- Title
-
Zebrafish as a model for apolipoprotein biology: Comprehensive expression analysis and a role for ApoA-IV in regulating food intake
- Authors
- Otis, J.P., Zeituni, E.M., Thierer, J.H., Anderson, J.L., Brown, A.C., Boehm, E.D., Cerchione, D.M., Ceasrine, A.M., Avraham-Davidi, I., Tempelhof, H., Yaniv, K., Farber, S.A.
- Source
- Full text @ Dis. Model. Mech.
|
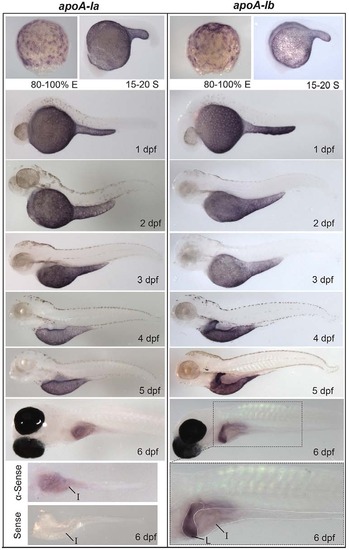
Developmental mRNA expression patterns of apoA-Ia and apoA-Ib. In situ hybridization of apoA-Ia and apoA-Ib during gastrulation [80-100% epiboly (E)], somitogenesis [15-20 somite (S)], and daily until 6 dpf. Both genes localize to the YSL from somitogenesis through 5 dpf, apoA-Ia localizes to the intestine (I) at 6 dpf, and apoA-Ib localizes strongly to the liver (L) and weakly to the intestine at 6 dpf. All zebrafish are wild type except 6-dpf larvae, which are nacre-/-. Larvae studied at 2-5 dpf were treated with PTU to prevent pigment formation. No signal was observed for either gene at the eight-cell stage or blastulation (supplementary material Fig. S1). Experiments were performed three times for each gene at each stage with ne5 embryos or larvae per probe per experiment. EXPRESSION / LABELING:
|
|
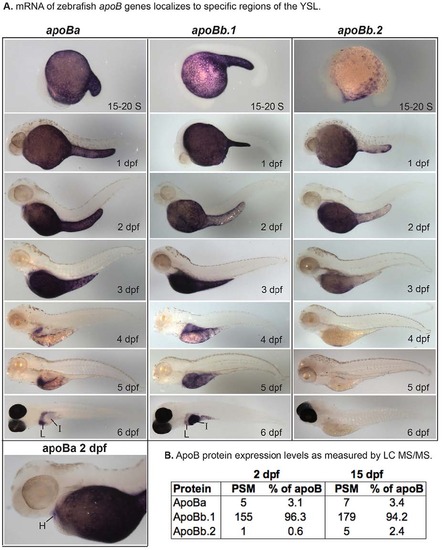
Zebrafish apoB mRNA and protein expression. (A) Localization of apoBa, apoBb.1 and apoBb.2 mRNA by ISH from somitogenesis [15-20 somite (S)] to 6 dpf. All apoB genes are expressed in the yolk syncytial layer (YSL), but mRNAs localize to distinct subregions of the YSL at various developmental stages. apoBa mRNA is present in the heart (H) at 2 dpf. At 6 dpf apoBa mRNA is expressed strongly in the liver (L) and weakly in the intestine (I), apoBb.1 is strongly expressed in the intestine and weakly in the liver, and apoBb.2 is not detectable by ISH. All zebrafish are wild type except 6-dpf larvae which are nacre-/-. Larvae collected at 2-5 dpf were treated with PTU to prevent pigment formation. No signal was observed at the eight-cell stage or at 30% or 80-100% epiboly (supplementary material Fig. S2). Experiments were performed three times for each gene at each stage with ne5 embryos or larvae per probe per experiment. (B) Relative abundance of zebrafish ApoB paralogs as identified by peptide spectrum matches (PSM) in 2- and 15-dpf larvae (20-30 pooled larvae per experiment) as determined by LC-MS/MS of total larval proteins ≥250 kDa. Two experiments were performed for each developmental stage and both experiments yielded similar results. Data presented are from one experiment. EXPRESSION / LABELING:
|
|
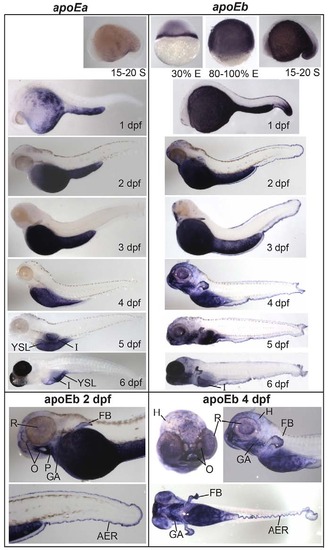
