C) and 36 hpf (D-F) in wild type. Mandibular (1) and hyoid (2) arches are labeled, as well as dorsal (D), intermediate (I) and ventral (V) domains. (G-I) Ventral views (top) and schematics (below) of 5 dpf facial skeletons in control hsp70I:Gal4 (G) and hsp70I:Gal4; UAS:Bmp4 larvae subjected to a 4-hour heat-shock (H), and hsp70I:Gal4; UAS:Edn1 larvae subjected to an 8-hour heat-shock (I). Cartilage is blue and bone red. Schematics show dorsal (green), intermediate (red) and ventral (blue) regions with dermal bones lightly shaded. Hm (black arrow), Pq (blue arrow) and Ptp (grey arrow) were transformed (arrowheads) in UAS:Bmp4 and UAS:Edn1 larvae. In the intermediate second arch, the joint (asterisk) and symplectic bone were lost in UAS:Bmp4 but not UAS:Edn1 larvae. M, Meckel’s cartilage; Pq, palatoquadrate cartilage; Ptp, pterygoid process; Sy, symplectic cartilage; Hm, hyomandibular cartilage; Ch, ceratohyal cartilage; Op, opercular bone; Br, branchiostegal ray bone. Scale bars: 50 μm. |
|
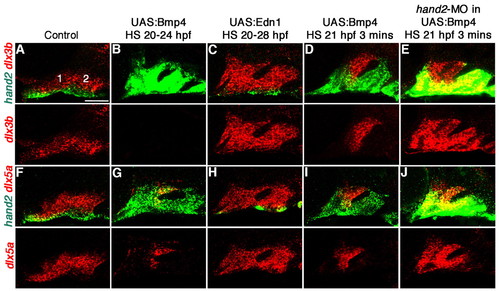
Distinct effects of Bmp4 and Edn1 misexpression on hand2 and dlx3b/5a expression. (A-J) Confocal sections of in situ hybridization for hand2 (green) with dlx3b (red, A-E) or dlx5a (red, F-J) in the mandibular (1) and hyoid (2) arches of 36 hpf control hsp70I:Gal4 (A,F) and hsp70I:Gal4; UAS:Bmp4 embryos subjected to a 20-24 hpf heat-shock (B,G), as well as hsp70I:Gal4; UAS:Edn1 embryos subjected to a 20-28 hpf heat-shock (C,H). (A,F) In controls, hand2 (n=48/48) was restricted to the ventral domain, and dlx3b (n=30/30) and dlx5a (n=18/18) to intermediate domains. (B,G) In UAS:Bmp4 embryos, hand2 was upregulated (n=23/32), dlx3b was variably expanded (n=11/21) or reduced (n=9/21), and dlx5a was also variably expanded (n=4/20) or reduced (n=9/20). (C,H) In UAS:Edn1 embryos, hand2 was reduced (n=34/34) and dlx3b (n=20/20) and dlx5a (n=14/14) were expanded. (D,E,I,J) Un-injected hsp70I:Gal4; UAS:Bmp4 embryos subjected to a 3-minute heat-shock at 21 hpf (D,I) never showed co-localization of hand2 with dlx3b (n=0/29) or dlx5a (n=0/21), whereas hand2 colocalized with dlx3b (n=21/35) and dlx5a (n=16/16) in hand2-MO-injected embryos (E,J). Anterior is towards the left and dorsal is upwards. Scale bar: 50 μm. |
|
DV gene expression in Bmp4 and Edn1 misexpression embryos. (A-R) In situ hybridization shows gene expression in the mandibular (1) and hyoid (2) arches of control hsp70I:Gal4 and hsp70I:Gal4; UAS:Bmp4 embryos subjected to a 20-24 hpf heat-shock and hsp70I:Gal4; UAS:Edn1 embryos subjected to a 20-28 hpf heat-shock. (A-C) msxe (36 hpf): compared with controls (A, n=22), expression was markedly expanded in UAS:Bmp4 (B, n=17/18) and slightly expanded in UAS:Edn1 (C, n=10/12) embryos. (D-F) epha4b (36 hpf): compared with controls (D, n=11), expression was markedly expanded in UAS:Bmp4 (E, n=12/12) and moderately expanded in UAS:Edn1 (F, n=20/20) embryos. (G-I) dlx6a (36 hpf): compared with controls (G, n=8), expression was variably expanded (n=4/7) or reduced (n=3/7) in UAS:Bmp4 (H), and markedly expanded in UAS:Edn1 (I, n=28/28) embryos. (J-L) nkx3.2 (44 hpf): compared with controls (J, n=13), expression was lost in UAS:Bmp4 (K, n=9/9) and expanded in UAS:Edn1 (L, n=21/25) embryos. (M-O) jag1b (36 hpf): compared with controls (M, n=14), expression was reduced in UAS:Bmp4 (N, n=14/20) and UAS:Edn1 (O, n=21/25) embryos. (P-R) hey1 (36 hpf): compared with controls (P, n=36), expression was reduced in UAS:Bmp4 (Q, n=26/27) and UAS:Edn1 (R, n=19/26) embryos. Arrow indicates hey1 staining in ventral mesoderm. (S-U) Confocal sections of in situ hybridizations for dlx3b (green) and msxe (red) in a 36 hpf wild-type embryo. Merged (S) and single (T,U) panels are shown. Intermediate (I) and ventral-intermediate (VI) domains are depicted. Scale bars: 50 μm. EXPRESSION / LABELING:
|
|
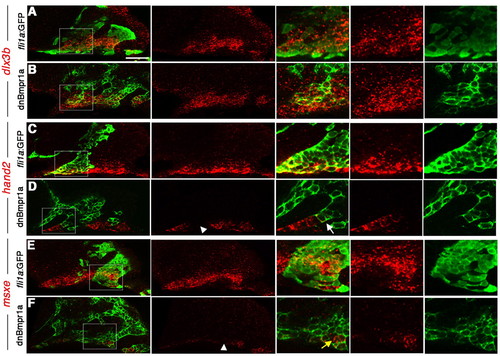
Cell-autonomous regulation of DV gene expression by BMP. (A-F) Confocal sections of anti-GFP staining (green) and dlx3b (A,B), hand2 (C,D) and msxe (E,F) expression (red). Merged and individual channels are shown, as well as higher magnification views of boxed regions. Wild-type hosts received CNCC precursor transplants from either wild-type fli1a:GFP (A,C,E) or hsp70I:dnBmpr1a-GFP (B,D,F) donors. hand2 and msxe were cell-autonomously reduced in hsp70I:dnBmpr1a-GFP clones (white arrowheads), whereas dlx3b was largely unaffected. In high magnification views, the white arrow indicates a hsp70I:dnBmpr1a-GFP clone displaying loss of hand2, and the yellow arrow indicates a small clone of wild-type host cells still expressing msxe. Scale bar: 50 μm. |
|
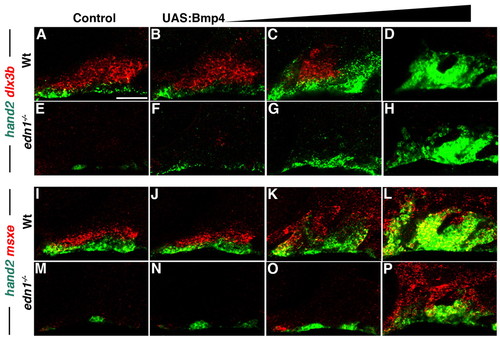
Bmp4 induces hand2 and msxe expression in the absence of Edn1. (A-P) Confocal sections of 36 hpf in situ hybridizations for hand2 (green) with dlx3b (red, A-H) or msxe (red, I-P) in control hsp70I:Gal4 (A,I), edn1-/- mutant (E,M), UAS:Bmp4 (B-D,J-L) and edn1-/-; UAS:Bmp4 (F-H,N-P) embryos. Increasing periods of Bmp4 heat-shock induction [1 minute at 21 hpf (B,F,J,N), 3 minutes at 21 hpf (C,G,K,O) and 4 hours from 20-24 hpf (D,H,L,P)] resulted in progressive recovery of hand2 and msxe but not dlx3b expression in edn1-/- mutants. Consistent phenotypes were observed for the following: (A) n=55, (B) n=31, (C) n=15, (D) n=9, (E) n=8, (F) n=3, (G) n=4, (H) n=1, (I) n=37, (J) n=11, (K) n=6, (L) n=9, (M) n=20, (N) n=2, (O) n=3 and (P) n=2. Scale bar: 50 μm. EXPRESSION / LABELING:
|
|
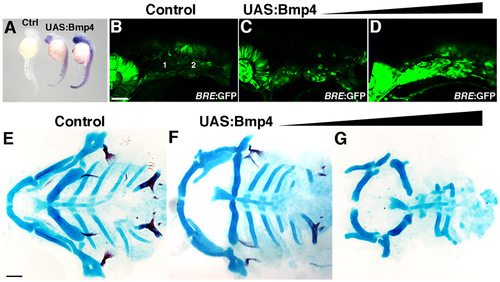
