- Title
-
Requirement for frzb and fzd7a in cranial neural crest convergence and extension mechanisms during zebrafish palate and jaw morphogenesis
- Authors
- Kamel, G., Hoyos, T., Rochard, L., Dougherty, M., Tse, W., Shubinets, V., Grimaldi, M., and Liao, E.C.
- Source
- Full text @ Dev. Biol.
|
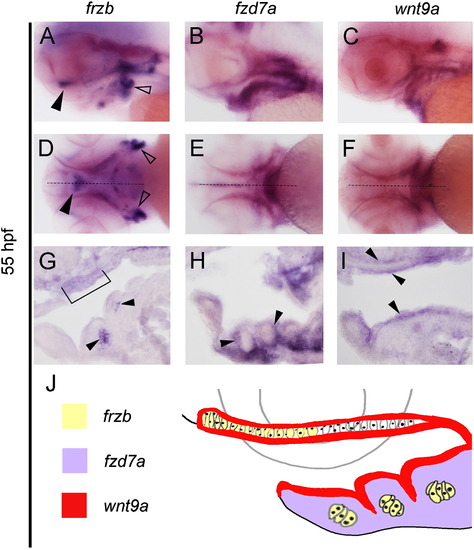
Expression patterns of wnt9a, frzb, and fzd7a within pharyngeal structures. (A–K) Whole-mount in situ hybridization with embryos oriented with head towards the left in lateral view (A, B, C), ventral views (D, E, F) and sagittal sections (G, H, I). Midline sagittal section plane is indicated by dotted line in D–F. frzb transcripts localize to the chondrocytes located in the distal portion of the palate (arrow in A, bracket in G), lower jaw (arrow in G) and lateral ceratobrachials (open arrows in A and D). Expression of fzd7a is restricted to the neural crest tissue surrounding the chondrocytes (B, E, arrows in H). wnt9a is expressed in oropharyngeal ectoderm, and in the tissue above the palate (C, F, arrows in I). Diagram summary of the expression domains and their spatial relationship (J). EXPRESSION / LABELING:
|
|
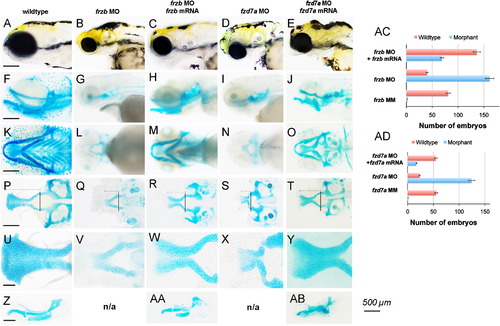
Depletion of frzb and fzd7a disrupts craniofacial development. (A–Y) Live embryo morphology (A–E) and Alcian blue cartilage stain (F-AB). Lateral view (A–J), ventral view (K–O), ventral view with lower jaw dissected away (P–T), taken at 10× magnification, scale bar 500 µm A–L, 100 μm M–P. View of the palate flat-mounted in ventral view (U–Y) and the lower jaw flat-mounted in lateral view (Z-AB), taken at 40× magnification. Anterior is to the left in all views. Wildtype embryos (A, F, K, P, U and Z), injected with frzb translation-blocking morpholino alone (MO) (B, G, L, Q and V), co-injection of frzb MO and non-complementary frzb mRNA (C, H, M, R, W and AA), embryos injected with fzd7a translation-blocking morpholino (MO) (D, I, N, S and X), and co-injection of fzd7a MO with non-complementary fzd7a mRNA (E, J, O, T, Y and AB) at 4.5 dpf. Normal craniofacial morphology (A) and craniofacial structures were seen in wildtype embryos, with normal lower jaw (F,K, Z) and palate (P,U). Knockdown of frzb and fzd7a produced smaller craniofacial structures grossly and narrow mouth openings (B, D). In frzb and fzd7a morphants, the proximal trabeculae form and converge at the midline, but fail to elongate and the palate does not form (Q, S). The chondrocytes in the leading edge of the trabeculae in frzb and fzd7a morphants appear dysmorphic and rounded and fail to adopt an organized intercalated pattern compared to wildtype (V,X). In frzb and fzd7a morphants, the lower jaw is absent and undetected by Alcian blue cartilage staining (G,I,L,N). Graphs summarize the representative phenotypes, where the total number of embryos scored for wildtype (red bar) and morphant phenotypes (blue bar) are shown with 5% error bars (AC,AD). Injection of non-complementary m RNA of frzb and fzd7a to rescue frzb MO and fzd7a, respectively, were statistically significant. PHENOTYPE:
|
|
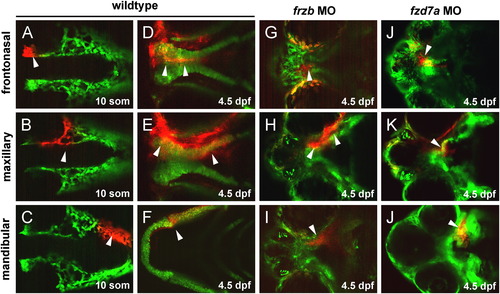
Directed migration of CNCC proceed in frzb and fzd7a morphants but fail to undergo morphogenesis. Migration of the frontonasal CNCCs (A–D), maxillary CNCCs (E–H) and mandibular CNCCs (I–L). Anterior is to the left in all views. Photoconversion was performed unilaterally, with the contralateral side serving as internal control. CNCCs anterior to the eye at 14-somites stage (A) migrate to form the frontonasal process at 4.5 dpf (D), CNCCs medial to the eye at 14-somites (arrow, B) migrate to form the maxillary processes at 4.5 dpf (arrows, E), and CNCCs at 14-somites posterior to the eye (arrow, C) form the mandible at 4.5 dpf (arrow, F). In frzb and fzd7a morphants, CNCCs destined to form the frontonasal process migrated to the median portion of the dysmorphic and shortened palate (arrows, G, J). In frzb and fzd7a morphants, CNCCs destined to form the maxillary process converged in the midline (H, K), while CNCCs destined to form Meckel′s cartilage migrated to form the mandibular prominence but failed to converge in the midline (arrows, I, L). EXPRESSION / LABELING:
PHENOTYPE:
|
|
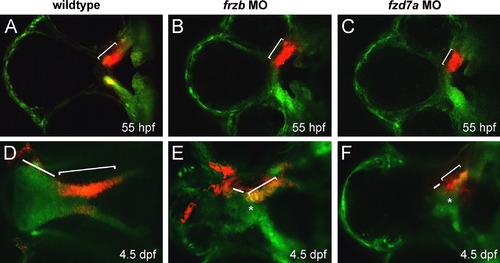
frzb and fzd7a are required for palatal extension. Anterior is to the left in all views. (A–F) Photoconversion was performed unilaterally, with the contralateral side serving as internal control. Wildtype trabeculae/maxillary prominence was photoconverted at 55 hpf and followed through 4.5 dpf, where the paired maxillary process CNCCs converge in the midline (asterisk) and extend (bracket) with distal proliferative front (bar). In frzb and fzd7a morphants, paired maxillary process converged in the midline (asterisk in E, F) but failed to extend (brackets in E and F are shorter than in D) and the proliferative domain is diminished in the distal portion of the palate (bars in E and F are shorter than in D). |
|
frzb and fzd7a are required for chondrocyte proliferation. Anterior is to the left and all images are in ventral view. (A–F) EdU staining for new cell division from 55–60 hpf, a period of early maxillary extension, revealed a proliferative front of chondrocytes at the leading edge of the developing palate during normal development (A, arrows, dotted line) and the paucity of new cell division during this same period in frzb (B) and fzd7a morphants (C). TUNEL assay for apoptosis demonstrated that the shortened palate is not a result of increased cell death (D–F). |
|
