- Title
-
Organ-specific requirements for Hdac1 in liver and pancreas formation
- Authors
- Noël, E.S., Casal-Sueiro, A., Busch-Nentwich, E., Verkade, H., Dong, P.D., Stemple, D.L., and Ober, E.A.
- Source
- Full text @ Dev. Biol.
|
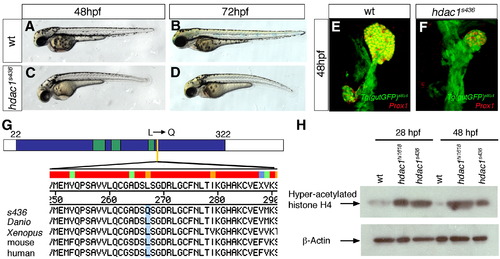
Mutant line s463 encodes a novel hdac1 allele. Lateral brightfield views of sibling (A, B) and hdac1s436 mutant (C, D) embryos at 48 hpf and 72 hpf, anterior to the left. (E, F) Projections of confocal stacks showing ventral views of Tg(gutGFP)s854, anterior to the top. (E) Wild type embryo at 48 hpf, expressing Prox1 (red) in the liver and exocrine pancreas. (F) s436 mutant exhibits a reduced liver and absent exocrine pancreas at 48 hpf. (G) s436 encodes a novel allele of Hdac1. A T to A base pair change at position 800 results in a leucine to glutamine transition at position 267. Schematic representation of the predicted zebrafish Hdac1 derived from alignment of vertebrate sequences; catalytic domain is depicted in blue, HDAC superfamily signature sequences in green, s436 lesion in yellow. Protein alignments with amino acid transition shown in blue. (H) Western blot showing increased levels of acetylated histone H4 in both hdac1hi1618 and hdac1s436 mutant embryos at 28 hpf and 48 hpf. β-actin levels were used as loading control. |
|
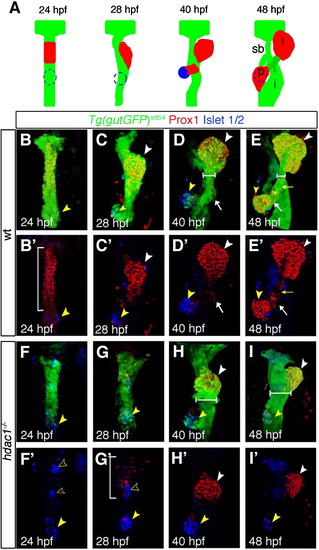
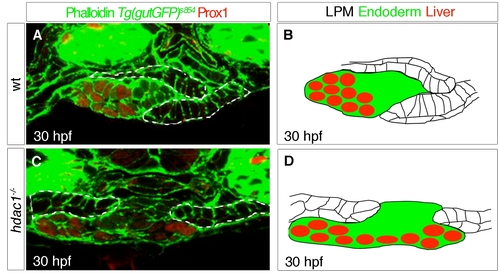
Specific requirements for Hdac1 in hepatic and pancreatic development. (A) Schematic depicting stages of endodermal organogenesis in wild type. Endoderm in green, Prox1-positive liver and exocrine pancreas in red, and endocrine pancreas in blue. l, liver; p, pancreas; dotted line, endocrine pancreas; sb, swim bladder; and i, intestinal bulb. (B–I′) Time course analysis comparing endodermal organogenesis of wild type siblings and hdac1 mutants, using the Tg(gutGFP)s854 line stained for Prox1(red) and Islet 1/2 (blue). Projection of confocal stacks showing ventral views of the Tg(gutGFP)s854 line expressing GFP throughout the endoderm (green), anterior to top. (B–E′), Wild type siblings, (F–I′), hdac1 mutants. (B, B′) At 24 hpf Prox1-expressing hepatoblasts are present throughout the organ-forming region of the endodermal rod (bracket). The endocrine pancreatic islet, stained with Islet 1/2, is situated posteriorly in the organ-forming region (yellow arrowhead). (C, C′) By 28 hpf the Prox1-positive hepatoblasts aggregate on the left side of the endodermal rod (white arrowhead) forming the liver bud. The exocrine pancreas expresses Prox1 at 40 hpf (arrow, D, D′) and 48 hpf; the liver and ventral pancreas are connected to the adjacent digestive tract by the extrahepatopancreatic duct (yellow arrow, E, E′). (F, F′) hdac1 mutants lack Prox1 expression at 24 hpf. The endocrine islet is present, although anterior groups of endocrine cells have formed (empty arrowheads). By 28 hpf, Prox1 expression is initiated in hepatoblasts (bracket, G, G′) which subsequently aggregate to form a medial liver bud by 40 hpf (white arrowhead, H, H′). By 48 hpf, in hdac1 mutants, the liver bud is located asymmetrically on the left side of the digestive tract (white arrowhead, I, I′). Exocrine pancreas and swim bladder fail to form at this time. Additionally, the width of the digestive tract is greater in hdac1 mutants than in wild type embryos once liver bud outgrowth has been initiated (compare horizontal bars, D, E with H, I). |
|
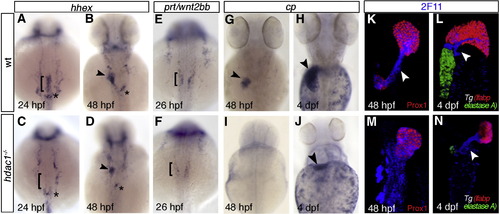
Hdac1 is required for timely liver specification and differentiation. (A–J) In situ hybridisation analyses of hhex, prt/wnt2bb and cp expression in sibling and hdac1 mutant embryos; dorsal views, anterior to the top. (A, B) Wild type embryos express hhex in hepatoblasts at 24 hpf (bracket, A) and 48 hpf (arrowhead, B). Additionally, hhex is expressed in pancreatic tissue (asterisk). (C) hdac1 mutants lack hepatic hhex, though retain pancreatic expression at 24 hpf. At 48 hpf hhex expression is present in the liver of hdac1 mutants, although in a reduced domain (arrowhead, D). (E, F) Wild type embryos express prt/wnt2bb in the LPM at 26 hpf (bracket, E), however hdac1 mutants show reduced prt/wnt2bb expression (bracket, F). (G–J) cp is expressed in differentiating hepatocytes at 48 hpf and 4 dpf in wild type embryos (arrowheads, G, H). hdac1 mutants lack cp expression at 48 hpf (I); a subset of embryos express cp at 4 dpf (arrowhead, J). (K–N) Ventral projection of confocal stacks. Analysis of Tg(gutGFP)s854 embryos (K, M; GFP not shown) and Tg(lfabp:dsRed;elastaseA:eGFP) embryos (L, N) stained for 2F11 (blue) and Prox1 (red) reveals that in wild type embryos 2F11 expression is heightened in the hepatopancreatic ducts at 48 hpf (arrowhead, K) and 4 dpf (arrowhead L). hdac1 mutant embryos express 2F11 throughout the organ-forming region at 48 hpf (M), however expression is heightened in the duct at 4 dpf (N). In addition, wild type embryos express dsRed in the liver and GFP in the pancreas at 3 and 4 dpf (L) while only a subset of hdac1 mutants express dsRed and GFP at 4 dpf (N). |
|
Cell-autonomous requirement for Hdac1 in hepatocyte differentiation. Ventral views of wild type (A) and hdac1 mutant (B) organ-forming region at 48 hpf using the Tg(gutGFP)s854 line. (C, D) Morphological analysis of cell transplantation experiments; wild type cells (red) transplanted into hdac1 mutants expressing the Tg(gutGFP)s854 transgene. (C) Wild type cells contributing to the mesoderm fail to rescue liver morphology in hdac1 mutants. (D) Wild type cells contributing to the endoderm of hdac1 mutants rescue liver morphology. (E–P) Cell transplantation experiments were analysed by fluorescent in situ hybridisation in conjunction with immunostaining in transverse sections at liver level, using the Tg(gutGFP)s854 line (blue) to highlight the endoderm, cp (red) to assess hepatocyte differentiation, and