- Title
-
Marcksb plays a key role in the secretory pathway of zebrafish Bmp2b
- Authors
- Ye, D., Wang, X., Wei, C., He, M., Wang, H., Wang, Y., Zhu, Z., Sun, Y.
- Source
- Full text @ PLoS Genet.
|
(A) Knockdown of EXPRESSION / LABELING:
|
|
(A) WISH analysis showed that the transcriptional levels of downstream targets of BMP signaling— |
|
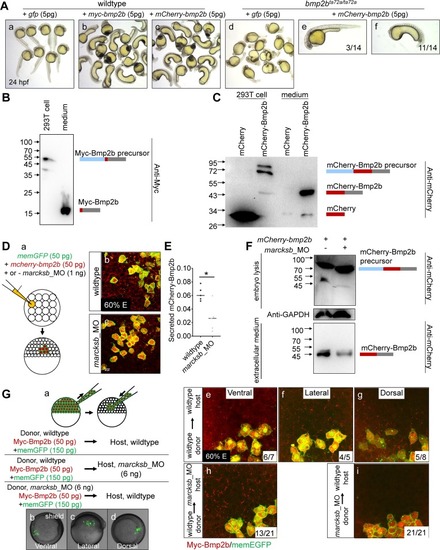
(A) Overexpression of |
|
(A) Injection of either |
|
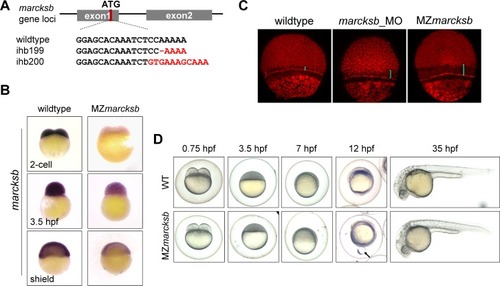
(A) The diagram shows the gRNA target of |
|
(A, B) Injection of |
|
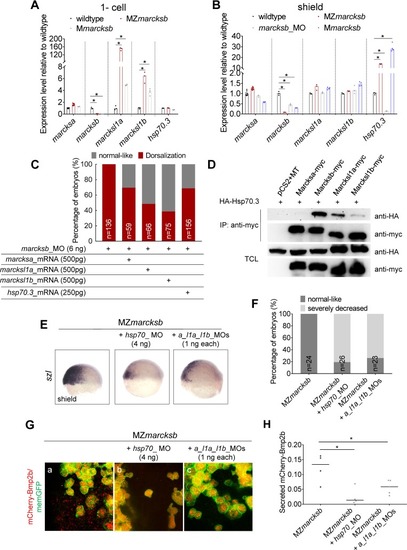
(A, B) RT-qPCR analysis on the expression of |
|
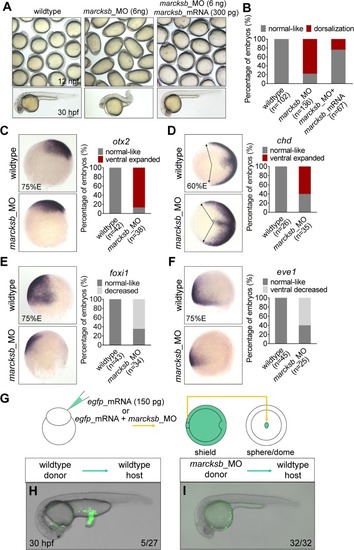
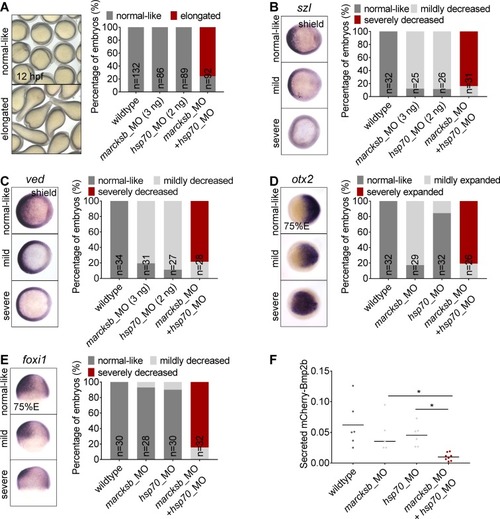
(A) Morphological phenotypes of embryos at 12 hpf, injected with sub-dosages of morpholinos against EXPRESSION / LABELING:
PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. EXPRESSION / LABELING:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |