- Title
-
The polycomb group protein ring1b/rnf2 is specifically required for craniofacial development
- Authors
- van der Velden, Y.U., Wang, L., Querol Cano, L., and Haramis, A.P.
- Source
- Full text @ PLoS One
|
Ring1b mutants lack almost all head cartilage elements. Lateral view of WT and ring1b live embryos at 72 hpf (A, B). Alcian-Blue stained head cartilages of WT (C, E, G and H) and ring1b (D, F, H and J) mutants at the indicated developmental points, ventral views. The paired trabeculae have elongated and fused posteriorly in WT embryos at 56 hpf (E) and by 72 hpf the elaborate cartilagenous skeleton of the head has been established (I). In contrast, no cartilage is visible in ring1b mutants except for two minute cartilage deposits at 72 hpf ring1b mutants (J: arrowheads). ch: ceratohyal; ep: ethmoid plate; hys: hyosymplectic; m: Meckel’s cartilage; pc: parachordal, pq: palatoquadrate; tc: trabeculae. PHENOTYPE:
|
|
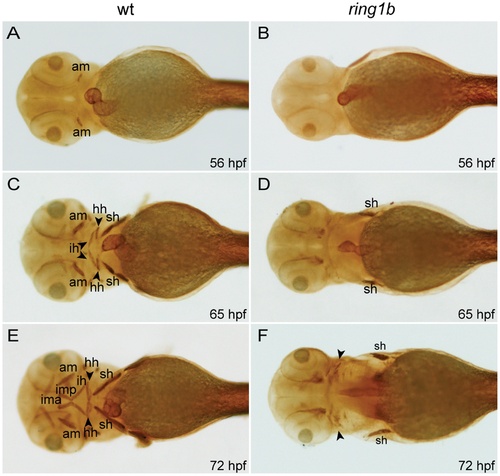
Cranial musculature development is severely impaired in ring1b mutants. Ventral views of WT and ring1b embryos stained with the MF20 antibody (A-F). The anterior mandibularis (am) has not formed in ring1b mutants at 56 hpf (B), the sternohyoideus (sh) is reduced at 65 hpf (D) and at 72 hpf, cranial muscles are almost completely absent (F). hh: hyohyoideus; ih: interhyoideus; ima: intermandibularis anterioris; imp: intermandibularis posterioris. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Migration and initial differentiation of trunk neural crest is largely unaffected in ring1b mutants. Sox10 staining of WT and ring1b mutants at 24 and 32 hpf shows that sox10 expressing NC cells have migrated timely and correctly to their ventral positions (A–D). Expression of foxD3 in cranial ganglia-associated glia is comparable to WT siblings in ring1b mutants at 32 hpf (E, F), but reduced in the postotic ganglia of ring1b mutants at 56 hpf (G, H). NeuroD expression in cranial ganglia precursors is reduced in ring1b mutants at 32 hpf (I, J), but remains detectable in the hindbrain region of ring1b mutants at 56 hpf (L). Lateral view of 32 hpf embryos shows normal distribution of melanocytes in ring1b mutants at 32 hpf (M, N). At 60 hpf, ring1b melanophores remain more stellate than WT melanophores, which have started to round up (S, T). Iridophores are present in near normal numbers in ring1b mutants at 60 hpf (O, P), whereas ring1b xanthophores are smaller and less stellate (Q, R, U, V). Abbreviations: gX: vagal ganglia; mll: medial lateral line ganglia; o/gVIII: octaval/statoacustic ganglia; pll/gP: posteriolateral line ganglia; pog: preotic ganglia; ptg: postotic ganglia; tg: trigeminal ganglia. |
|
Cranial neural crest cells migrate into the pharyngeal arches of ring1b mutants. Dorsal (A–H) and lateral (A′–H′) views of WT and ring1b mutants at 32 hpf stained with the indicated genes. Pre-chondrogenic gene expression in CNCs is largely unaffected in ring1b mutants. Dlx2a is expressed at normal levels in the pharyngeal arches of ring1b mutants (A–B′). Separation of dlx2a-positive CNCs into distinct groups of cells that populate the posterior arches is slightly affected in ring1b mutants A–B′). Hand2 expression in the ring1b pharyngeal arches is identical to that of WT siblings (C–D′). Hoxa3a expression is reduced in the ring1b posterior pharyngeal arches (E–F′). Expression of the pharyngeal pouch marker cyp26a1 is severely reduced in ring1b mutants (G–H′). |
|
