- Title
-
Zebrafish msxB, msxC and msxE function together to refine the neural-nonneural border and regulate cranial placodes and neural crest development
- Authors
- Phillips, B.T., Kwon, H.J., Melton, C., Houghtaling, P., Fritz, A., and Riley, B.B.
- Source
- Full text @ Dev. Biol.
|
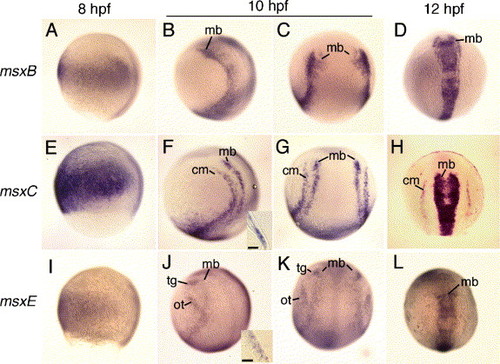
Expression of msxB, msxC, and msxE. Wild type embryos at 8 hpf (A, E, I), 10 hpf (B, C, F, G, J, K) and 12 hpf (D, H, L) showing msxB expression (A–D), msxC expression (E–H), and msxE expression (I–L). The inset in panel F shows a cross section through the cranial mesoderm (cm) domain of msxC. The inset in panel J shows a cross section through the otic (ot) domain of msxE, where expression is limited to the ectoderm. Scale bars in the insets = 10 μm. Abbreviations: cm, cranial mesoderm; mb, midbrain; ot, otic; tg, trigeminal. Anterior is to the top in all specimens. Panels A, B, E, F, I, J show lateral views with dorsal to the right. Panels C, D, G, H, K, L show dorsal views. EXPRESSION / LABELING:
|
|
Expression of msx genes relative to preplacodal and neural markers. (A, B) Expression of msxB (black) and dlx3b (red) at 11 hpf. Panel B is a fluorescent enlargement of the left otic placode of the specimen in panel A. Brackets show extent of overlap in gene expression. (C, D) Comparison of preotic domains of msxE (C) and pax8 (D) at 10 hpf. Expression of krox20 (red) is shown to facilitate alignment of embryos, and the preotic placode is outlined (dashed white line). (E) Expression of msxC (black) and dlx3b (red) at 10 hpf. The cranial mesoderm domain of msxC is not visible in this image. (F, G) Expression of msxC (black) and dlx3b (red) at 12 hpf. Panel G is a fluorescent enlargement of the right otic placode of the specimen in panel F. (H) expression of msxC (black) and krox20 (red) at 10 hpf. Abbreviations: mb, midbrain; op, otic placode. All specimens are dorsal views with anterior to the top except for panel C which is a lateral view with anterior to the top and dorsal to the right. Scale bars, 50 μm. EXPRESSION / LABELING:
|
|
Disruption of msx. (A–F) Lateral views of live embryos at 28 hpf showing the whole embryo and an enlargement of the midbrain–hindbrain region. (A) Wild-type, (B) x8/x8 mutant, (C) msxB morphant, (D) msxC morphant, (E) msxE morphant, and (F) msxBCE morphant. Regions of the brain with a darkened granular appearance are indicative of elevated cell death (cd). (G) Schematic of general intron–exon structure of msx genes and mature mRNAs. Splice-blocking morpholinos were designed to bind to splice donor sites as shown (msx-MO). For each gene, PCR primers (arrows) were designed to amplify exon1 plus the 52 end of exon2 (primers 1 and 2) or the entire coding region (primers 1 and 3). (H) Analysis of morpholino efficacy. Primers for the indicated genes (left) were used to amplify RT-PCR products from lysates of embryos injected with the indicated morpholinos (top). All morpholinos are highly specific and affect only the targeted transcript. The effects of msxE-MO were the strongest, with no detectable PCR products from either primer set. In the case of msxC-MO, amplification of the entire coding region (c1/c3 primers) yielded no signal corresponding to the mature mRNA, although a weak band was detected at a higher molecular weight corresponding to unspliced message (not shown). This residual msxC transcript presumably accounts for the appearance of a weak band corresponding to exon1 (c1/c2 primers). msxB-MO gave a more complex pattern: Primers b1 and b2 gave only a faint band of the expected size whereas amplification of the entire coding region (primers b1 and b3) gave three faint bands, one corresponding to mature mRNA and two with lower molecular weight (not shown). The latter presumably reflect activation of cryptic splice sites. odc expression was used as a constitutive control (Draper et al., 2001). |
