- Title
-
Wnt3 and Wnt3a are required for induction of the mid-diencephalic organizer in the caudal forebrain
- Authors
- Mattes, B., Weber, S., Peres, J., Chen, Q., Davidson, G., Houart, C., and Scholpp, S.
- Source
- Full text @ Neural Dev.
|
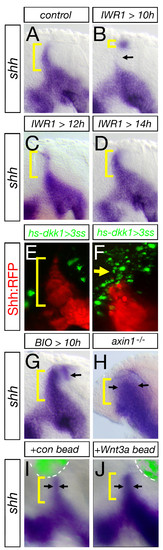
Canonical Wnt signaling is required for induction of Shh expression in the MDO. MD organizer in lateral views of embryos with anterior to the left at 28 hpf. Blockage of canonical Wnt signaling by treatment of embryos with 40 μM IWR1 at different time points leads to the following alterations in the formation of the shh positive MDO: 10 to 28 hpf - strong decrease (B, arrow), 12 to 28 hpf - mild decrease (C) 14 to 28 hpf - no alteration (D). Shh expression at the MDO is unaltered if Dkk1-GFP positive cells are located in the telencephalon and the pretectum (E), however, the shh positive MDO is absent if Dkk1-GFP cells are located in the thalamic complex (F, n = 6/10, arrow). Ectopic activation of the Wnt signaling pathway by BIO treatment (G, n = 12/18), in the axin1-/- mutant embryos (H) as well as embryos with an implanted Wnt3a loaded bead (I, J, n = 3/5) leads to a increase of MD organizer shown by expanded shh expression (black arrows). |
|
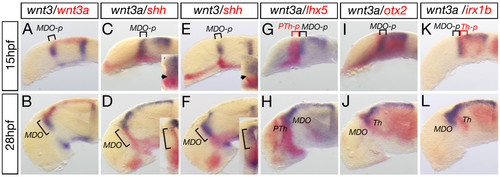
Wnt3 and Wnt3a mark the MDO and the dorsal thalamus. Analysis of expression dynamics of indicated marker genes at 15 and 28 hpf. At 15 hpf, wnt3 and wnt3a are expressed in the MDO primordium (MDO-p) and maintained in the organizer at 28 hpf. Cross section analysis of the left hemisphere reveals that wnt3a expression is adjacent to shh expression at the ventricular zone of the organizer (D), whereas wnt3 expression co-localizes with shh expression at 28 hpf (F). In the diencephalon, expression of wnt3a is located between the lhx5 expression domain (G, H) and the irx1b expression domain (K, L). Consistently, wnt3a is co-expressed with otx2 (I, J) suggesting that wnt3a marks the MD organizer territory at 15 hpf and 28 hpf. MDO, PTh, prethalamus, Th, thalamus, *-p, *-primordium. EXPRESSION / LABELING:
|
|
Analysis of knock-down of Wnt3 and Wnt3a. Injection of wnt3 MO leads to a decrease in splicing of mRNA at the exon3-exon4 boundary and an increase in non-spliced RNA product visualized by amplification at the exon3-intron3 boundary and subsequently to a reduction of Wnt3 protein in morphant embryos at 28 hpf (A). Knock-down of either Wnt3 or Wnt3a leads to an indistinguishable morphology to the control Morpholino-injected embryos whereas the double morphant embryos show a significant alteration in brain morphology, a kinked tail and a smaller otic vesicle (B, arrows). Similarly, analysis of marker genes in the forebrain midbrain area shows no alteration in control, wnt3 or wnt3a morphant embryos (C-H). Cer, cerebellum. |
|
