- Title
-
Basal constriction during midbrain-hindbrain boundary morphogenesis is mediated by Wnt5b and focal adhesion kinase
- Authors
- Gutzman, J.H., Graeden, E., Brachmann, I., Yamazoe, S., Chen, J.K., Sive, H.
- Source
- Full text @ Biol. Open
|
Basal constriction at the zebrafish MHBC occurs prior to apical expansion and results in wedge-shaped cells. (A–D) Live scanning confocal imaging of wild-type embryos injected with mGFP mRNA and imaged at 14 ss, 22 ss, 28 ss, and prim-6. Cells at the MHBC are outlined in yellow, red, and blue. (E–H) 3D reconstruction of red outlined cell using 3D Doctor (Able Software). Each reconstruction is shown from two viewpoints: face-on as in the confocal image, and with a 45° rotation of the same image. (I) Histogram comparing the basal width of cells at each time point. (J–M) Schematics of wild-type MHBC formation. Anterior is to the left in all images. Arrowheads indicate the MHBC. M, midbrain; H, hindbrain. A–D, n>8 embryos per stage; I, n=3 embryos with 6 cells measured per embryo for each time point. Error bars reflect ±s.d. Scale bars: 9 µm. |
|
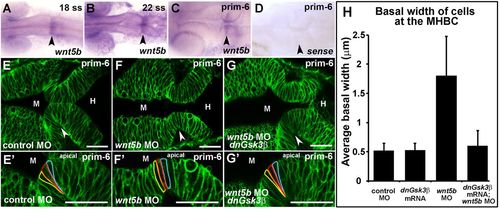
Wnt5b regulates basal constriction possibly through Gsk3β. (A–D) In situ hybridization of wnt5b expression during MHB development at 18 ss (A), 22 ss (B), and prim-6 (C). (D) prim-6 sense probe control. (E–G′) Live confocal images of the MHB region in prim-6 stage embryos. Single-cell wild-type embryos were injected with mGFP to label cell membranes and co-injected with control MO (E,E′), wnt5b MO (F,F′), or wnt5b MO and dnGsk3β mRNA (G,G′). (H) Quantification of basal cell width in control MO, wnt5b MO, dnGsk3β mRNA (image not shown), and wnt5b MO+dnGsk3β mRNA injected embryos. (H) For each treatment group, n=3 embryos. For each embryo, 6 cells located at the MHBC were measured, 3 cells on each side. Arrowheads indicate MHBC. M, midbrain; H, hindbrain. Scale bars: 26 µm. EXPRESSION / LABELING:
PHENOTYPE:
|
|
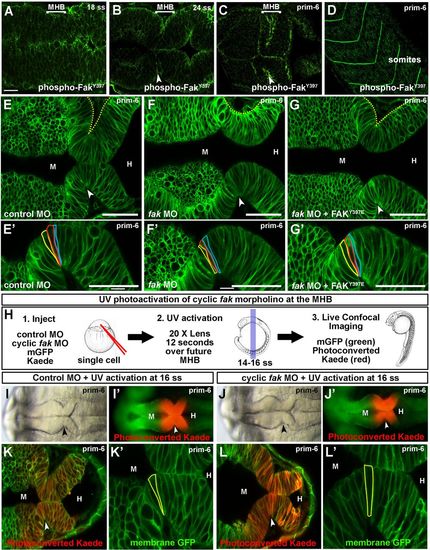
Fak is required at the MHB for basal constriction. (A–D) Wild-type embryos stained with anti-phospho-FakY397 antibody and imaged by scanning confocal microscopy. (A,B) phospho-FakY397 is localized at the basal and apical sides of the neural tube at 18 and 24 ss. (C) Activated Fak is enriched at the MHBC at prim-6. (D) Phospho-FakY397 is localized to somite boundaries at prim-6. (E–G′) Live confocal images of embryos co-injected with mGFP and control MO (E,E′), fak MO (F,F′), or fak MO+FAKY397E mRNA (G,G′). (E′–G′) Magnifications of individual cells outlined at the MHBC. (H) Schematic for fak caged MO experiments. (1.) One-cell stage wild-type embryos were co-injected with mGFP and photoconvertable Kaede mRNA and either control MO or cyclic fak MO. (2.) Cyclic fak MO was uncaged at 16 ss by UV activation. (3.) Embryos were incubated until prim-6 and then imaged using brightfield, fluorescence, and live scanning confocal microscopy. (I–J′) Gross morphology using brightfield imaging (I,J) and corresponding fluorescent (I′,J′) images of embryos injected with control MO (I,I′) or photoactivatable fak MO (J,J′) after UV photoconversion. (K–L′) Live confocal images showing the MHB region of prim-6 embryos after photoconversion. (K′,L′) Magnifications of the neuroepithelium shown in K and L with individual cells outlined at the MHBC. (n>6). Anterior is to the left in all images. Arrowheads indicate MHBC. M, midbrain; H, hindbrain. Scale bars: A–C=20 µm. E–G′=50 µm. EXPRESSION / LABELING:
PHENOTYPE:
|
|
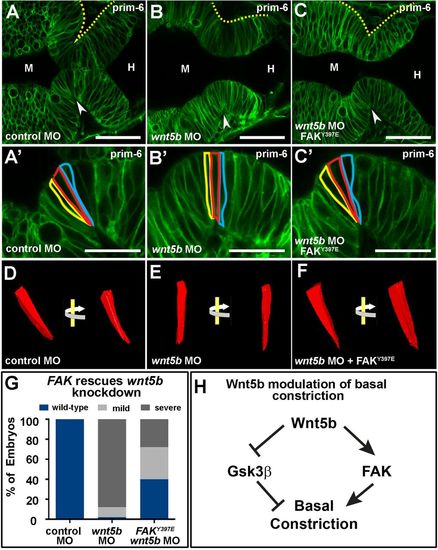
Fak rescues effects of wnt5b knockdown on basal constriction. (A–C′) Live confocal images of the MHB region in prim-6 stage embryos. Single-cell wild-type embryos were co-injected with mGFP and control MO (A,A′), wnt5b MO (B,B′), or wnt5b MO and FAKY397E mRNA (C,C′). (D–F) 3D reconstruction of cells outlined in (A′–C′) using 3D doctor with view of the same cell rotated 45°. (G) Quantification of MHBC gross morphology and basal constriction following FAK397E mRNA rescue of wnt5b knockdown (n>60 per condition). (H) Model pathway for Wnt5b regulation of basal constriction. Arrowheads indicate MHBC. M, midbrain. H, hindbrain. Scale bars: A–C, 50 µm; A′–C′, 25 µm. |
|
Tissue patterning and polarity are retained in wnt5b and fak morphants. (A-F) in situ hybridization patterns for fgf8 and pax2a expression in control MO (A,D), wnt5b MO (B,E), and fak MO (C,F) injected embryos at 18 ss. Expression patterns are normal in both wnt5b and fak morphants. RNA probes containing digoxigenin-11-UTP were synthesized from linearized plasmid DNA for pax2.1 (Krauss et al., 1991), and fgf8 (Reifers et al., 1998) as previously described (Harland, 1991). Standard methods for hybridization and for single color labeling were used as described (Sagerstrom et al., 1996). After staining, embryos were de-yolked, flatmounted in glycerol and imaged with a Nikon compound microscope. (G-L) Immunohistochemistry staining for the apical junction marker ZO-1 (G-I) and the cell polarity maker aPKC (J-L) in control MO (G,J), wnt5b MO (H.K), and fak MO (I,L) injected embryos at prim-6. Apical localization of ZO-1 appears normal in each condition and indicates establishment of apical junctions. aPKC is also normal in each condition indicating that the cells have established cell polarity. For immunostaining experiments, embryos were fixed in 4% paraformaldehyde or Dent’s (70% methanol: 30% DMSO) for ZO-1. Embryos were blocked in 2% normal goat serum, 1% BSA, and 0.1% Triton-X100 in PBT; incubated overnight at 4C in primary antibody (anti-aPKC (C-20), SC-216, Santa Cruz Biotechnology, 1:1000; anti-ZO1, 33- 9100, Invitrogen, 1:200); then incubated in secondary antibody (goat anti-rabbit or anti- mouse IgG conjugated with Alexa Fluor 488, Invitrogen, 1:500). Arrowheads indicate MHBC. Scale bars: G-I, 20 m; J-L, 35 m. |
|
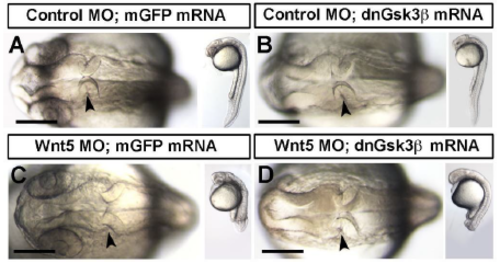
Gross morphology images of dnGsk3 and wild-type Gsk3 overexpression phenotypes and dnGsk3 rescue of wnt5b morphants. (A-D) Brightfield dorsal and lateral images of control MO (A,B) and wnt5b MO (C,D) injected embryos, co-injected with mGFP (A,C), or dnGsk3 (B,D) mRNA. (A) Control morphants co-injected with mGFP mRNA demonstrating a normal MHBC basal constriction phenotype. (B) Control morphants co-injected with dnGsk3 mRNA exhibit an eyeless phenotype, but undergo basal constriction normally with this concentration of dnGsk3 (n=9). (C) Wnt5b morphants co-injected with control mRNA exhibit abnormal MHBC morphogenesis, tail defects, and fail to undergo basal constriction (n=6). (D) Wnt5b morphants co-injected with dnGsk3 mRNA exhibit a loss of eyes and tail defects, but the gross morphology of basal constriction is rescued and occurs normally (n=6). Arrowheads indicate the MHBC. Scale bars: 100m. |
|
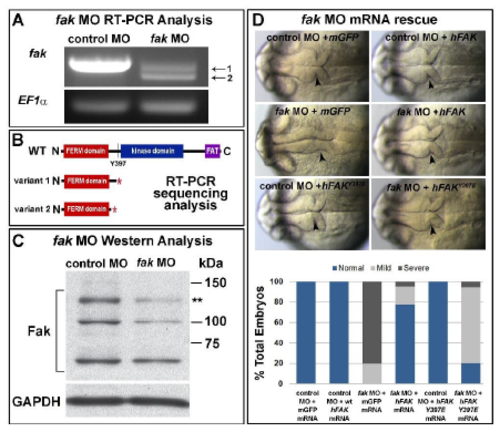
fak MO efficacy and specificity. (A) RT-PCR for control MO and fak MO injected embryos. Whole tissue lysate was analyzed. fak MO injection resulted in 2 abnormal mRNA products (arrows 1 and 2). EF1 was used as an RT-PCR control. Primers used for RT-PCR: fak exon 11 forward 5-CACCTTGCCAACTTCACTCA-3; fak exon 22 reverse 5- GTGAATCGTGGGCGTTTACT-3; EF1 forward, 5-GATGCACCACGAGTCTCTGA-3; and EF1 reverse, 5-TGATGACCTGAGCGTTGAAG-3. fak RT-PCR products were cloned into pGEM using the pGEM T-Easy Vector System Kit (Promega) and sequenced. (B) Sequence analysis of the two RT-PCR product variants from (A) resulted in the detection of two truncated mRNA amplicons caused by partial or complete exon deletion, each resulting in early stop codons as indicated in the diagram. Both truncations eliminate the autophosphorylation site Y397. (C) Western Blot analysis of fak morphant lysate demonstrating downregulation of full length Fak (asterisks ** at 125 kDa). Injected embryos were manually dechorionated and deyolked. Proteins were analyzed on 8% SDS-PAGE gels. Blots were blocked in 4% non-fat milk or 5% BSA and probed with antibody