- Title
-
Patterning of proneuronal and inter-proneuronal domains by hairy- and enhancer of split-related genes in zebrafish neuroectoderm
- Authors
- Bae, Y.K., Shimizu, T., and Hibi, M.
- Source
- Full text @ Development
|
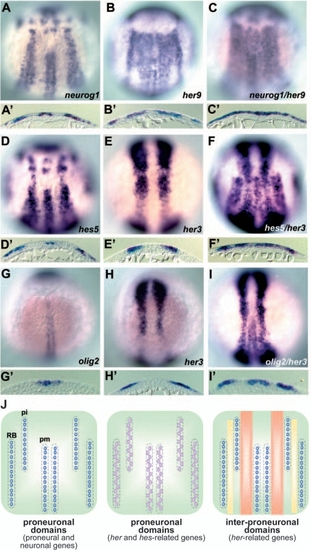
Expression of Her genes in the zebrafish posterior neuroectoderm. Expression of neurogenin1 (neurog1; A,A′), her9 (B,B′), hes5 (D,D′), and her3 (E,E′) at the one-somite stage, and olig2 (G,G′) and her3 (H,H′) at the three-somite stage. Co-staining with probes for neurog1 and her9 (C,C′, one-somite stage), hes5 and her3 (F,F′, one-somite stage), and olig2 and her3 (I,I′, three-somite stage). Dorsal views of the hindbrain and spinal cord region. (J) Schematic representation of expression profiles of the genes expressed in the proneuronal and inter-proneuronal domains. pm, primary motoneuron; pi, primary interneuron; RB, Rohon Beard neurons. neurog1 and olig2 are expressed in the proneuronal domains (indicated by purple circles, left). hes5 and her4 are also expressed in the proneuronal domains, but not in the cells that express proneural genes (indicated by pink circles, middle). her3 is expressed between the primary motoneurons and interneurons (orange stripes), and her9 is expressed in all the inter-proneuronal domains (orange and yellow stripes, right). EXPRESSION / LABELING:
|
|
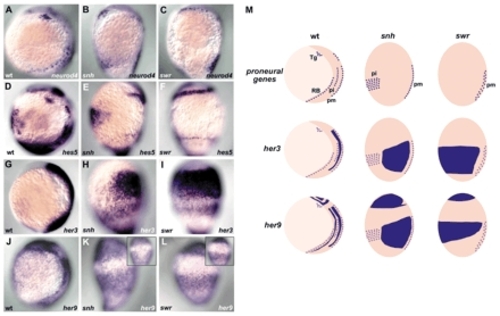
Regulation of her3 and her9 expression by Bmp signaling. Expression of neurod4 (A-C), hes5 (D-F), her3 (G-I) and her9 (J-L) in wild-type (A,D,G,J), snail house/bmp7 (snh; B,E,H,K) and swirl/bmp2b (swr; C,F,I,L) mutant embryos at the one-somite stage. Lateral views with dorsal towards the right and ventral views (insets in K,L). (M) Schematic representation of the expression profiles of the proneuronal gene neurod4, and of her3 and her9 in the wild-type, snh and swr mutant embryos. Tg, trigeminal neurons. In the snhty68a mutant embryos, the RB neurons are lost and the primary interneurons are shifted ventrally (primary neurons are marked by neurod4 expression). In the swrtc300 mutant embryos, both the RB and primary interneurons are absent. In correlation with the expansion of the inter-proneuronal domains in these mutant embryos, the expression domains of her3 and her9 were strongly expanded ventrally. The expression reached the most ventral side in the swrtc300 mutant embryos. EXPRESSION / LABELING:
|
|
her3 and her9 expression is independent of Notch signaling. (A-L) Expression of neurod4 (A,E), her3 (B,F), her9 (C,G) and hes5 (D,G) in wild-type (A-D), mind bombm178 mutant (mib; E-H) and DAPT (γ-secretase inhibitor)-treated embryos (+DAPT; I-L) at the one-somite stage. Dorsal views with anterior towards the top. The expression of neurod4 became homogeneous within the proneuronal domains, and the expression of hes5 was abolished in the mibm178 mutant and the DAPT-treated embryos. The expression of her3 and her9 was not affected in the mibm178 mutant or the DAPT-treated embryos. (M-T) Expression of neurod4, her3, her9 and hes5 in embryos that had received an injection of β-galactosidase RNA (50 pg) and RNA for a constitutively active zebrafish notch5 (50 pg, +zN5ICD; M-P) or an antimorphic Xenopus Delta-1 (100 pg, +xDlstu; Q-T) at the one-somite stage. Co-staining with the riboprobes and X-gal. Dorsal views with anterior towards the top. Misexpression of zN5ICD abolished the neurod4 expression and reduced the her3 expression at variable levels, and strongly induced the ectopic expression of hes5. By contrast, the expression of xDlstu increased the neurod4 expression within the proneuronal domain and abolished the hes5 expression, but did not affect the her3 expression. The expression of her9 was not affected by the expression of either zN5ICD or xDlstu. EXPRESSION / LABELING:
|
|