transplanted wild type cells labelled with fluorescein (green). (E, H, K, N) Wild type embryos express cp in the liver at 48 hpf. Wild type cells contributing to the hepatic endoderm of hdac1 mutants express cp (arrowhead, F, I, L, G, J, M), however wild type cells contributing to the adjacent digestive tract fail to express cp (F, G). To validate that transplanted host cells injected with cas mRNA contribute to the endoderm, Tg(gutGFP)s854 embryos were used as donors in a subset of transplants (E, N, G, P); due to the mild mosaicism of the Tg(gutGFP)s854 expression, we observe in a subset of transplanted cells very low GFP expression. Asterisks indicate liver, white dashed lines outline the digestive tract. EXPRESSION / LABELING:
|
|
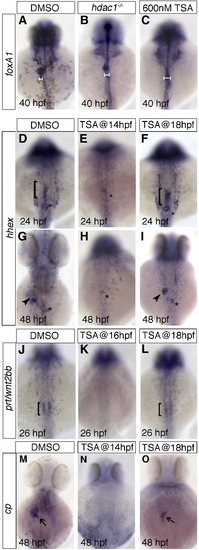
Histone deacetylase function is required during late somitogenesis for liver specification and differentiation. TSA was applied at various time points, and digestive tract morphogenesis, liver specification and differentiation were examined by foxA1, hhex, prt/wnt2bb, and cp expression, respectively. 600 nM TSA produced the closest phenocopy of the hdac1s436 phenotype when assessed by foxA1 expression (compare panel B and C). This concentration was used in subsequent experiments. Horizontal bars show an expansion of the digestive tract in both TSA treated and hdac1 mutants when compared to wild type embryos (compare panels B, C with A). (D–I) Hepatoblast specification in TSA treated embryos was assessed by hhex expression. Application of TSA at 14 hpf resulted in an absence of hepatic hhex expression at both 24 and 48 hpf (E, H). However, treatment at 18 hpf resulted in wild type expression of hepatic hhex at 24 hpf (bracket, F), although strongly reduced at 48 hpf (arrowhead, I). (J–L) prt/wnt2bb expression was assessed in TSA treated embryos. TSA treatment at 16 hpf resulted in an absence of prt/wnt2bb expression in the LPM abutting the organ-forming endoderm (K), however application of TSA at 18 hpf resulted in wild type-like expression of prt/wnt2bb (bracket, L). (M–O) Hepatocyte differentiation in TSA treated embryos was assessed by cp expression. Embryos treated with TSA at 14 hpf showed no cp expression at 48 hpf (N), however embryos treated at 18 hpf express cp, but in a reduced domain (arrow, O). DMSO was added as a control in all cases (A, D, G, J, M), asterisk indicates position of the pancreas. Anterior to the top. |
|
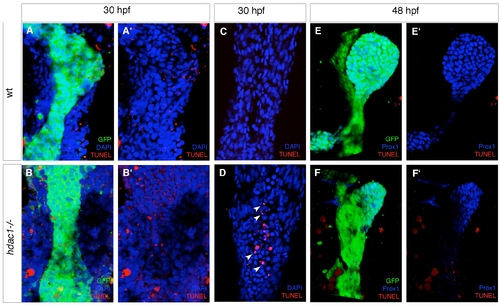
Loss of hdac1 results in reduced cell proliferation in the organ-forming endoderm. (A–D′) Ventral projections using Tg(gutGFP)s854 line, stained for PH3-positive cells (red) and Prox1-positive hepatoblasts (blue) at 24 hpf (A, C), and 48 hpf (B, D); dorsal to the top. (E–H) In situ hybridisation analysis of pes expression. Wild type embryos express pes in the organ-forming region at 24 hpf (bracket, E) and hepatic region at 30 hpf (arrowhead, F). hdac1 mutants lack pes expression in the organ-forming endoderm at both 24 and 30 hpf (brackets, G, H). (I, J) hdac1 mutants display a decreased number of PH3-positive cells the endoderm and liver between 24 and 48 hpf. Numbers are supplied in Table 1 and Table 2. |