in 3% BSA. FAK C-20 (1:1000 dilution; sc-558) and FAK (1:200 dilution; Invitrogen AHO0502). GAPDH was used as a loading control (1:25,000 dilution, ab22555). Blots were visualized using Enhanced Chemiluminscence. (D) Representative brightfield images and quantification of fak morphant embryo rescue experiments. Control MO or fak MO was co-injected with mGFP, wild-type human FAK, or human FAKY397E mRNA. Each condition had equal total amounts of mRNA injected. Embryos were analyzed at prim-6 for MHB defects. Each embryo was scored as having a normal, mild, or severe phenotype. Mild phenotypes are represented in D. Control MO + mGFP (n=38), control MO + wt hFAK (n=42), fak MO + mGFP (n=55), fak MO + hFAK (n=63), control MO + hFAKY397E (n=51), fak MO + hFAKY397E (n=54). Anterior is to the left in all images. Arrowheads indicate MHBC. |
|
Fak is required at the MHBC for basal constriction. Schematic for transplant procedure. 1. One-cell stage wild-type embryos were co-injected with mGFP (donor) or mCherry (host) and control or fak MO. 2. Cells from donors (sphere stage) were transplanted into hosts (shield stage). Transplanted cells were targeted to the presumptive MHB region (Woo and Fraser, 1995). 3. Embryos were incubated until prim-6 then imaged with live confocal microscopy. (E-F’) Donor cells are outlined in yellow and host cells in blue. (B,B’) Control donor and control host cells basally constricted normally when transplanted to the MHBC, (n=6). (C,C’) Cells from fak morphant donors transplanted into control hosts failed to undergo basal constriction at the MHBC, (n=4). Basal constriction occurred normally in control host cells, even when immediately adjacent to fak morphant donor cells. |
|
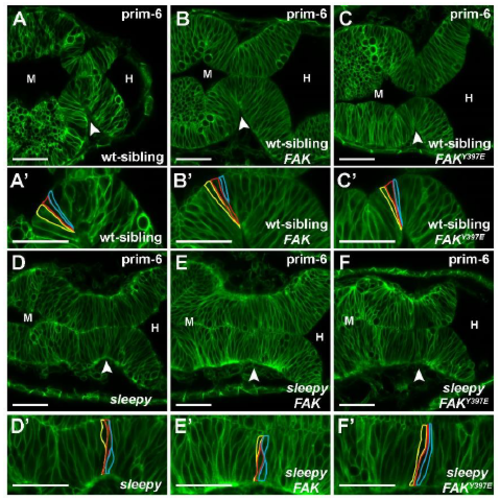
Expression of human FAK does not rescue the laminin mutant basal constriction phenotype. (A-F’) Live confocal images showing the MHB region of prim-6 embryos injected with mGFP (A,D), mGFP + wt FAK mRNA (B,E), mGFP + FAK-Y397E (C,F). (A-C’) sleepy (slym86) (Schier et al., 1996) heterozygous sibling or wild-type sibling embryos showing normal basal constriction at the MHBC. (D-F’) sleepy mutants showing defects in basal constriction both without and with the co-injection of human wild-type FAK and human phosphomimetic FAKY397E mRNA. Representative images from 3 independent experiments with n>3 for each condition. (A’-F’) Magnifications of the neuroepithelium shown in A-F with individual cells outlined at the MHBC. Arrowheads indicate MHBC. M, midbrain. H, hindbrain. Scale bars: 35 m. Biology Open (2018): doi:10.1242/bio.034520: Supplementary information Biology Open • Supplementary information |