- Title
-
Gon4l regulates notochord boundary formation and cell polarity underlying axis extension by repressing adhesion genes
- Authors
- Williams, M.L.K., Sawada, A., Budine, T., Yin, C., Gontarz, P., Solnica-Krezel, L.
- Source
- Full text @ Nat. Commun.
|
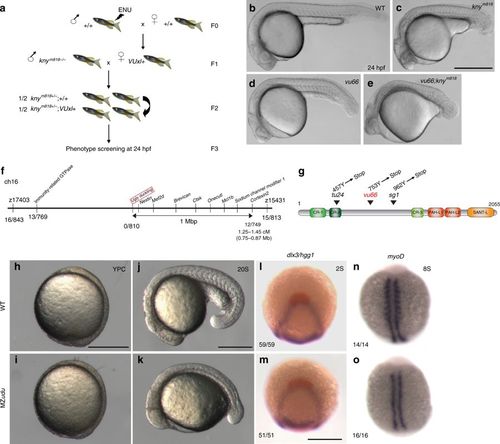
A forward genetic screen identifies ugly duckling(udu)/gon4l as a regulator of axis extension in zebrafish embryos. a Schematic of a synthetic screen to identify enhancers of the short axis phenotype in knym818/m818 zebrafish mutants. b–e Phenotypes at 24 hpf: wild type (WT) (b), knym818/m818 (c), vu66/vu66 (d), knym818/m818; vu66/vu66 compound mutants (e). Images are representative of phenotypes observed at Mendelian ratios in multiple independent clutches. f Diagram of mapping vu66 mutation to Chromosome 16. Bold numbers below specify the number of recombination events between vu66 and the indicated loci. g Diagram of the Gon4l protein encoded by the udu locus. Arrowheads indicate residues mutated in vu66 and other described udu alleles. h–k Live WT (h, j) and maternal zygotic (MZ)udu (i, k) embryos at yolk plug closure (YPC) (h–i) and 20 somite stage (j–k). 100 percent of MZudu mutants from more than 15 germline-replaced females exhibited the pictured phenotypes. l–m Whole mount in situ hybridization (WISH) for dlx3 (purple) and hgg1 (red) in WT (l) and MZudu−/− (m) embryos at two-somite stage. n–o WISH for myoD in WT (n) and MZudu−/− (o) embryos at eight-somite stage. Anterior is to the left in b–e, j–k; anterior is up in h–i, l–o. Fractions indicate the number of embryos with the pictured phenotype over the number of embryos examined. Scale bar is 500 μm in b–e and 300 μm in h–o EXPRESSION / LABELING:
|
|
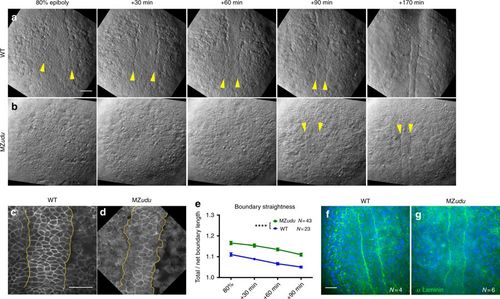
MZudu mutant gastrulae exhibit irregular notochord boundaries. a–b Still images from live Nomarski time-lapse series of the dorsal mesoderm in WT (a) and MZudu−/− embryos (b) at the time points indicated. Images are representative of over 40 MZudu−/− gastrulae. Arrowheads indicate notochord boundaries. c–d Live confocal microscope images of representative WT (c, N = 23) and MZudu−/− (d, N = 43) embryos expressing membrane Cherry. Yellow lines mark notochord boundaries. e Quantification of notochord boundary straightness in live WT and MZudu−/− gastrulae throughout gastrulation. Symbols are means with SEM (two-way ANOVA, ****p < 0.0001). f–g Confocal microscope images of immunofluorescent staining for pan-Laminin in WT (f) and MZudu−/− (g) embryos at two-somite stage. N indicates the number of embryos analyzed. Scale bars are 50 μm. Anterior is up in all images EXPRESSION / LABELING:
