- Title
-
Fast revascularization of the injured area is essential to support zebrafish heart regeneration
- Authors
- Marín-Juez, R., Marass, M., Gauvrit, S., Rossi, A., Lai, S.L., Materna, S.C., Black, B.L., Stainier, D.Y.
- Source
- Full text @ Proc. Natl. Acad. Sci. USA
|
The zebrafish heart displays fast revascularization of the damaged area. (A-C) Tg(fli1a:EGFP) ventricles at 0, 15, and 40 hpi. White arrowheads point to coronary vessel sprouts in the injured area; asterisk marks an area of coronary plexus (n = 4). Insets show high-magnification images of early coronary sprouts (B) and the coronary plexus (C). (D) Tg(kdrl:EGFP) heart at 15 hpi (n = 3). (E and F) Tg(fli1a:EGFP);Tg(kdrl:mCherry) ventricles extracted at 0 hpi and cultured in explant medium for 24 h (0+24 hpi) (n = 3). White arrowhead points to kdrl:EGFP+ vessels sprouting into the injured area. Inset shows overlay image of a sprouting coronary vessel. White arrows point to fli1a:EGFP+/kdrl:mCherry+ coronary vessel sprouting into the injured area. (G) Tg(Dll4in3:TagRFP) ventricle at 40 hpi (n = 5). Inset shows a Dll4in3:TagRFP+ sprouting coronary artery. (H and I) Tg(cxcr4a:YFP) ventricles at 40 hpi (n = 3) and 7 dpi (n = 4). Insets show cxcr4a:YFP+ sprouting coronaries; white arrowheads point to coronary arteries sprouting into the injured area. Dotted lines delineate the injured area. (Scale bars: 50 µm.) EXPRESSION / LABELING:
|
|
vegfaa mutants exhibit delayed and disorganized revascularization. (A) qPCR analysis of vegfaa expression at 6 hpi and 1, 2, 3, and 5 dpi. Data are expressed as mean ± SD, *P < 0.05 (n = 4). (B and C) Tg(etv2:EGFP) vegfaa-/- ventricles at 40 hpi (n = 3). White arrowheads point to coronary vessels sprouting into the injured area. (D and E) Tg(etv2:EGFP) WT (n = 5) and Tg(etv2:EGFP) vegfaa-/- ventricles at 4 dpi (n = 3). Asterisk marks an area of coronary plexus. Dotted lines delineate the injured area. (F and G) Sections of WT (n = 4) and vegfaa-/- (n = 4) ventricles at 45 dpi stained with AFOG to identify scar (blue), fibrin (red), and muscle (orange). (H) Microarray profiling of vegfaa+/+ and vegfaa-/- ventricles. Data are shown relative to vegfaa+/+ ventricles, which are set at 1 (dashed line). (Scale bars: B–E, 50 µm; F and G, 100 µm.) |
|
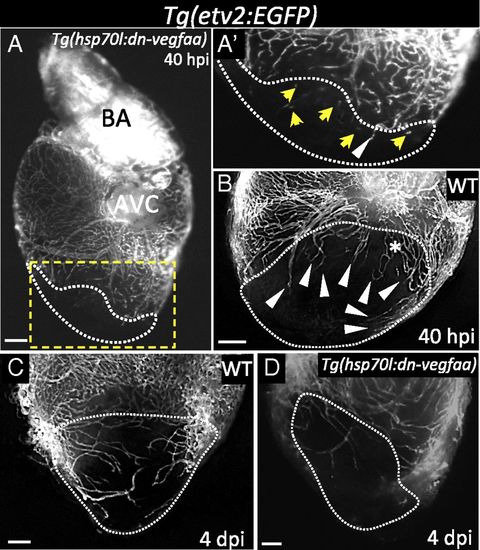
Induction of dn-vegfaa expression blocks revascularization of the damaged area. (A) Tg(hsp70l:dn-vegfaa);Tg(etv2:EGFP) ventricle at 40 hpi (n = 5). (A′) Inset shows high-magnification image of the injured area. (B) Tg(etv2:EGFP) WT ventricle at 40 hpi (n = 5). (C) Tg(etv2:EGFP) WT ventricle at 4 dpi (n = 5). (D) Tg(hsp70l:dn-vegfaa);Tg(etv2:EGFP) heart at 4 dpi (n = 3). White arrowheads point to coronary vessels sprouting into the injured area; yellow arrows point to disconnected endothelial cells; asterisk mark an area of coronary plexus. Dotted lines delineate the injured area. AVC, atrioventricular canal; BA, bulbus arteriosus. (Scale bar: 100 µm.) |
