- Title
-
Rotation and asymmetric development of the zebrafish heart requires directed migration of cardiac progenitor cells
- Authors
- Smith, K.A., Chocron, S., von der Hardt, S., de Pater, E., Soufan, A., Bussmann, J., Schulte-Merker, S., Hammerschmidt, M., and Bakkers, J.
- Source
- Full text @ Dev. Cell
|
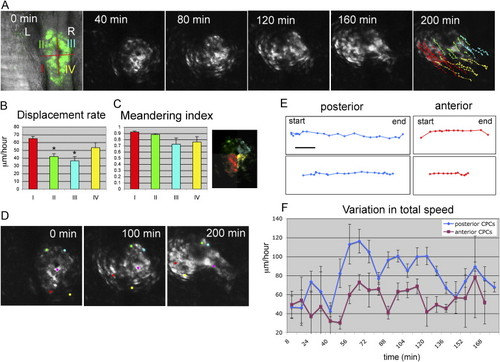
Rotation of the Cardiac Cone during Formation of the Linear Heart Tube (A) Selected images of a confocal time-lapse recording of a tg(cmlc2:gfp) embryo starting at the 23-somite stage. Individual GFP-positive cells were tracked and color-coded according to their location within the cardiac field at the 23-somite stage. Dorsal view with anterior to the top. (B and C) Displacement rates and meandering index (displacement/track length) of CPCs located at different positions within the cardiac field (color corresponds to position indicated in the confocal image) calculated from time-lapse recording shown in (A) (n = 30). Error bars indicate standard errors. Statistic significance (by paired t test) calculated for CPCs derived from sector I (red) compared with CPCs derived from sectors II (green) and III (blue) (* p < 0.005). (D) Selected images of confocal timelapse recording with individual cells labeled during formation of the heart tube. Individual cells are marked (yellow, red, green, and blue for laterally located atrial cells and pink for medially located ventricular cells) and tracked over a 200 min period, revealing the evident rotation of the cardiac cone. (E) Representative tracks of individual cells originating from the posterior (blue) or anterior (purple) region of the cardiac cone with 8 min intervals in between time points shown. Scale bar represents 25 μm. (F) Graphical representation of CPC speeds during the process of heart tube formation. Cells were grouped according to their original position in the cardiac field (posterior, blue; anterior, purple). EXPRESSION / LABELING:
|
|
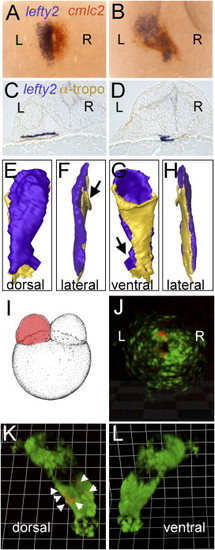
lefty2 Expression and Lineage Tracing Reveals a Dorsal-Ventral Orientation of the Left-Right Regions of the Heart Field, Respectively, in the Linear Heart Tube (A and B) Double ISH with cmlc2 (red) and lefty2 (blue) antisense riboprobes at 23-somite stage (A) and 24 hpf (B). Dorsal views with anterior to the top. (C and D) Serial section and 3D reconstruction (E?H) of an ISH embryo with lefty2 probe counterstained by using an anti-tropomyosin antibody at the level of the venous pole of the heart tube (C) and the arterial pole (D). (E?H) 3D reconstruction with blue representing lefty2-positive tissue and yellow representing tropomyosin-positive tissue. (E) Dorsal view with venous pole to the top and arterial pole to the bottom. Arrows in (F) and (G) indicate regions at the poles where some torsion of the tube is visible. (I?L) Lineage tracing of CPCs from the left or right cardiac field. (I) CPCs were mosaically labeled in the left or right cardiac field by injection of cmlc2:mRFP DNA into one cell at the two-cell stage. (J) In the cardiac cone, mRFP-expressing CPCs were observed on the left side, and these cells were later found to occupy the dorsal part (K, arrowheads) of the linear heart tube with no mRFP cells observable on the ventral side of the tube (L). EXPRESSION / LABELING:
|
|
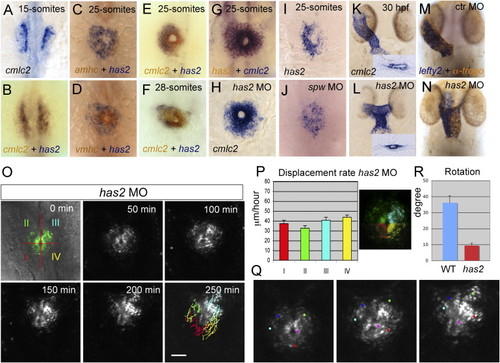