PHENOTYPE:
|
|
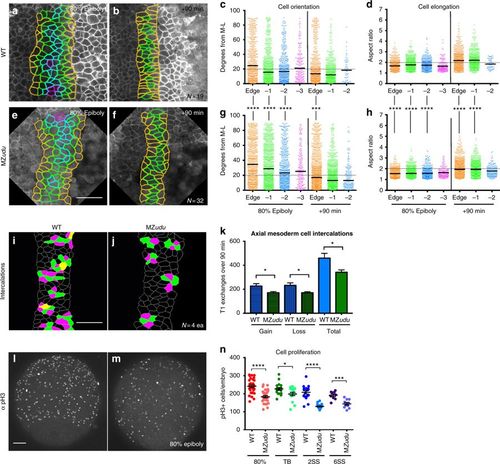
Mediolateral cell polarity and cell intercalations are reduced in the axial mesoderm of MZudu−/− gastrulae. a–b, e–f Still images from live time-lapse confocal movies of the axial mesoderm in WT (a, b) and MZudu−/− (e, f) gastrulae at the time points indicated. Cell outlines are colored according to a cell’s position with respect to the notochord boundary. c, g Quantification of axial mesoderm cell orientation at 80% epiboly (left side) and +90 min (right side) time points. Each dot represents the orientation of the major axis of a single cell with respect to the embryonic ML axis and is colored according to that cell’s position with respect to the notochord boundary (as in images to the left). Bars indicate median values. Asterisks indicate significant differences between WT and MZudu−/− (Kolmogorov–Smirnov test, ***p < 0.001, ****p < 0.0001). d, h Quantification of axial mesoderm cell elongation at 80% epiboly (left side) and +90 min (right side) time points. Each dot represents the aspect ratio of a single cell and is color-coded as in c. Bars indicate mean values. Asterisks indicate significant differences between WT and MZudu−/− (Mann–Whitney test, ****p < 0.0001). i–j Cell intercalations detected in the axial mesoderm of WT (i) and MZudu−/− gastrulae (j). Cells gaining contacts with neighbors are green, cells losing contacts are magenta, and cells that both gain and lose contacts are yellow. k Quantification of cell intercalation events (T1 exchanges) in WT (blue bars) and MZudu−/− gastrulae (green bars) over 90 min. N indicates the number of embryos analyzed. Bars are means with SEM (T-test, *p < 0.05). l–m 200 μm confocal Z projections of immunofluorescent staining for phosphorylated Histone H3 (pH3) in WT (l) and MZudu−/− gastrulae (m) at 80% epiboly. Images are representative of eight independent trials. n Quantification of pH3+ cells/embryo at the stages indicated. Each dot represents a single embryo, dark lines are means with SEM (T-test, *p < 0.05, ***p < 0.001, ****p < 0.0001). Scale bars are 50 μm. Anterior/animal pole is up in all images EXPRESSION / LABELING:
PHENOTYPE:
|
|
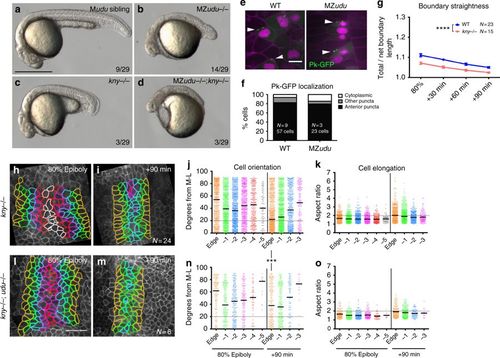
Gon4l regulates axis extension independent of PCP signaling. a–d Live embryos at 24 hpf resulting from a cross between a germline-replaced udu−/−;kny fr6/+ female and an udu+/-;knyfr6/+ male. Genotypes are indicated in the upper right corner, fractions indicate the number of embryos in the clutch with the pictured phenotype. e Mosaically expressed Prickle (Pk)-GFP in WT and MZudu−/− gastrulae. Arrowheads indicate anteriorly localized Pk-GFP puncta. Membrane Cherry marks cell membranes, nuclear-RFP marks cells injected with pk-gfp RNA. Images are representative of three independent experiments. f Quantification of Pk-GFP localization shown in e (chi-square, p = 0.07). g Quantification of notochord boundary straightness in WT and kny−/− gastrulae. Symbols are means with SEM (two-way ANOVA, ****p < 0.0001). h, i, l, m Still images from live time-lapse confocal movies of the axial mesoderm in kny−/− (h–i) and kny−/−;udu−/− (l–m) gastrulae at the time points indicated. Cell outlines are colored as in Fig. 3. j, n Quantification of axial mesoderm cell orientation as in Fig. 3, bars are median values (Kolmogorov–Smirnov test, ***p = 0.0003). k, o Quantification of axial mesoderm cell elongation as in Fig. 3, bars are mean values. N indicates number of embryos analyzed (and number of cells in f). Scale bar is 500 μm in a–d, 10 μm in e, 50 μm in h–m PHENOTYPE:
|
|
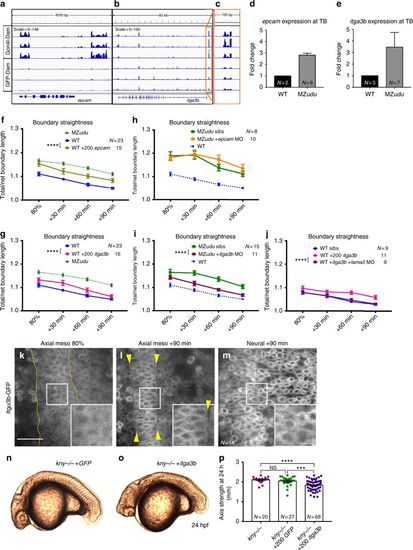
Gon4l regulates notochord boundary straightness by limiting itga3b expression. a–c Genome browser tracks of Gon4l-Dam and GFP-Dam association at the epcam (a) and itga3b (b) locus. An expanded view of the itga3b promoter is shown in c. d–e Quantitative RT-PCR for epcam (d) and itga3b (e) in WT and MZudu−/− embryos at tailbud stage. N indicates the number of independent clutches tested with technical triplicates of each, bars are means with SEM. f–g Quantification of notochord boundary straightness in WT epcam (f) or itga3b (g) overexpressing embryos (two-way ANOVA, ****p < 0.0001). h–i Notochord boundary straightness in MZudu−/− epcam (h) or itga3b (i) morphants and sibling controls (two-way ANOVA, ****p < 0.0001). j Quantification of notochord boundary straightness in WT itga3b-overexpressing embryos with or without lama5 MO (two-way ANOVA, ****p < 0.0001). N indicates the number of embryos analyzed, symbols are means with SEM. k–m Itgα3b-GFP localization in WT embryos in the tissues and at the time points indicated. Insets are enlarged from regions in white squares. Yellow lines and arrowheads mark notochord boundaries. Images are representative of three independent trials. Scale bar is 50 μm. n–o Live images of kny−/− embryos at 24 hpf injected with 200 pg GFP (n) or 200 pg itga3b (o) RNA. Images are representative of four independent experiments. p Quantification of axis length of injected kny−/− embryos at 24 hpf. Each dot represents one embryo, bars are means with SEM (T-tests, ***p < 0.001, ****p < 0.0001). N indicates number of embryos analyzed EXPRESSION / LABELING:
PHENOTYPE:
|
|
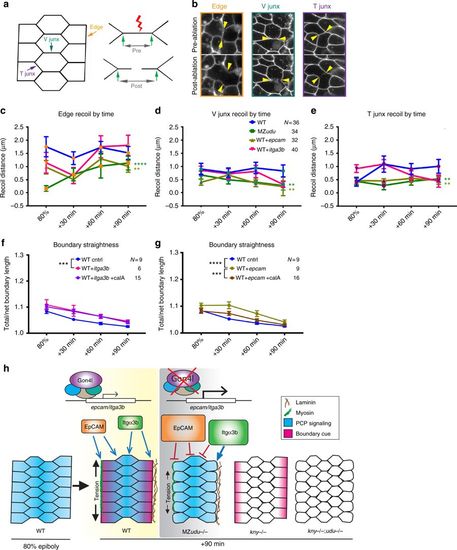