|
Placode development in msxBCE morphants. (A, D) foxe4 expression at 14 hpf in a control embryo (A) and msxBCE morphant (D). (B, E) Lens and nasal pit (np) at 28 hpf in a control embryo (B) and msxBCE morphant (E). (C, F) NCAM as revealed by zn12 antibody staining in trigeminal ganglia (tg) at 24 hpf in a control embryo (C) and msxBCE morphant (F). (G, L) pax8 expression at 10 hpf in a control embryo (G) and msxBCE morphant (L). The prospective otic placode (op) is indicated. (H, I, M, N) pax2a expression at 12 hpf in a wild-type control (H), msxBCE morphant (M), a b380/b380 mutant (I), and a b380/b380 mutant injected with msxBCE-MO (N). The otic placode domain (op) or the corresponding region in the b380/b380 mutant (asterisk) is indicated. Dilute probes for krox20 and myoD were included to help identify b380/b380 mutant embryos (by virtue of their fused somites) and to visualize the hindbrain. (J, K, O, P) Pax2 antibody staining of otic placode in 12 hpf control (J), 14 hpf control (K), 12 hpf msxBCE morphant (O) and 14 hpf msxBCE morphant (P). Numerous pax2a-expressing cells have failed to converge into the otic placode by 14 hpf in the msxBCE morphant (asterisks). (Q, U) Expression of pax5 in the otic vesicle at 24 hpf in a control embryo (Q) and msxBCE morphant (U). (R, V) Expression of otx1 in the otic vesicle at 24 hpf in a control embryo (R) and msxBCE morphant (V). (S, W) msxC expression in the otic vesicle at 72 hpf in a control embryo (S) and msxBCE morphant (W). The edges of the otic vesicle are outlined. Although morpholino injection impairs accumulation of mature mRNA, msxC transcripts can still be detected by prolonging the enzymatic reaction following in situ hybridization. Positions of the anterior crista (ac), lateral crista (lc) and posterior cristae (pc) are indicated. (T, X) Dorsal view of otic vesicle at 75 hpf in a control embryo (T) and msxBCE morphant (X). Semicircular canals (ssc) are indicated. Images show dorsal views with anterior to the top (A, D, H, I, M, N) and medial to the right (J, K, O, P, Tm, X), lateral views with dorsal to the top and anterior to the left (B, C, E, F, Q–S, U–W) or lateral views with dorsal to the right and anterior to the top (G, L). Scale bar, 50 μm. EXPRESSION / LABELING:
PHENOTYPE:
|
|
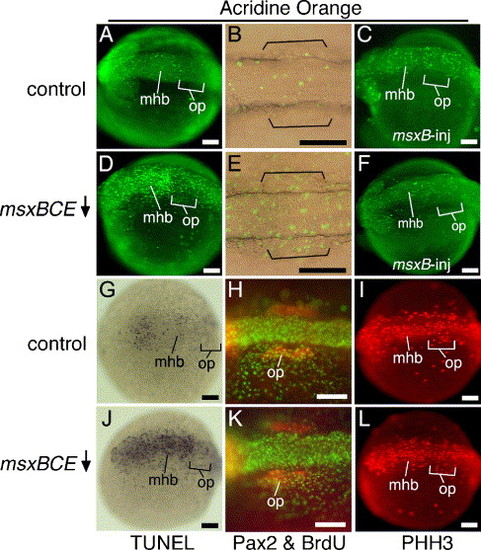
Cell division and cell death in msxBCE morphants. (A–F) Vital staining with acridine orange (AO) in wild-type embryos at 14 hpf (A) and 15 hpf (B), a 14 hpf wild-type embryo injected with msxB plasmid (C), msxBCE morphants at 14 hpf (D) and 15 hpf (E), and a 14 hpf msxBCE morphant injected with msxB plasmid (F). Images in panels B and E show transmitted light plus fluorescence to show otic placode morphology (brackets) in relation to AO-staining. (G, J) Wholemount TUNEL staining at 14 hpf in a control embryo (G) and msxBCE morphant (J). (H, K) Staining at 14 hpf for BrdU (green) and Pax2 (red) in a control embryo (H) and msxBCE morphant (K). (I, L) Staining at 14 hpf for Phosphohistone H3 (PHH3) in a control embryo (I) and msxBCE morphant (L). All images show dorsal or dorsolateral views with anterior to the left. Abbreviations: mhb, midbrain–hindbrain border; op, otic placode. Scale bars, 100 μm. EXPRESSION / LABELING:
|
|