Edn1 and Jag1b negatively regulate grem2 expression. (A-F) Confocal sections of in situ hybridization for grem2 (red) and dlx2a (green) in controls (A, n=62), jag1bb1105 mutant (B, n=9), edn1-/- mutant (C, n=5), UAS:Edn1 (D, n=17), hsp70I:dnBmpr1a-GFP (E, n=32) and UAS:Bmp4 (F, n=23) embryos at 36 hpf. In UAS:Edn1 embryos, grem2 expression was seen throughout the mandibular and hyoid arches, except for the ventral-most domain (white arrow). In hsp70I:dnBmpr1a-GFP embryos, grem2 expression shifted to ventral regions (yellow arrow) and was reduced dorsally (white arrowhead). Dorsal (D), intermediate (I) and ventral (V) domains of the mandibular (1) and hyoid (2) arches are labeled. Scale bar: 50 μm. (G,H) BRE:GFP expression increased throughout the dorsal and intermediate arch domains of edn1-/- mutants (n=4) compared with wild-type siblings (n=16). EXPRESSION / LABELING:
|
|
Grem2 promotes the dorsal and intermediate facial skeleton. (A) Structure of the grem2 gene and the Grem2:GFP fusion construct. grem2-MO #1 recognizes the ATG start site in Exon 2 and grem2-MO #2 recognizes the 5′ UTR. (B,C) Grem2:GFP fluorescence in uninjected (B) or grem2-MO-#1-injected (C) embryos at 9 hpf. (D,E) Confocal sections of 30 hpf BRE:GFP transgenic embryos. Compared with uninjected controls (D, n=5), injection of grem2-MO-#1 (E, n=8) increased BRE:GFP throughout the mandibular (1) and hyoid (2) arches. (F-H) Confocal sections of in situ hybridizations for hand2 (green) and dlx3b (red). Relative to the first pouch (white outlines), dlx3b and hand2 were mildly expanded in grem2-MO embryos (G, n=45/52) and lost in hsp70I:Gal4; UAS:Grem2 embryos subjected to a 16-17 hpf heat-shock (H, n=16/16). Arrow indicates expanded hand2 expression. (I-M) Ventral views of 5 dpf facial skeletons from control (I), grem2-MO-#1-injected (J), UAS:Grem2 (K), edn1–/– mutant (L) and an edn1–/– mutant injected with grem2-MO-#1 (M). Schematics show skeletal regions derived from dorsal (green), intermediate (red) and ventral (blue) arch domains, with bones lightly shaded. Elements of undefined morphology or derived from the maxillary domain or more posterior arches are grey. Scale bar: 50 μm. |
|
Variable BMP activation and facial skeletal defects in UAS:Bmp4 animals. (A-D) Colorimetric in situ hybridizations show variability of bmp4 upregulation in UAS:Bmp4 embryos subjected to a 20-24 hpf heat-shock after 20 minutes of staining. In hsp70I:Gal4 control embryos, 20 minutes was not enough time for endogenous bmp4 staining to appear (A). Similarly, various classes of GFP upregulation were observed in BRE:GFP; hsp70I:Gal4; UAS:Bmp4 embryos (C,D) compared with BRE:GFP; hsp70I:Gal4 controls (B). Mandibular (1) and hyoid (2) arches are numbered. (E-G) Ventral views of 5 dpf dissected facial skeletons in hsp70I:Gal4 control (E) and a phenotypic series of UAS:Bmp4 larvae (F,G). Scale bars: 50 μm. |
|