wnt9a, frzb and fzd7a are required for bapx1 expression. Dorsal views of embryo at 10-somites (A′–P′) and lateral views of embryo at 24 hpf (A–L) and 55 hpf (M–T). Wholemount RNA in situ hybridization for sox10 (A′–D′), sox9a (E′–H′), foxd3 (I′–L′) and dlx2a (M′–P′) in wildtype, wnt9a, frzb and fzd7a morphants; edn1 (A–D), endrA2 (E–H), dlx2a (I–L) sox9a (M–P) and bapx1 (Q–T) in wildtype, wnt9a, frzb and fzd7a morphants. Quantitative PCR for edn1, ednrA2, dlx2a in wildtype, wnt9a, frzb and fzd7a morphants at 24 hpf (U–W) and bapx1 at 55 hpf (X). Knockdown of wnt9a, frzb and fzd7a did not alter neural crest marker expression early in development (A′–P′). Similarly, edn1, ednrA2 and dlx2a expression (A–L) was unaffected in all morphants. Quantitative PCR analysis of whole embryos further confirms normal transcript levels at 24 hpf (U–W). Expression patterns of sox9a at 55 hpf in wnt9a, frzb and fzd7a morphants confirm the presence of CNCC mesenchyme within the first and second pharyngeal arches (M–P). However, the expression of bapx1 is reduced both qualitatively and quantitatively in wnt9a, frzb and fzd7a morphants at 55 hpf (Q–T, X). EXPRESSION / LABELING:
|
|
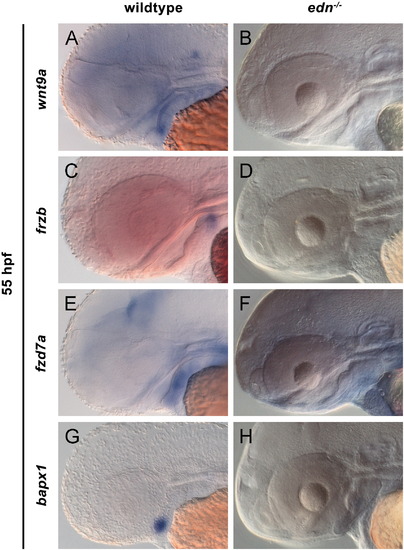
edn1 is required for expression of wnt9a, frzb and fzd7a. Lateral views at 55 hpf (A–F). Wholemount RNA in situ hybridization for wnt9a (A, B), frzb (C, D), fzd7a (E, F) and bapx1 (G, H) in wildtype and edn1-/- mutants. Expression of wnt9a, frzb and bapx1 was absent in the developing pharyngeal arches (B, D, H), while fzd7a expression was strongly reduced in both the forebrain and arches (F) in edn1-/- mutants. |
|
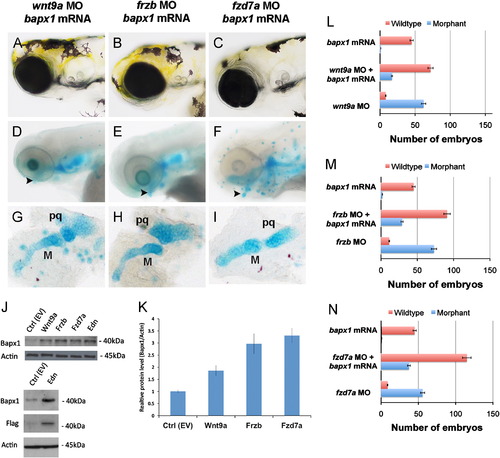
Overexpression of bapx1 partially rescues lower jaw elements in wnt9a, frzb and fzd7a morphants. Live embryo morphology (A–C) and Alcian blue cartilage stain (D–F) at 10× magnification, lower jaw flatmount at 20× magnification (G–I). Anterior is to the left in all images. Overexpression of bapx1 mRNA in wnt9a, frzb and fzd7a morphants partially rescued distal palatoquadrate and Meckel′s cartilage elements (arrowhead, D–F, G–I). Graphs summarize the representative phenotypes, where the total number of embryos scored for wildtype (red bar) and morphant phenotypes (blue bar) are shown with 5% error bars (L–N). Overexpression of wnt9a, frzb and fzd7a in HEK293T cells increased Bapx1 protein production as shown by Western blot (J) where actin serves as an internal control and Flag represents mammalian expression vector tag. Bapx1 expression was increased in a statistically significant manner, P<0.05 (K). Abbreviations: M (Meckel′s cartilage), pq (palatoquadrate) |
Reprinted from Developmental Biology, 381(2), Kamel, G., Hoyos, T., Rochard, L., Dougherty, M., Tse, W., Shubinets, V., Grimaldi, M., and Liao, E.C., Requirement for frzb and fzd7a in cranial neural crest convergence and extension mechanisms during zebrafish palate and jaw morphogenesis, 423-33, Copyright (2013) with permission from Elsevier. Full text @ Dev. Biol.