|
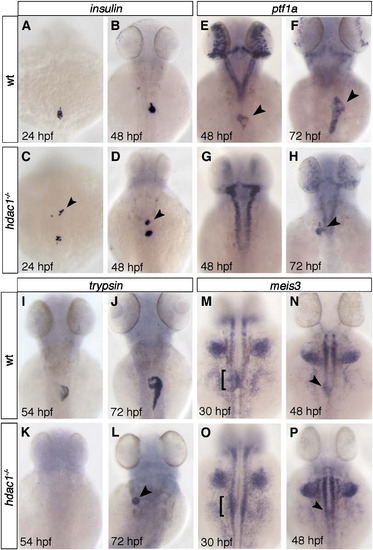
Hdac1 depletion results in ectopic endocrine tissue formation and defects in exocrine pancreas specification. (A–D) insulin is expressed in a single cluster of endocrine β-cells in wild type embryos at 24 and 48 hpf (A, B). hdac1 mutants display ectopic anterior clusters of insulin-expressing cells at 24 and 48 hpf (arrowheads, C, D). (E–H) ptf1a is expressed in the exocrine pancreas at 48 and 72 hpf (arrowhead, E, F). hdac1 mutant embryos lack pancreatic ptf1a expression at 48 hpf (G), but express ptf1a at 72 hpf in a reduced domain (arrowhead, H). (I–L) trypsin expression in the exocrine pancreas in wild type siblings at 54 hpf (I) is absent in hdac1 mutants (K), however is detected in a subset of hdac1 mutants at 72 hpf (arrowhead, L). (M–P) Wild type embryos express meis3 medially in the LPM at 30 hpf (bracket, M) and 48 hpf (arrowhead, N). hdac1 mutants display meis3 expression bilateral to the endoderm at 30 hpf (bracket, O) and medially at 48 hpf (P). EXPRESSION / LABELING:
|
|
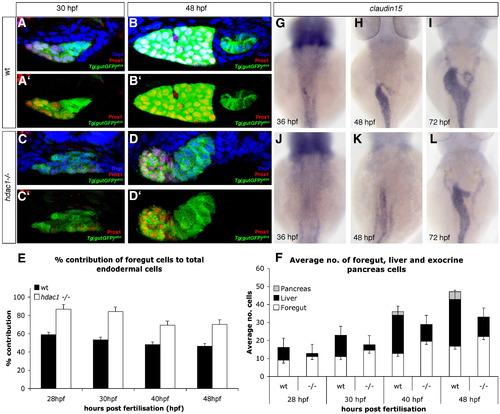
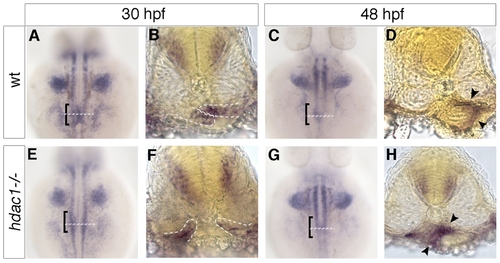
Loss of Hdac1 function results in an increase of non-hepatic foregut endoderm. (A–D) Transverse sections taken through the level of the liver, at 30 hpf and 48 hpf in wt (A–B′) and hdac1 mutants (C–D′). To count endodermal cells, Tg(gutGFP)s854 embryos were stained for Prox1 (red), highlighting hepatoblasts, and Dapi (blue). (E) hdac1 mutants display an increased percentage contribution of foregut cells to total endodermal cells between 28 hpf and 48 hpf when compared to wild type siblings. (F) hdac1 mutant embryos have an increased number of foregut cells compared to wild type embryos between 28 and 48 hpf (white bars). However, hdac1 mutant embryos display an overall reduction in number of total endodermal cells when compared to wild type embryos (compare total bar height). (E, F) Number of wild type embryos analysed: 28 hpf n = 12, 30 hpf n = 14, 40 hpf n = 13, 48 hpf n = 13. Number of hdac1 mutants analysed: 28 hpf n = 10, 30 hpf n = 11, 40 hpf n = 10, 48 hpf n = 10. (G–L) hdac1 is required for correct temporal expression of claudin15. Wild type embryos express claudin15 in the endoderm from 36 hpf onwards (G–I), whereas hdac1 mutants fail to express claudin15 at 36 hpf (J), however, expression is detected at 48 hpf (K) and 72 hpf. EXPRESSION / LABELING:
|
|
|
|
|
|
|
Reprinted from Developmental Biology, 322(2), Noël, E.S., Casal-Sueiro, A., Busch-Nentwich, E., Verkade, H., Dong, P.D., Stemple, D.L., and Ober, E.A., Organ-specific requirements for Hdac1 in liver and pancreas formation, 237-250, Copyright (2008) with permission from Elsevier. Full text @ Dev. Biol.