|
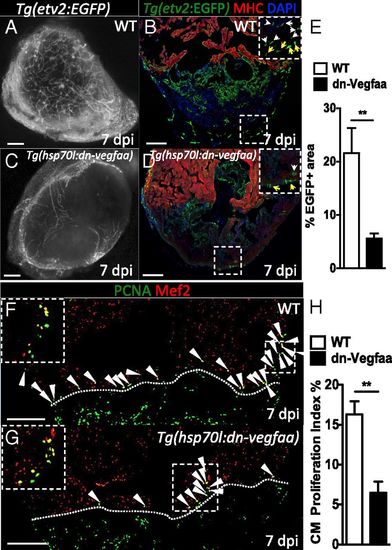
Revascularization of the damaged area supports CM proliferation. (A and C) Images of the apex of Tg(etv2:EGFP) WT and Tg(hsp70l:dn-vegfaa);Tg(etv2:EGFP) ventricles at 7 dpi. WT and Tg(hsp70l:dn-vegfaa) were daily heatshocked. (B and D) Sections of Tg(etv2:EGFP) WT and Tg(hsp70l:dn-vegfaa);Tg(etv2:EGFP) ventricles. CMs are immunostained with anti-MHC antibody (red), and nuclei counterstained with DAPI (blue). Insets show high-magnification images of the coronaries on the surface of the heart (yellow arrows) and in the damaged area (white arrows). (E) Quantification of coronary vessel formation in the injured area. The percentage of etv2:EGFP+ area was measured relative to total injured area in WT ventricles (n = 6) and Tg(hsp70l:dn-vegfaa) ventricles (n = 5). (F and G) Sections from Tg(etv2:EGFP) WT and Tg(hsp70l:dn-vegfaa);Tg(etv2:EGFP) ventricles stained with anti-Mef2 (red) and anti-PCNA (green) antibodies. Arrowheads point to Mef2+/PCNA+ nuclei. Insets show high-magnification images of Mef2+/PCNA+ nuclei. (H) Quantification of CM proliferation in WT (n = 5) and Tg(hsp70l:dn-vegfaa) (n = 5) ventricles at 7 dpi. Data are expressed as mean ± SEM, **P < 0.001. Dotted lines delineate the injured area. (Scale bars: 100 µm.) |
|
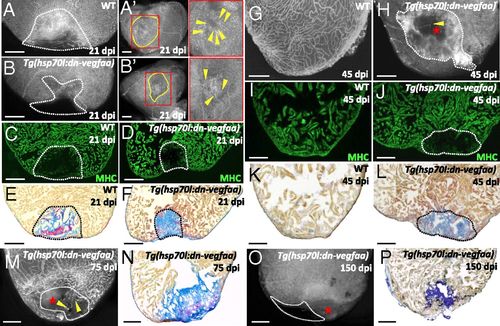
Blockade of early revascularization prevents vascular invasion and heart regeneration. (A and B) Tg(etv2:EGFP) WT (n = 5) and Tg(hsp70l:dn-vegfaa);Tg(etv2:EGFP) (n = 5) ventricles at 21 dpi. (A¹ and B′) View of the apex of Tg(etv2:EGFP) WT and Tg(hsp70l:dn-vegfaa);Tg(etv2:EGFP) ventricles at 21 dpi. Yellow dotted lines delineate the injured area. Insets (red dotted boxes) show high-magnification images of the injured area. Yellow arrowheads point to coronaries in the injured area. (C and D) Sections of Tg(etv2:EGFP) WT (n = 5) and Tg(hsp70l:dn-vegfaa);Tg(etv2:EGFP) (n = 5) ventricles at 21 dpi. CMs are immunostained with anti-MHC antibody (green). (E and F) Sections of WT (n = 4) and Tg(hsp70l:dn-vegfaa) (n = 5) ventricles at 21 dpi stained with AFOG to identify collagen (blue) and fibrin (red) deposition. (G and H) Tg(etv2:EGFP) WT (n = 5) and Tg(hsp70l:dn-vegfaa);Tg(etv2:EGFP) (n = 4) ventricles at 45 dpi. (I and J) Sections of Tg(etv2:EGFP) WT (n = 5) and Tg(hsp70l:dn-vegfaa);Tg(etv2:EGFP) (n = 4) ventricles at 45 dpi. CMs are immunostained with anti-MHC antibody (green). (K and L) Sections of WT (n = 4) and Tg(hsp70l:dn-vegfaa) (n = 4) ventricles at 45 dpi stained with AFOG to identify scar (blue) and fibrin (red) deposition. (M and O) Tg(hsp70l:dn-vegfaa);Tg(etv2:EGFP) ventricles at 75 (n = 4) and 150 (n = 4) dpi. (N and P) Sections of Tg(hsp70l:dn-vegfaa) ventricles at 75 (n = 4) and 150 (n = 4) dpi stained with AFOG. Dotted lines delineate the injured area. Red stars mark nonvascularized areas. (Scale bars: 100 µm.) |
|
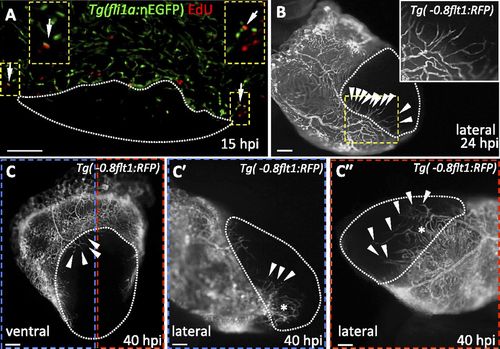
Revascularization of the damaged area starts mainly from the dorsal half of the ventricle. (A) Section from a Tg(fli1a:nEGFP) ventricle stained for EdU incorporation. Arrows point to fli1a:nEGFP+/EdU+ nuclei in the ventricular wall. Insets show high-magnification images of fli1a:nEGFP+/EdU+ nuclei. (B) Lateral view of 24 hpi Tg(-0.8flt1:RFP) ventricle. Inset shows high-magnification image of vessels sprouting from preexisting coronaries. (C) Ventral view of 40 hpi Tg(-0.8flt1:RFP) ventricle. (C′ and C′′) Lateral views of 40 hpi Tg(-0.8flt1:RFP) ventricle. Arrowheads point to coronary vessels sprouting into the injured area; asterisk marks an area of coronary plexus (n = 5 ventricles examined). Dotted lines delineate the injured area. Arrowheads point to coronaries sprouting into the injured area. (Scale bars: 100 µm.) |
|
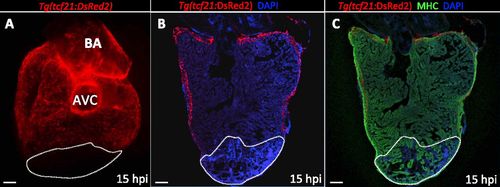
No epicardial cells are found in the damaged area at 15 hpi. (A) Dorsal view of a 15 hpi Tg(tcf21:DsRed2) ventricle (n = 3). (B and C) Sections of a Tg(tcf21:DsRed2) ventricle at 15 hpi (n = 3). Cardiomyocytes are immunostained with anti-MHC antibody (red), and nuclei are counterstained with DAPI (blue). White dotted lines delineate the injured area. AVC, atrioventricular canal; BA, bulbus arteriosus. (Scale bars: 100 µm.) |
|
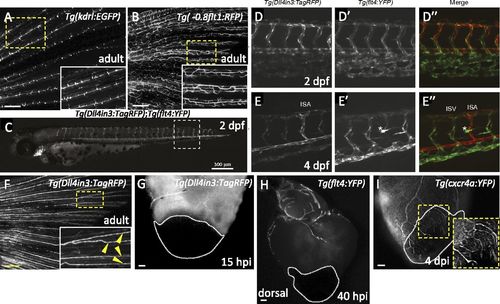
Expression patterns of different arterial and venous markers at different developmental stages and after injury. (A and B) Images of Tg(kdrl:EGFP) (n = 4) and Tg(-0.8flt1:RFP) (n = 4) adult caudal fins. Insets show high-magnification images of the fin rays displaying Tg(kdrl:EGFP) expression only in the medial arteries (A), or Tg(-0.8flt1:RFP) (B) expression in arteries and veins. (C) At 2 dpf, Tg(Dll4in3:TagRFP) expression is visible throughout the arterial endothelium, the dorsal aorta, intersegmental vessels, endocardium, branchial arch arteries, and cranial arterial endothelium. (D and E) Detailed views of posterior body segments of Tg(Dll4in3:TagRFP) (an arterial-specific EC reporter) and Tg(flt4:YFP) (a venous and lymphatic EC-specific reporter) animals. At 2 