Loss of Gon4l and excess epcam reduce notochord boundary tension. a Diagram of laser ablation experiments to measure tension at axial mesoderm cell interfaces. b Still images from confocal time-lapse movies of each of the three types of cell interfaces (Edge, V junctions, and T junctions) before and after laser ablation. Arrowheads indicate cell vertices adjacent to the ablated interface. Images are representative of 36 independent experiments. c–e Quantification of cell vertex recoil distance immediately after laser ablation of Edge (c), V junction (d), and T junction (e) interfaces at the time points indicated in WT, MZudu−/−, WT epcam overexpressing, and WT itga3b-overexpressing gastrulae. Symbols are means with SEM. Asterisks are colored according to key and indicate significant differences compared to WT controls (two-way ANOVA, **** p < 0.0001, **p < 0.01). f–g Quantification of notochord boundary straightness in WT itga3b (f) or epcam (g) overexpressing embryos with or without Calyculin A (two-way ANOVA, ****p < 0.0001, ***p < 0.001). N indicates the number of embryos analyzed, symbols are means with SEM. h Graphical model of the roles of Gon4l and PCP signaling in regulating ML cell polarity of axial mesoderm cells and notochord boundary formation PHENOTYPE:
|
|
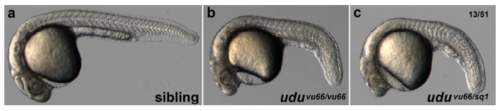
vu66 is a new allele of ugly duckling Live day 1 WT (a) and udu-/- (b,c) embryos. c) Representative embryo from a complementation cross between a vu66 heterozygote and an udusq1 heterozygote. Phenotype of vu66/sq1 mutants (c) resembles vu66/vu66 (b), indicating failure to complement. Fraction indicates the number of embryos with the pictured genotype/phenotype over total embryos in the clutch. |
|
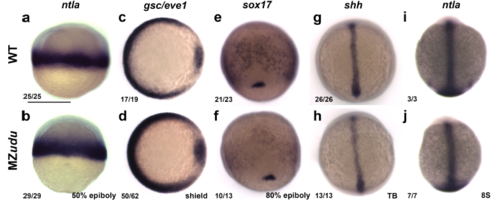
Maternal zygotic (MZ)udu-/- embryos specify all three germ layers Whole mount in situ hybridizations (WISH) in WT (top row) and MZudu-/- embryos (bottom row) at the stages indicated. a-b) ntla marks nascent mesoderm. c-d) gsc and eve1 mark the embryonic shield and ventral mesoderm, respectively. e-f) sox17 marks endoderm and dorsal forerunner cells. g-j) shh (g-h) and ntla (i-j) mark axial mesoderm. Fractions indicate the number of embryos with the pictured phenotype over the number of embryos examined. Images depict a dorsal view with anterior/animal pole up in all images except c-d in which dorsal is to the right. Scale bar is 300μm. |
|
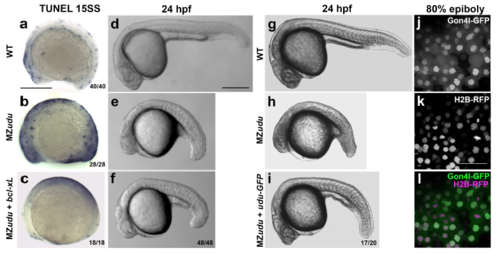