apoEa localization to the YSL and intestine contrasts with widespread expression of apoEb. apoEa and apoEb mRNA expression observed by ISH during blastulation [30% epiboly (E)], gastrulation (80% E), somitogenesis [15-20 somite (S)] and 1-6 dpf. apoEa mRNA is expressed in distinct subregions of the YSL at somitogenesis and 1 dpf, and ubiquitously throughout the YSL from 2-5 dpf. At 6 dpf (but not 5 dpf), apoEa is observed in the intestine (I). apoEb is expressed not only throughout the YSL (30% E to 5 dpf), but also in the tail bud (15-20 S and 1 dpf), the apical ectodermal ridge (AER) (1-6 dpf), the head (H) (1-6 dpf), mouth and nose orifices (O) (2-6 dpf), the find buds (FB) (2-6 dpf), pharynx (P) (2-6 dpf), gill arches (GA) (3-6 dpf), periderm (4-6 dpf), retina (R) (4-6 dpf), intestine (6 dpf) and swim bladder (6 dpf). No expression of apoEa was observed during blastulation or gastrulation, or for either gene at the eight-cell stage (supplementary material Fig. S3). Zebrafish are wild type, except for 6-dpf larvae which are nacre-/-; 1-6 dpf larvae were treated with hydrogen peroxide to remove pigmentation. ISH was performed in triplicate with ne5 embryos or larvae per probe per experiment. EXPRESSION / LABELING:
|
|
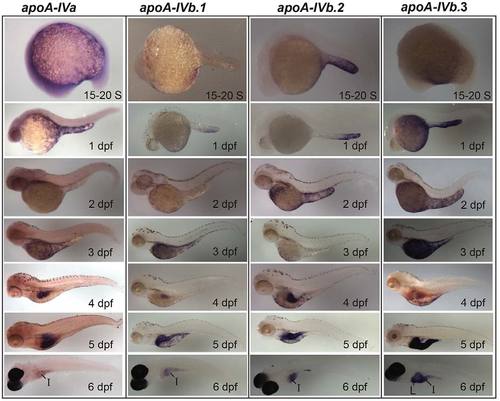
The zebrafish apoA-IV genes have unique expression patterns in the YSL, intestine and liver. Developmental mRNA expression dynamics of the zebrafish apoA-IV genes, as measured by ISH, during somitogenesis [15-20 somite (S)] and at 1-6 dpf. All four apoA-IV genes showed earliest mRNA expression at 15-20 S in the YSL; the mRNAs localize to distinct subregions of the YSL until 5 dpf. All four genes are expressed in the intestine (I) at 4-6 dpf, with weak intestinal expression observed for apoA-IVa, and apoA-IVb.3 is expressed in the liver (L) at 4-6 dpf. No expression was observed for the apoA-IV genes at the eight-cell stage, 30% epiboly or 80-100% epiboly (supplementary material Fig. S4). Larvae from 15-20 S to 5 dpf are wild type (2-5 dpf treated with PTU; 6-dpf larvae are nacre-/-. Experiments were performed in triplicate with ne5 embryos or larvae per probe in every experiment. EXPRESSION / LABELING:
|
|
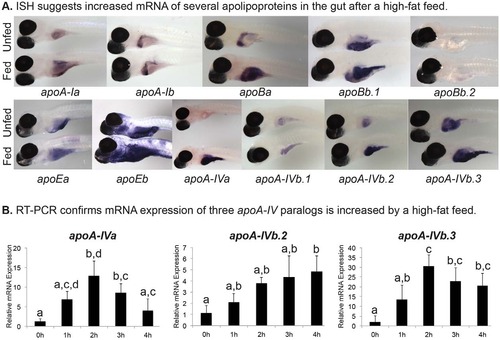
High-fat feed increases expression of apolipoproteins in larval gut. (A) Representative images of mRNA expression as observed by ISH of the zebrafish apoA-I, apoB, apoE and apoA-IV genes in 6-dpf nacre-/- larvae. Larvae have either never eaten exogenous food (Unfed), or have been fed a high-fat, 5% chicken egg yolk meal for 4 hours (Fed). ISH was performed in triplicate with n≥5 larvae; no staining was observed in sense probed (supplementary material Figs S1-S4). (B) A