Impaired neural crest development in msxBCE morphants. (A, F) foxd3 expression at 11 hpf in a control embryo (A) and msxBCE morphant (F). (B, G) snail2 expression at 11 hpf in a control embryo (B) and msxBCE morphant (G). (C, H) hoxb3 expression at 14 hpf in a control embryo a control embryo (C) and msxBCE morphant (H). Arrowhead shows neural crest cells migrating from the hindbrain. (D, I) Anti-Pax7 staining at 24 hpf in a control embryo (D) and msxBCE morphant (I). (E, J) Staining with 39.4D5 antibody reveals Islet1 and 2 in the trunk region at 19 hpf in a control embryo (E) and an msxBCE morphant (J). Rohon–Beard sensory neurons (rb) and primary motoneurons (pmn) are indicated. (K–O) Alcian blue cartilage staining at 75 hpf embryos knocked down for the indicated genes. Abbreviations: d, dorsal palate; g, gill arches; m, mandibular cartilage; pm, prechordal mesoderm. Images show dorsal views with anterior up (A–C, F–H), lateral views with dorsal up and anterior to the left (D, E, I, J) and ventral views with anterior to the left (K–O). EXPRESSION / LABELING:
PHENOTYPE:
|
|
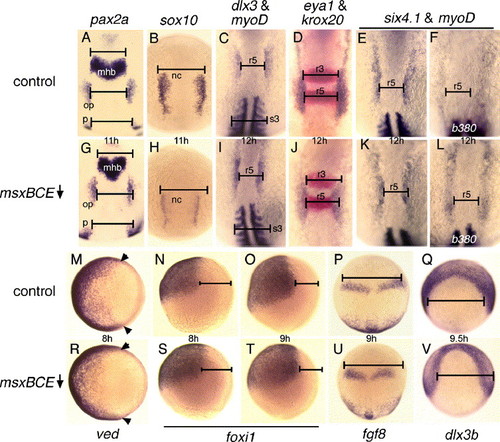
Positioning of the neural border is regulated by opposing activities of msx and dlx genes. (A, G) pax2a expression in the otic placode (op), midbrain–hindbrain border (mhb) and pronephros (p) at 11 hpf in a control embryo (A) and msxBCE morphant (F). (B, H) sox10 expression in neural crest (nc) at 11 hpf in a control embryo (B) and msxBCE morphant (H). (C, I) Expression of dlx3b and myoD at 12 hpf in a control embryo (C) and msxBCE morphant (I). (D, J) Expression of eya1 (blue) and krox20 (red) at 12 hpf in a control embryo (D) and msxBCE morphant (J). (E, F, K, L) Expression of six4.1 and myoD at 12 hpf in a wild-type embryo (E), msxBCE morphant (K), a b380/b380 mutant (F), and a b380/b380 mutant injected with msxBCE-MO (L). b380 mutants were unambiguously identified by the absence of clearly defined somite boundaries. (M, R) Animal pole views showing expression of the ventral marker ved (Shimizu et al., 2002) at 8 hpf in a control embryo (M) and msxBCE morphant (R). Arrowheads mark the dorsal limit of expression. (N, O, S, T) Lateral views of foxi1 expression in ventral ectoderm in control embryos at 8 hpf (N) and 9 hpf (O) and msxBCE morphants at 8 hpf (S) and at 9 hpf (T). (P, U) Expression of fgf8 in the hindbrain at 9 hpf in a control (P) and msxBCE morphant (U). (Q, V) Expression of dlx3b in preplacodal ectoderm at 9.5 hpf in a control (Q) and msxBCE morphant (V). To facilitate comparisons, black bars show the normal width (as measured in control embryos) of the indicated regions. Bars are aligned on the left side but extend past the structures on the right in msxBCE morphants, revealing the degree of neural plate reduction. Abbreviations: mhb, midbrain–hindbrain border; nc, neural crest; op, otic placode; p, pronephros; r3, rhombomere 3; r5, rhombomere 5; s3, somite 3. Unless otherwise indicated, images show dorsal views with anterior to the top. EXPRESSION / LABELING:
|
|
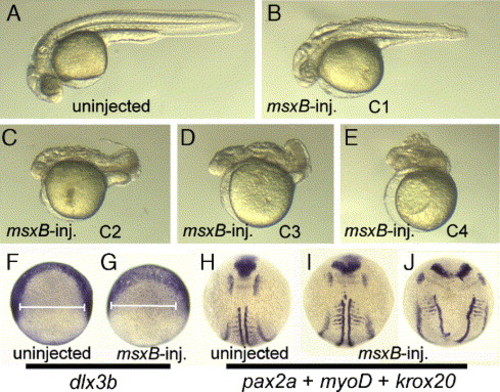
Dorsalization caused by misexpression of msxB. (A–E) Lateral views of a live embryos at 28 hpf showing a control embryo (A) or msxB-injected embryos showing varying degrees of dorsalization (B–E). Dorsalization indices (C1–C4, after Nguyen et al., 1998) are indicated. (F, G) Dorsal views of dlx3b expression at 9.5 hpf in a control embryo (F) and an embryo injected with msxB plasmid (G). (H–J) Dorsal views expression of pax2a, myoD and krox20 at 11.5 hpf in a control embryo (H) and embryos injected with msxB plasmid (I, J). EXPRESSION / LABELING:
|

Unillustrated author statements EXPRESSION / LABELING:
PHENOTYPE:
|
Reprinted from Developmental Biology, 294(2), Phillips, B.T., Kwon, H.J., Melton, C., Houghtaling, P., Fritz, A., and Riley, B.B., Zebrafish msxB, msxC and msxE function together to refine the neural-nonneural border and regulate cranial placodes and neural crest development, 376-390, Copyright (2006) with permission from Elsevier. Full text @ Dev. Biol.