Increased apoptosis in MZudu-/- embryos is not causative of axis extension defects a-c) TUNEL staining in WT (a), MZudu-/- (b), and MZudu + bcl-xL RNA (c) at 15 somite stage. d-i) Live WT (d,g), MZudu-/- (e,h), MZudu + bcl-xL (f), and MZudu + 25pg udu-gfp (i) embryos at 24hpf. Fractions indicate the number of embryos with the pictured phenotype over the number of embryos examined. j-l) Live WT embryos at 80% epiboly expressing Gon4l-GFP (j) and Histone2B-RFP (k), merged channels in l. Images are representative of four independent trials. Scale bars are 300μm in a-i, 50μm in k. Anterior is to the left. |
|
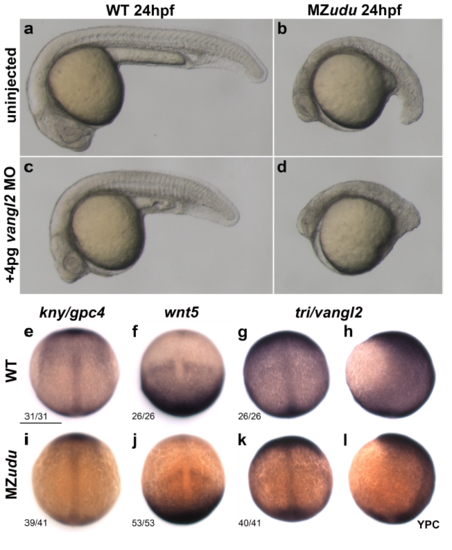
Gon4l functions in parallel to PCP signaling a-b) Live uninjected WT (a) and MZudu-/- (b) embryos at 24hpf. c-d) Live 24hpf WT (c) and MZudu-/- (d) embryos injected with 4pg MO1-vangl2 morpholino. Images are representative of two independent trials. e-l) WISH for the PCP genes kny/gpc4 (e,i), wnt5 (f,j), and tri/vangl2 (g-h, k-l) in WT (top row) and MZudu-/- (bottom row) embryos at YPC stage. Fractions indicate the number of embryos with the pictured phenotype over the number of embryos examined. Anterior is up, dorsal is to the right in h,l. Scale bar is 300 μm. |
|
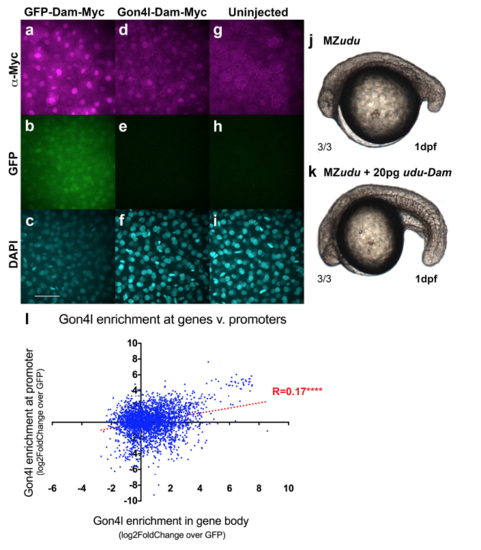
Dam fusion proteins are functional a-i) Anti-Myc Immunofluorescent staining for Dam-Myc fusion proteins in WT embryos injected with gfp-dammyc (a-c) or udu-dam-myc (d-f) RNA or uninjected controls (g-i) at blastula stage. Images are representative of multiple embryos from a single trial. j) Uninjected MZudu-/- sibling at 1 dpf. k) MZudu-/- injected with 20pg ududam RNA. Fractions indicate number of embryos with the pictured phenotype over the number of embryos examined. l) Correlation between Gon4l enrichment across promoters and enrichment across gene bodies of genes with regions of significant Gon4l association (Spearmann correlation ****p<0.0001). Dotted line is linear regression of correlation. Negative values indicate depletion of Gon4l relative to GFP controls. Scale bar is 50μm. |
|
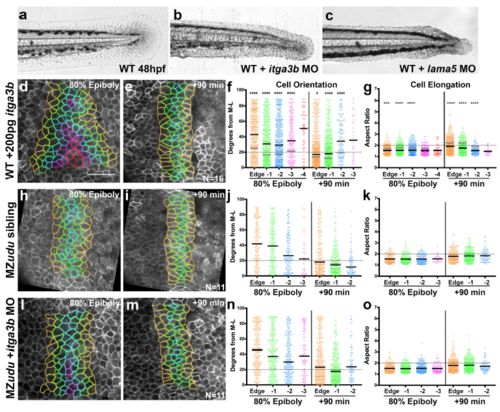
Reduction of Integrinα3b does not suppress cell polarity defects in MZudu-/- gastrulae a-c) Live images of caudal fins of an uninjected WT embryo (a) and those injected with 2ng itga3b MO (b) or 1ng lama5 MO (c) at 48hpf (note recapitulation of mutant phenotypes1). Images are representative of all MOinjected embryos from three (itga3b) or four (lama5) independent trials. d-g) Quantification of axial mesoderm cell orientation (f) and elongation (g) in itga3b overexpressing WT gastrulae. Asterisks indicate significant differences compared to WT controls (Kolmogorov-Smirnov (f) and Mann-Whitney (g) tests, *p<0.05, ***p<0.001, ****p<0.0001). Black bars are median values in f, mean values in g; medians and means of WT are shown as gray bars in f-g. h-o) Quantification of axial mesoderm cell orientation and elongation in control MZudu-/- siblings (h-k) and MZudu-/- injected with 2ng itga3b MO (l-o) at the time points indicated. Graphs and color coding as in Fig.3. Bars are median values in j, n; bars are mean values in k, o. N indicates the number of embryos analyzed, scale bar is 50μm. |