high-fat meal increases zebrafish apoA-IVa, apoA-IVb.2, and apoA-IVb.3 mRNA expression in the gut (intestine, liver, pancreas). Real-time PCR quantification of apolipoprotein transcriptional response in the 6-dpf zebrafish gut to 1, 2, 3 or 4 hours of a 10% chicken egg yolk feed (n=3; 10 pooled larval guts per experiment). Groups with the same letter are not significantly different (one-way ANOVA, P<0.05, n=3). |
|
In situ hybridization analysis of apoA-Ia and apoA-Ib does not reveal maternal transcripts at the 8-cell (C) stage, and no induction of transcription at 30% epiboly (E). No staining is present in zebrafish treated with sense probes at any stage. Larvae are wild type ( 2-5 dpf treated with PTU) and nacre-/- (6 dpf). Experiments were performed in triplicate; n ≥ 5 larvae for each probe in every experiment. S: somite, dpf: days post-fertilization. EXPRESSION / LABELING:
|
|
apoBa, apoBb.1, and apoBb.2 in situ hybridization does not reveal mRNA transcripts at the 8-cell (C) stage, 30% epiboly (E), 80% E. No staining was observed for sense probes. Experiments were performed in triplicate on wild type ( 2-5 dpf treated with PTU) and nacre-/- (6 days post-fertilization (dpf)) larvae; n ≥ 5 larvae for each experiment. |
|
apoEa has no or low expression in 8-cell (C), 30% epiboly (E), or 80% E larvae; apoEb mRNA is not present in 8-C embryos. Sense probes show no staining at any stage (S: somite). In situ hybridization (ISH) was performed in wild type and nacre-/- zebrafish. Larvae from 2 to 6 days post-fertilization (dpf) were treated with hydrogen peroxide. ISH was performed in triplicate; n ≥ 5 larvae for each experiment. |
|
Zebrafish apoA-IVa, apoA-IVb.1, apoA-IVb.2, and apoA-IVb.3 mRNA are not revealed at the 8-cell (C) stage, 30% epiboly (E), or 80% E, as measured by in situ hybridization (ISH). Experiments were performed in triplicate (n ≥ 5 larvae each) on wild type (2–5 dpf PTU treated) and nacre-/- (6 dpf). No staining was observed with sense probes for any of the apoA-IV genes at any stage. S: somite. Data for apoA-IVb.1, apoA-IVb.2 sense and antisense probes and apoA-IVb.3 sense probe at 30% E not shown. |
|
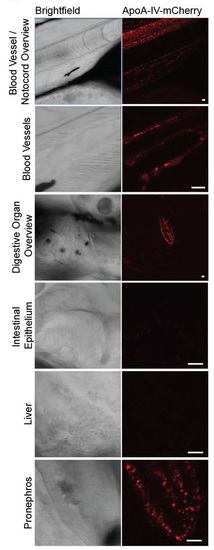
Representative images of Tg(hsp70:apoA-IVb.1:mCherry) larvae ~18 hours post-heat shock. mCherry signal represents mCherry protein; mCherry signal in the digestive organ overview is visible in the pronephros. Larvae are 7 days post-fertilization, anesthetized in tricane and immobilized in 1.2% low melt agarose. Imaging was conducted with a 25X water immersion objective on a Leica SP5 confocal microscope. Scale bars represent 10 microns. EXPRESSION / LABELING:
|