Expression of her3 and her9 is independent of proneural genes. (A-C) Expression of her3, her9 and hes5 in embryos that had received an injection of RNA for Myc-tagged Neurog1 (Neurog1MT, 50 pg) and ß-galactosidase (50 pg) (the injection was into one blastomere of two- to four-cell stage embryos) at the one-somite stage. Co-staining with the riboprobes and X-gal. (D-L) Expression of her3, her9 and islet1 in embryos that received injections of control MO (2 ng, D-F) or 1 ng neurog1-MO and 2 ng olig2-MO (N1/O2 MO, G-I) at the one-somite stage. Dorsal views with anterior towards the top. Misexpression of Neurog1MT reduced the her3 expression and strongly induced the ectopic expression of hes5, but did not affect the her9 expression. neurog1/olig2 morphant embryos showed the loss of RB and primary motoneurons, which express islet1, but displayed a normal expression of her3 and her9. EXPRESSION / LABELING:
|
|
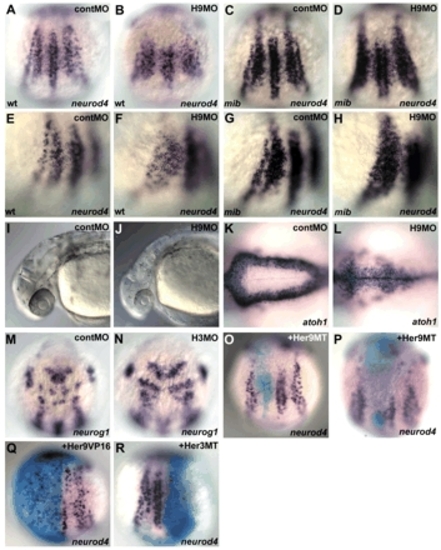
Her3 and Her9 repress the expression of proneural genes. (A-H) Expression of neurod4 in the wild-type control embryos (A,E), embryos that received an injection of 2 ng her9ATG-MO (H9MO; B,F), mib mutant embryos (C,G) and mib mutant embryos that received an injection of her9ATG-MO (D,H) at the one-somite stage. Dorsal views (A-D) and dorsolateral views (E-H). neurod4 was expressed in the inter-proneuronal domain between the RB neurons and primary interneurons in the her9ATG-MO-injected wild-type and mib mutant embryos, where neurod4 was not expressed in the control embryos. (I-L) The her9-MO-injected embryos had a reduced and narrow fourth ventricle at 30 hours post fertilization (hpf) (J,L), compared with the control embryos (I,K). atoh1 marks neuronal precursor cells located in the subventricular zone of the fourth ventricle. Lateral views (I,J) and dorsal views of the hindbrain region (K,L). (M-N) Expression of neurog1 in 1 ng of control MO (M) and her3-MO1-injected embryos (H3MO; N) at the one-somite stage. Dorsal views for the anterior hindbrain. Ectopic expression of neurog1 was detected only in rhombomeres 2 and 4 of the her3 morphant embryos. (O-R) Her9 functions as a transcriptional repressor. RNAs for Myc-tagged Her9 (Her9MT, 5 pg; O,P), Her9-VP16 fusion protein (Her9VP16, 5 pg; Q) or Myc-tagged Her3 (Her3MT, 20 pg; R) together with 50 pg ß-galactosidase RNA, were injected into one blastomere of two- to four-cell stage embryos. The embryos were fixed at the one-cell stage, and stained with X-gal and the neurod4 probe. In the majority of the her9MT RNA-injected embryos, cells that received her9MT RNA were distributed to only the inter-proneuronal domains (O). However, when the cells were located in the proneuronal domains, the neurod4 expression was reduced or abrogated (P). Expression of Her9VP16 led to an ectopic but spotty expression of neurod4 in the neural plate (Q). Expression of Her3MT led reduction of the neurod4 expression in the proneuronal domains (R). Quantification of the results is shown in Table 1. EXPRESSION / LABELING:
|
|
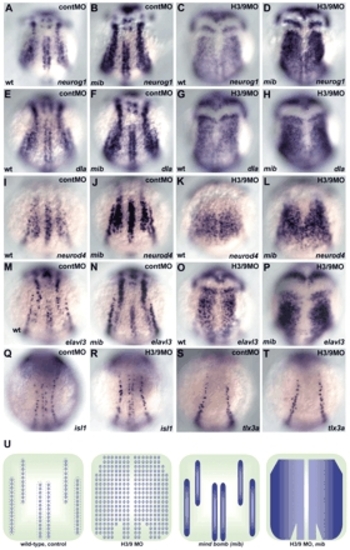
Her3 and Her9 are required for formation of the inter-proneuronal domains. (A-P) Expression of neurog1 (A-D), deltaA (dla; E-H), neurod4 (I-L) and elavl3 (M-P) in wild-type embryos that received 2 ng control MO (A,E,I,M) or 1 ng her3-MO1 and 2 ng her9ATG-MO (C,G,K,O), or in mib mutant embryos that received control MO (B,F,J,N) or her3/her9-MO (D,H,L,P) at the one-somite stage. Dorsal views. (Q-T) Expression of islet1 (isl1, a marker for primary motoneurons and RB neurons) and tlx3a (a marker for RB neurons) in the control (Q,S) and the her3/her9-MO-injected embryos (R,T) at the three-somite stage. Expression of islet1 and tlx3a was not affected in the her3/her9 morphant embryos. (U) Schematic representation of the expression profiles of neurog1, dla, neurod4 and elavl3 in wild-type or mib embryos that received control or her3/her9-MO injections. The expression of these genes became ubiquitous in the neural plate, except for the midline region including the floor plate in the her3/her9 morphant embryos, and became homogenous within the proneuronal domains in the mib mutant embryos. The her3/her9 morphant mib mutant embryos showed ubiquitous and homogeneous expression of these genes in the neural plate, except for the midline region. EXPRESSION / LABELING:
|

Unillustrated author statements EXPRESSION / LABELING:
|