|
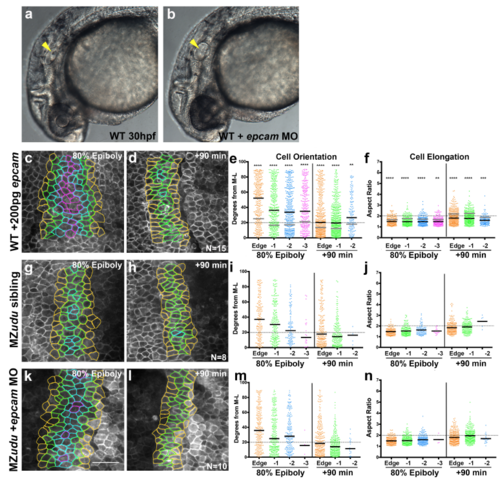
Reduction of EpCAM does not suppress cell polarity defects in MZudu-/- gastrulae a-b) Live images of otic vesicles in WT embryos at 30hpf either (a) uninjected or (b) injected with 1ng epcam MO. Arrowheads mark otoliths (or lack thereof). Approximately 36% of epcam MO-injected embryos from three independent trials lacked otoliths as shown in b, an additional 56% exhibited smaller otoliths (both phenotypes are observed in epcam mutants2). c-f) Quantification of axial mesoderm cell orientation (e) and elongation (f) in epcam overexpressing WT gastrulae. Asterisks indicate significant differences compared to WT controls (Kolmogorov-Smirnov (e) and Mann-Whitney (f) tests, **p<0.01, ***p<0.001, ****p<0.0001). Black bars are median values in e, mean values in f; medians and means of WT are shown as gray bars in e-f. g-n) Quantification of axial mesoderm cell orientation and elongation in control MZudu-/- siblings (g-j) and MZudu-/- injected with 1ng epcam MO (k-n) at the time points indicated. Graphs and color coding as in Fig. 3. Bars are median values in i, m; bars are mean values in j, n. N indicates the number of embryos analyzed, scale bar is 50μm. |
|
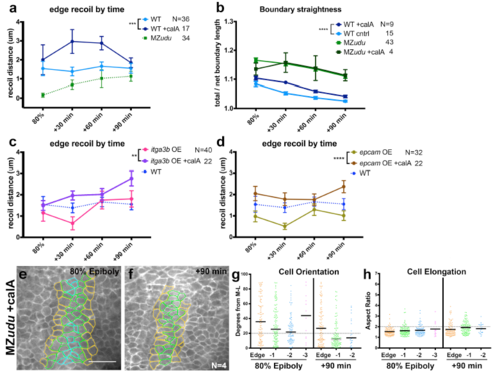
Calyculin A treatment increases notochord boundary tension without improving MZudu-/- boundary straightness or cell polarity a) Quantification of cell vertex recoil distance upon laser ablation of Edge cell interfaces at the time points indicated in MZudu-/-, WT, and Calyculin A-treated WT gastrulae. Symbols are means with SEM (2-way ANOVA, ***p=0.0006). b) Quantification of boundary straightness in WT and MZudu-/- gastrulae with or without Calyculin A treatment. Symbols are means with SEM (2-way ANOVA, ****p<0.0001). c-d) Cell vertex recoil distance upon laser ablation of Edge cell interfaces at the time points indicated in WT embryos overexpressing itga3b (c) or epcam (d) with or without Calyculin A treatment. Symbols are means with SEM (2- way ANOVA, **p=0.027, ****p<0.0001). e-h) Quantification of axial mesoderm cell orientation (g) and elongation (h) in Calyculin A-treated MZudu-/- gastrulae. Graphs and color coding as in Fig.3. Bars are median values in g and mean values in h. Scale bar is 50μm. N indicates the number of embryos analyzed. |