Impaired formation of prechondrogenic condensations and abolished cartilage differentiation in ring1b mutants. Ventral (A–J) and lateral (A′–J′) views of WT and ring1b mutants at 50 hpf stained with the indicated genes. Prechondrogenic condensation markers sox9a and sox9b are expressed in ring1b CNCs residing in pharyngeal arches 1 and 2, albeit the expression domain is smaller (A–D′). Similarly, the expression domain of CNC markers sox10 and dlx2a is reduced in ring1b pharyngeal arches 1 and 2 (E–H′). Expression of sox9a, sox9b and sox10 is not detected at the presumptive location of the trabeculae in ring1b mutants (B, B′, D, D′, F, F′). Col2a1 is expressed in WT trabeculae and pharyngeal arches I–II, but expression is abolished in ring1b mutants (I–, J′). Note that expression of col2a1, a marker for differentiating chondrocytes, is normal in the otic vesicles and parachordals of ring1b mutants. tc: trabeculae cranii; pc: parachordal; ov: otic vesicle. Numbers indicate the respective pharyngeal arches. EXPRESSION / LABELING:
PHENOTYPE:
|
|
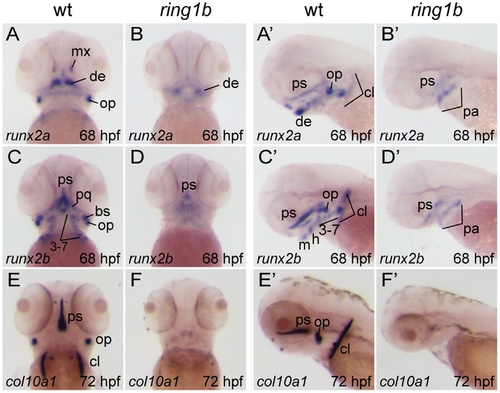
Loss of endochondral and dermal ossification in ring1b mutants. Ventral (A–D, E, F) and lateral (A′–D′, E′,) views of in situ hybridizations with riboprobes against the indicated genes in WT and ring1b mutants at 68–72 hpf. In WT embryos, runx2a and runx2b are expressed in hypertrophic pharyngeal arch-derived chondrocytes, as well as in the dermal ossification centers of the operculum, parasphenoid and cleithrum. Weak expression is detected in pharyngeal arches and the parasphenoid of ring1b mutants (B, B′, D, D′). At 72 hpf, col10a1 is expressed in developing dermal bones in WT embryos, but not in ring1b mutants (E–F). cl: cleithrum; de: dentary; h: hyoid; m: mandibular; mx: maxilla; pa: pharyngeal arches; pq: palatoquadrate; ps: parasphenoid, op: operculum cl: the cleithrum. Numbers indicate the respective pharyngeal arches. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Apoptosis is slightly increased in the pharyngeal arch of ring1b mutants. Lateral views of WT and ring1b embryos at 36 hpf stained for TUNEL. In WT embryos two small clusters of TUNEL-positive apoptotic cells were detected in the pharyngeal arch region just posterior to the eye (A, arrows). These clusters appear to contain more apoptotic cells in the ring1b mutants (B, arrows). The arrowhead indicates the otic vesicle (ov). WT embryos at 36 hpf contain few apoptotic cells in the trunk (C), whereas there is an increase in overall apoptosis particularly in the trunk and the tail in ring1b mutants (D, arrows). PHENOTYPE:
|
|
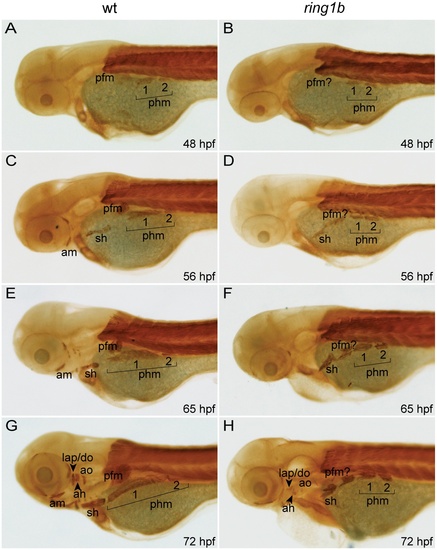
Cranial musculature development is severely impaired in ring1b mutants. Lateral views of embryonic musculature in WT and ring1b mutants (A–H). The posterior hypaxial muscle (phm), had delaminated from the somites in both WT and ring1b mutants at 48 hpf (A, B). At 56 hpf, the sternohyoideus (sh) is formed in both WT and ring1b mutants and the pectoral fin muscle is prominently visible (C, D). During later development, the sh and phm elongated and attached to the cleithrum, a bone of the fin girdle in WT embryos, (E, G). Elongation of these muscles was completely abrogated in ring1b mutants (F, H). Cranial musculature is almost completely absent in ring1b mutants (H). Abbreviations: am: anterior mandibularis; ah: adductor hyomandibulae; ao: adductor opercule; do: dilator operculi; hh: hyohyoideus; ih: interhyoideus; ima: intermandibularis anterioris; imp: intermandibularis posterioris, lap: levator arcus palatine; phm: posterior hypaxial muscle; pfm: pectoral fin muscle; sh: sternohyoideus. |
|
Persistence of apoptotic cells in ring1b mutants. Ventral views of WT and ring1b embryos at 48 hpf stained with Acridine Orange. No apoptotic cells are detected in the pharyngeal arch region of WT embryos (A) whereas few AO-positive apoptotic cells have persisted in the ring1b mutants (B) Arrows in (B) indicate apoptotic clusters in the prospective jaw region and anteriorly to the otic vesicle. Figure also shows images of live WT and ring1b embryos depicting the fin and pharyngeal cartilages in WT (C, arrows) and their absence in the ring1b mutants (D, *). |