dpf, Tg(Dll4in3:TagRFP) expression is clearly restricted to the dorsal aorta, but is also present in all intersegmental vessels (D). Tg(flt4:YFP) is preferentially expressed in the vein plexus, but coexpressed with Tg(Dll4in3:TagRFP) in intersegmental vessels and the dorsal aorta (D′). At 4 dpf, arterial Tg(Dll4in3:TagRFP) and venous Tg(flt4:YFP) expression are mutually exclusive (E and E′). In the trunk, Tg(Dll4in3:TagRFP) expression is restricted to the dorsal aorta and to intersegmental vessels that by now have become specified as arterial (E). In contrast, Tg(flt4:YFP) expression is only observed in the posterior cardinal vein and venous intersegmental vessels (vISVs) (E′). At this time, Tg(flt4:YFP) is also expressed in lymphatic vessels (asterisk) (E′ and E′′). (F) Image of a Tg(Dll4in3:TagRFP) adult caudal fin (n = 6). Inset shows high-magnification image of the arteries in the fin rays displaying Dll4in3:TagRFP expression (yellow arrowheads). (G) Dorsal view of Tg(Dll4in3:TagRFP) ventricle at 15 hpi (n = 5). (H) Dorsal view of Tg(flt4:YFP) ventricle at 40 hpi (n = 5). (I) Lateral view of Tg(cxcr4a:YFP) ventricle at 4 dpi (n = 4). Inset shows cxcr4a:YFP+ sprouting coronaries. Dotted lines delineate the injured area. (Scale bars: A, B, and F, 100 µm; C, 300 µm; G-I, 50 µm.) |
|
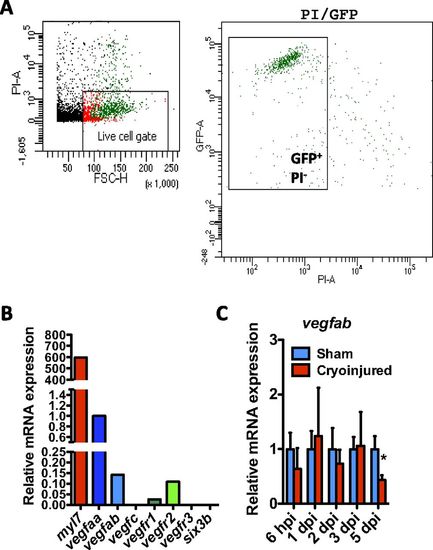
Expression of Vegf family members in zebrafish CMs. (A) FACS isolation of EGFP+ CMs from uninjured Tg(myl7:EGFP) adult zebrafish hearts. Cellular debris were excluded, and the gated population of EGFP+/propidium iodide (PI)- cells was sorted. (B) qPCR analysis of myl7 (as a positive control), vegfaa, vegfab, vegfc, vegfr1, vegfr2, vegfr3, and six3b (as a negative control) expression in FACS-sorted myl7:EGFP+ CMs. (C) qPCR analysis of vegfab expression at 6 hpi and 1, 2, 3, and 5 dpi. Data (mean ± SD) are shown relative to their respective sham controls. *P < 0.05 (n = 4, each replicate consists of a pool of 4 ventricles). |
|
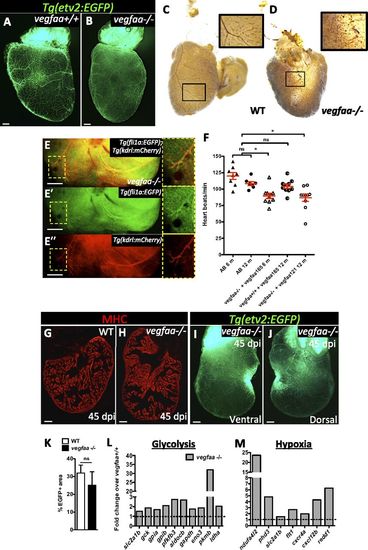
vegfaa-/- hearts exhibit features of myocardial hibernation and can regenerate. (A and B) Ventral views of Tg(etv2:EGFP) vegfaa+/+ and vegfaa-/- adult hearts. (C and D) Erythrocytes in WT and vegfaa-/- hearts stained with O-dianisidine to assess vascular integrity. Insets show high-magnification images of coronaries containing erythrocytes. (E-E′′) Dorsal view of Tg(fli1a:EGFP);Tg(kdrl:mCherry) vegfaa-/- ventricle. Insets show fli1a:EGFP+ and kdrl:mCherry+ coronaries. (F) Doppler measurements of heart rate in WT fish (AB) of 6 (n = 8) and 12 (n = 8) months of age, 6-mo-old vegfaa-/- (n = 10) and 12-mo-old vegfaa+/+ (n = 10) injected with 200 pg of vegfaa 165, and 