Wnt3 and Wnt3a are required for MDO formation. Knock-down of Wnt3 and Wnt3a leads to a down-regulation of the expression of indicated markers in the central part of the MDO (A-L). Time-lapse analysis of Shh::RFP shows an independent induction of the dorsal remaining organizer spot (M, N). Treatment of wnt3/wnt3a morphant embryos with the Wnt signaling agonist BIO from 10 to 28 h leads to a rescue of shh expression at the MDO (O, P). Brackets mark the extent of the MDO, whereas the arrows indicate the gap between the basal plate and the remaining spot of dorsal MDO identity. Eth, epithlamus, HyTh, hypothalamus, MHB, midbrain-hindbrain boundary, Tel, telencephalon. |
|
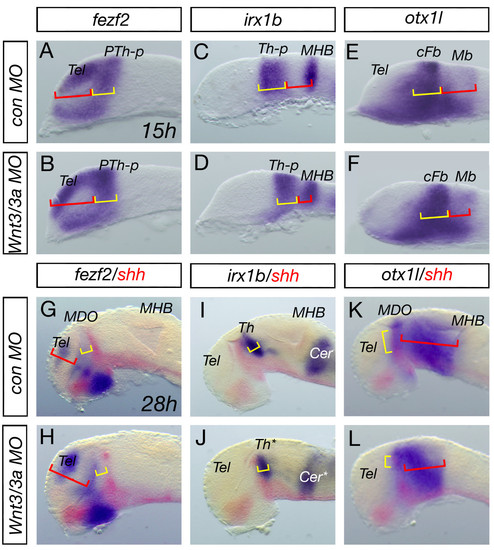
Primordia of the prethalamus and the thalamus are not affected in Wnt3/Wnt3a morphant embryos. Analysis of the fezf2 positive anterior forebrain area at 15 and 28 hpf reveals an expansion of the telencephalic domain (red brackets), however, the prethalamus (yellow brackets) is unaltered wnt3/wnt3a morphant embryos (A, B, G, H). Posteriorly analysis of the irx1b as well as the otx1l expression shows that the thalamic domain does not change significantly (yellow brackets), whereas, the midbrain territory shrinks in compound morphant embryos (red brackets, C-F; I-L). Notably, although both organizers - the MDO and MHB - disappeared in wnt3/wnt3a morphant embryos, expression of competence factors like fezf2 and irx1b are unaltered at 28 hpf, suggesting a specific function of Wnt3/Wnt3a in organizer formation. cFb, caudal forebrain, Mb, midbrain. EXPRESSION / LABELING:
|
|
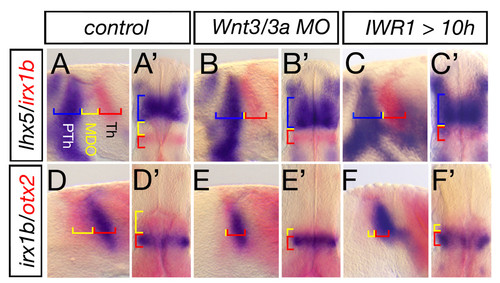
Blockage of Wnt signaling lead to lack of MDO tissue. Lateral views and dorsal views (marked by ′) of embryonic heads at 28 hpf. Wnt3/wnt3a morphant embryos lack the organizer tissue and the lhx5 positive prethalamus (PTh, blue) abuts the irx1b thalamus (Th, red) (A-B′, n = 54/78). A similar phenotype is observed in embryos treated with 30 μM IWR1 from 10 to 28 h to inhibit canonical Wnt signaling (n = 15/17). Hence, the otx2 positive MDO (yellow) is lacking if Wnt3/Wnt3a function is knocked-down (n = 44/80, D-E′), likewise after inhibition of canonical Wnt signaling (F, F′, n = 14/24). |
|
Lack of Wnt signaling leads to apoptosis in the MDO. Lateral views and dorsal views of the left hemisphere (marked by ′) of Shh::RFP transgenic embryos at 36 hpf. Wnt3/wnt3a morphant embryos as well as embryos treated with IWR1 display an increase of apoptotic cells within the MDO at 36 hpf (n = 4/6, A-C′). Upon blockage of Tp53 function apoptosis is down-regulated within the organizer tissue of wnt3/wnt3a morphant embryos as well as embryos treated with IWR1 and consistently, shh:RFP expression is restored (D-F′). |
|
FezF2 and Irx1b limit the MDO at the AP axis. Shh expression in the MDO is decreased in Wnt3/Wnt3a deficient embryos (A, B, n = 45/55), whereas, prethalamic identity as well as thalamic identity is maintained (E, F, n = 18/23). In triple morphant embryos lacking Wnt3, Wnt3a and Fezf2 function, a restoration of shh expression is observed in the MDO (C, n = 28/31) accompanied by a reduction of lhx5 expression in the pre-thalamus (G, n = 10/34). In wnt3/wnt3a/irx1b triple morphant embryos, we observe a similar restore of the shh positive MDO (D, n = 28/31); however, here the neurog1 positive thalamus shrinks (H, n = 25/32, N, n = 20/25). For explanation of the summary (I-L) see the text. |