Fig. 3
|
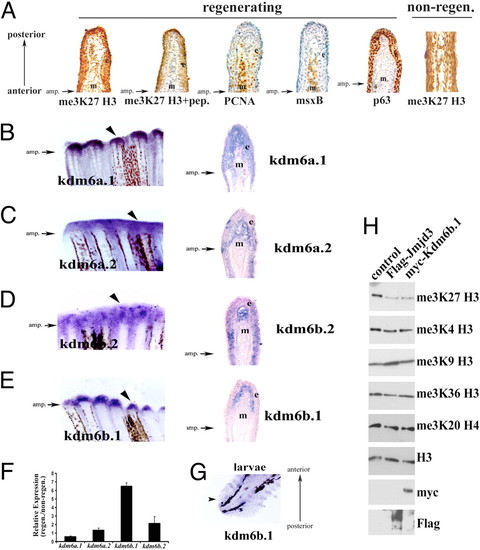
me3K27 H3 demethylases in caudal fin. (A) Global levels of me3K27 H3 remain intact during regeneration. Immunohistochemistry of longitudinal sections of 48 hpa regenerating caudal fins. From left to right, me3K27 H3 antibody; me3K27 H3 antibody with peptide competition; msxB; PCNA; p63. The right-most panel is a non-regenerating caudal fin stained for me3K27 H3. Antibody signal (brown) and hematoxylin counterstain (blue) are shown. (B–E) Expression of putative me3K27 H3 demethylases in regenerating caudal fins. Whole mount in situ hybridization of regenerating caudal fins at 48 h post amputation (B) kdm6a.1, (C) kdm6a.2, (D) kdm6b.2, and (E) kdm6b.1. Longitudinal sections of the fins in (B–E) reveal unique expression patterns and are shown in the right panels. The epidermis (e) and mesenchyme (m) are indicated in each panel. (F) Upregulation of kdm6b.1 during regeneration. The expression of putative me3K27 H3 demethylases was examined in non-regenerating and regenerating (48 hpa) adult caudal fins by qPCR and normalized to Ribosomal Protein L18 expression. Data are expressed as average expression levels relative to non-regenerating fins from two different cohorts (12 animals each) and error bars represent the deviation from the means derived from each cohort. Similar results were obtained in several independent experiments. (G) kdm6b.1 expression in larvae caudal fin at 24 hpa [72 h post fertilization (hpf)] by in situ hybridization. (H) Kdm6b.1 demethylates me3K27 H3. myc-tagged Kdm6b.1 was immunoprecipitated from transfected 293T cells and subjected to a histone demethylase assay, followed by immunoblotting. Some panels contain blots that were stripped and re-probed with different antibodies. |
| Genes: | |
|---|---|
| Antibodies: | |
| Fish: | |
| Condition: | |
| Anatomical Terms: | |
| Stage: | Day 4 |