12-mo-old vegfaa-/- (n = 10) injected with 50 pg of vegfaa 121. Each data point represents a single fish. Mean ± SEM is indicated in red. ns, no significant differences. *P < 0.05. (G and H) Sections of WT (n = 4) and vegfaa-/- (n = 4) hearts at 45 dpi. Cardiomyocytes are immunostained with anti-MHC antibody (red). (I and J) Ventral and dorsal views of a Tg(etv2:EGFP) vegfaa-/- heart at 45 dpi. (K) Percentage of etv2:EGFP+ area in the apex of WT (n = 5) and Tg(hsp70l:dn-vegfaa) ventricles (n = 3) at 45 dpi. (L and M) Differentially expressed genes involved in glycolysis or stimulated by hypoxia obtained by microarray profiling of vegfaa+/+ and vegfaa-/- adult ventricles. Data are expressed relative to vegfaa+/+ ventricles (dashed lines). (Scale bars: 100 µm.) |
|
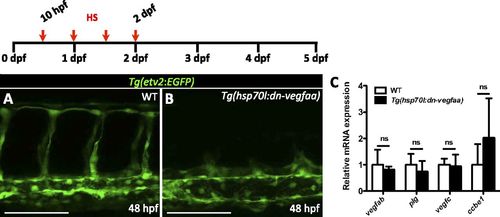
dn-vegfaa blocks angiogenesis. (A and B) High-magnification images of the trunk of 48 hpf Tg(etv2:EGFP) WT and Tg(hsp70l:dn-vegfaa);Tg(etv2:EGFP) larvae heatshocked for 1 h every 12 h from tailbud stage until 48 hpf. (C) qPCR analysis of vegfab, plg, vegfc, and ccbe1 expression in ventricles from WT and Tg(hsp70l:dn-vegfaa) fish heatshocked for 4 d. Data (mean ± SD) are shown relative to wild types. (n = 4, each replicate consists of a pool of 4 ventricles). ns, no significant differences. (Scale bars: 100 µm.) |
|
Cryoinjury elicits a heatshock-like response. Bright field (A-E) and fluorescent images (A′-E′) of Tg(hsp70l:GFP-podxl) hearts untouched, 1 d after sham, 1 dpi, 2 dpi, and 1 d after 1-h 37 °C heatshock. Dotted lines delineate the injured area. (F) qPCR analysis of hsp70l expression at 6 hpi and 1, 2, 3, and 5 dpi. Data (mean ± SD) are expressed relative to their respective sham controls. *P < 0.05 (n = 4, each replicate consists of a pool of 4 ventricles). |
|
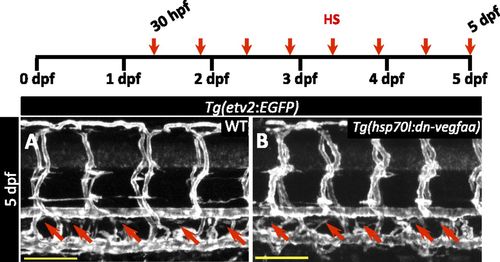
dn-vegfaa does not affect thoracic duct formation. (A and B) Confocal images of the trunk vasculature in 5 dpf Tg(etv2:EGFP) WT (A) and Tg(hsp70l:dn-vegfaa);Tg(etv2:EGFP) (B) larvae heatshocked from 30 hpf every 12 h until 5 dpf. Red arrows point to thoracic duct. (Scale bars: 100 µm.) |
|
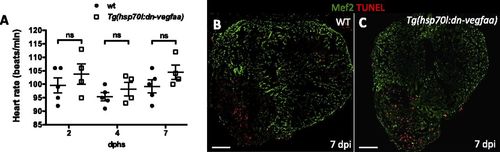
dn-vegfaa does not affect heart rate or cardiomyocyte survival. (A) Doppler measurements of heart rate in WT (AB) (n = 5) and Tg(hsp70l:dn-vegfaa) (n = 4) adult fish heatshocked daily for 7 d. Each data point represents a single fish. Data are expressed as mean ± SEM; dphs, days after heatshock. ns, no significant differences. (B and C) Sections from WT and Tg(hsp70l:dn-vegfaa) adult ventricles. Sections were stained with anti-Mef2 antibody (green) and TUNEL staining (red). (Scale bars: 100 µm.) |