Has2 Expression in CPCs Is Required for Jogging and Rotation of the Cardiac Cone (A?L) One- and two-color ISH (probes indicated at bottom) on wild-type embryos (A?G, I, and K) or embryos injected with a southpaw MO (J) or a has2 MO (H and L) during various stages (indicated at the top) of heart tube formation. Evident is the dynamic expression pattern of has2 in the CPCs. All images are dorsal views with anterior to the top and left to the left. (M and N) Combined ISH with an antisense lefty2 riboprobe (blue) and anti-tropomyosin antibody staining (brown, labeling all cardiomyocytes) of a control MO-injected embryo (M) or has2 MO-injected embryo (N). (O) Selected images of a confocal time-lapse recording of a tg(cmlc2:gfp) embryo injected with a has2 MO starting at the 23-somite stage. Individual GFP-positive cells were tracked and color coded according to their location within the cardiac field at the 23-somite stage. Dorsal views with anterior to the top. (P) Displacement rates of CPCs localized at different positions within the cardiac field (color corresponds to position indicated in the confocal image) calculated from time-lapse recording shown in (O) (n = 55, mean � SEM). Evident is the absence of leftward displacement of the CPCs and their equal displacement rates. (Q) Selected images of a confocal time-lapse recording shown in (O) with individual cells marked (red, light blue, dark blue, and green for atrial cells located lateral and pink for ventricular cells located medial) and tracked over a 250 min period. (R) Quantification of clockwise cardiac cone rotation in wild-type and has2 MO-injected embryos (mean ± SEM). EXPRESSION / LABELING:
PHENOTYPE:
|
|
Asymmetric Activation of Smad 1,5,8 Protein in the LPM. Confocal images of the LPM at the cardiac region in 23-somite stage embryos stained for tropomyosin (red, A and D), phospho-Smad 1,5,8 (white, B and E), and the overlay (C and F). Staining in WT embryos (A?C) shows asymmetric activation of Smad 1,5,8 on the left side (yellow arrowheads) compared with right (green arrows). Cell counting showed this difference to be statistically significant ([G]; mean � SEM, # p < 0.05). Activation of Smad 1,5,8 was significantly lower in tg(hsp70:noggin3) embryos (D?F) compared with WT embryos ([H]; mean ± SEM, * p < 0.001), and the difference between left and right activation of Smad 1,5,8 was not significant ([I]; mean ± SEM). In all images anterior is to the top, left is to the left side, and the midline is demarcated by the dashed yellow line. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Leftward Displacement of CPCs and Cardiac Rotation Are Affected upon Reduced or Ectopic BMP Signaling (A?C) Selected images of confocal time-lapse recordings of laf/alk8 (A) heat-shock induced (at 15-somite stage) tg(hsp70:noggin3) (B) and tg(hsp70:bmp2b) (C) embryos with individual cells marked and tracked over a 200 min period. (D) Quantification of clockwise cardiac cone rotation in wild-type laf/alk8 mutant tg(hsp70:noggin3) and tg(hsp70:bmp2b) embryos (mean � SEM). (E) ISH with a cmlc2 riboprobe on tg(hsp70:bmp2b) embryos heat shocked at various time points (indicated at the top). Arrows and arrowheads indicate CPCs not properly incorporated into the cardiac tube. Heat shock at 15-somite stage, no cardiac jogging (n = 14/16); 18-somite stage, no cardiac jogging (n = 12/20); 22-somite stage, leftward jogging (n = 17/19). EXPRESSION / LABELING:
PHENOTYPE:
|
|
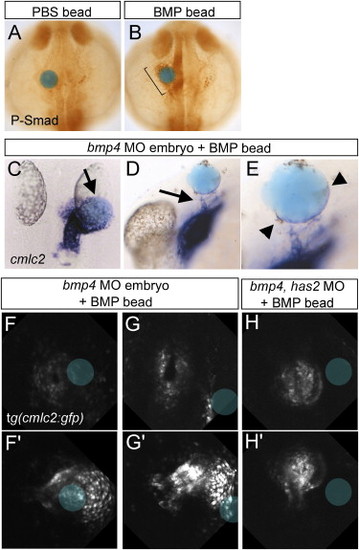
BMP Beads Direct Cardiac Jogging (A and B) Phospho-Smad 1,5,8 antibody staining after implantation at the 15-somite stage of a PBS bead (A) or a BMP bead (B). Bracket in (B) indicates the induced phospho-Smad staining in close proximity of the BMP bead. Dorsal view with anterior to the top of 25-somite stage embryos. (C?E) ISH with antisense cmlc2 riboprobe of embryos injected with a bmp4 MO and a BMP bead implanted at the 15?18-somite stage. (C) The heart tube is redirected to a BMP bead implanted on the right side (11/15), dorsal view with anterior to the top, arrow indicates position of the BMP bead. (D) In a lateral view, arrows indicate CPCs that have been recruited toward the BMP bead (n = 7/8). Arrowheads (E) point to a layer of CPCs that form a layer on the bead's surface. (E) is an enlargement of (D). (F?H′) Selected images of a confocal time-lapse recording of bmp4 morphants (F?G′) or a bmp4 has2 double morphant (H and H′) with a BMP bead placed on the right side. Images represent the start (F?H) and end point (F′,G′,H′) of the time-lapse of the three individual embryos. Position of the BMP beads is marked by the blue circle. |
|
Stages of Zebrafish Heart Development (A-C) Dorsal views of double labeling in situ hybridisation with ventricle myosin heavy chain (vmhc, blue) and atrial myosin heavy chain (amhc, red) probes. After fusion of the bilateral heart fields (23-somites, 20 hours post fertilization hpf) a cardiac disk is formed which is located ventrally to the neural tube and endoderm (A). While future ventricle cells (blue), which are positioned centrally in the disk, start to form a cone the future atrial cells (red), located at the periphery of the disk, remain spread out on the yolk (B). At the same time the venous pole of the heart is displaced towards the left while the arterial pole of the tube remains positioned at the midline, a process referred to as cardiac jogging. Slightly later, when extension of the tube has proceeded, also the atrial cells are incorporated into the linear heart tube, which is clearly jogged towards the left. D) Dorsal view of a similar stage embryo as shown in (C). Anterior and posterior have been reversed to allow better comparison with succeeding looping stages during which the heart becomes located more anteriorly due to extension of the embryo. E,F) Anterior views of looping stage embryos. At the onset of rightward looping a Cloop is formed during which the atrioventricular canal becomes positioned at the midline (E). When looping has completed a S-shaped heart is formed with the ventricle positioned to the right and the atrium to the left (F). AVC, atrioventricular canal, A atrium, hpf hours post fertilization, IF inflow, L left, OF outflow, R right. EXPRESSION / LABELING:
|
|
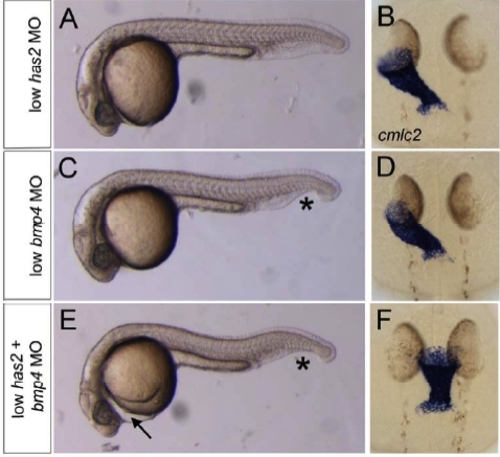
Defects in Cardiac Jogging Are Synergistically Enhanced upon Combined Moderate Knockdown of Has2 and Bmp4 (A,B) Injection of moderate amount of has2 MO has no visible effect on the embryo nor on cardiac jogging (0.4 ng has2 MO, 68/73 embryos). (C,D) Injection of a moderate amount of bmp4 MO results in a mild reduction of the ventral tail fin (*) but no effect on cardiac jogging (0.4 ng bmp4 MO, 71/73 embryos). (E,F) Co-injection of moderate amounts of has2 and bmp4 MO leads to synergistic enhancement of the cardiac jogging defect (17/63 embryos) without affecting dorsalventral patterning indicated by the only very mild reduction of the ventral tail fin (*). Arrow indicatFi> and bmp4 MO les pericardial edema. (A,C,E) Lateral views of live embryos, (B,D,F) dorsal views with anterior to the top of in situ hybridizations with an antisense cmlc2 riboprobe. |

Unillustrated author statements |
Reprinted from Developmental Cell, 14(2), Smith, K.A., Chocron, S., von der Hardt, S., de Pater, E., Soufan, A., Bussmann, J., Schulte-Merker, S., Hammerschmidt, M., and Bakkers, J., Rotation and asymmetric development of the zebrafish heart requires directed migration of cardiac progenitor cells, 287-297, Copyright (2008) with permission from Elsevier. Full text @ Dev. Cell