CNCC death and skeletal loss in severely affected UAS:Bmp4 embryos. (A,B) Ventral views of 5 dpf whole-mount skeletons in hsp70I:Gal4 control (A) and severely affected UAS:Bmp4 larvae (B). Presumptive high levels of Bmp4 misexpression resulted in reductions of most of the craniofacial skeleton, including ventral mandibular (M) and hyoid (Ch) elements denoted by arrows in (A). (C,D) Confocal projections of control fli1a:GFP (n=3) and UAS:Bmp4; fli1a:GFP (n=7) embryos stained with Lysotracker Red to mark dying cells at 36 hpf. fli1a:GFP (green) marks CNCCs of the pharyngeal arches. Increased numbers of Lysotracker-positive cells were observed in UAS:Bmp4 embryos, and colocalization with fli1a:GFP showed death in both pharyngeal CNCCs and epithelial cells. First and second arch are labeled in control (C); asterisks mark the oral epithelia. |
|
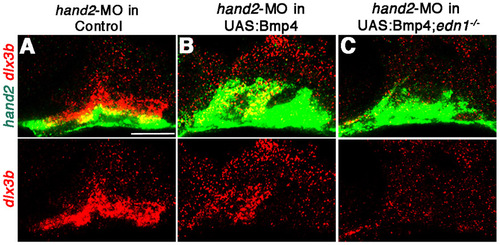
BMPs fail to induce dlx3b in the absence of both Edn1 and Hand2. (A-C) Confocal sections of in situ hybridizations at 36 hpf for dlx3b (red) and hand2 (green). In controls injected with hand2-MO, a slight ventral expansion of dlx3b was seen (A, n=12) compared to un-injected embryos (see Fig. 5A). UAS:Bmp4 embryos subjected to a three-minute heat-shock treatment and injected with hand2-MO show expansion of dlx3b and hand2 (B, n=7), yet no upregulation of dlx3b was observed in UAS:Bmp4; edn1/ embryos injected with hand2-MO (C, n=5). Scale bar: 50 μm. |
|
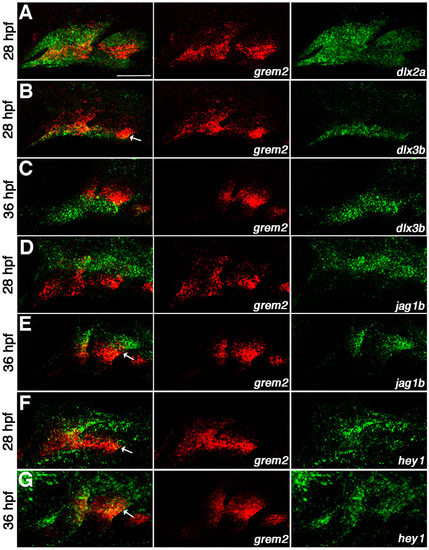
Colocalization of grem2 with arch markers. (A-G) Confocal projections of in situ hybridizations for grem2 (red) with dlx2a (green, A), dlx3b (green, B,C), jag1b (green, D,E) or hey1 (green, F,G) at 28 hpf (A,B,D,F) or 36 hpf (C,E,G). Arrows show partial colocalization of grem2 with dlx3b, jag1b and hey1. Scale bar: 50 μm. |
|
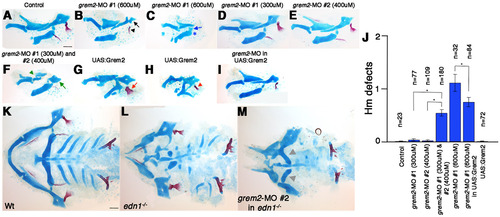
Validation of grem2-MO. (A-I) Unilateral flat-mount dissections of 5 dpf facial skeletons. The moderately affected grem2-MO #1 (600 μM) example (B) displayed a mild Hm defect (black arrow) and Op shape change (black arrowhead), whereas the more severe example (C) showed a severely reduced/transformed Hm (blue arrow) and missing Sy and joint (blue asterisk). grem2-MO #1 (300 μM) (D) and grem2-MO #2 (400 μM) (E) caused no defects on their own, but together (F) caused synergistic skeletal defects, including transformed Hm (green arrow), missing Sy and joint, and altered Pq (green arrowhead). Two examples of hsp70I:Gal4; UAS:Grem2 larvae subjected to a 16-17 hpf heat-shock (G,H) show increasing reduction/transformation of Ch, Sy and joints. The Op was expanded in less severely affected UAS:Grem2 animals (G, red arrow), yet lost in more severely affected animals (H, red arrowhead). (J) Quantification of dorsal Hm defects, scored as: 0, not affected; 1, mildly defective (e.g. B); and 2, transformed/reduced (e.g. C,F). Standard errors of the mean are shown, and asterisks show statistically significant comparisons. (K-M) Ventral views of dissected facial skeletons in wild-type (K), edn1-/- mutant (L) and edn1-/- embryos injected with grem2-MO #2 (400 μM) (L). Grey arrowheads depict rescue of ventral Ch. Scale bars: 50 μm. |