- Title
-
A morpholino-based screen to identify novel genes involved in craniofacial morphogenesis
- Authors
- Melvin, V.S., Feng, W., Hernandez-Lagunas, L., Artinger, K.B., and Williams, T.
- Source
- Full text @ Dev. Dyn.
|
External morphology and craniofacial cartilage organization in wild-type 5-day post-fertilization (dpf) Danio rerio larvae. A: Lateral view of a fixed zebrafish larva. B: Ventral view of larval head skeleton. C: Lateral view of same fish imaged in B. D: Flat-mount of viscerocranial skeleton after removal of the neurocranium (100� magnification). E: Flat-mount of neurocranium (100� magnification). cb, ceratobranchials; ch, ceratohyal; ep, ethmoid plate; hs, hyosymplectic; m, Meckel′s; pc, parachordals; pq, palatoquadrate; and t, trabecula. Bone and pharyngeal teeth (arrowheads) and otic vesicles (arrow) are also shown. Scale bars = 500 μm. |
|
Defects in the viscerocranium of Class I smoc2 morphants. A: Lateral view of 5dpf zebrafish larva injected with 10 ng smoc2 UTR Morpholino; inset more severe smoc2 morphant with greatly reduced head. B: Lateral view of 5dpf zebrafish larva injected with 10 ng smoc2 SPL Morpholino. C: Ventral view of the craniofacial skeleton of an uninjected larva at 5dpf. D: Ventral view of the cranial skeleton of a smoc2 UTR morphant at 5dpf; inset: a lateral view of a severe smoc2 UTR morphant showing lack of discrete craniofacial cartilages. E: Ventral view of the cranial skeleton of a smoc2 SPL morphant. Asterisk denotes flattened or inverted ceratohyal and arrowhead indicates the presence of pharyngeal teeth in some smoc2 morphants. Abbreviations as in Figure 1. PHENOTYPE:
|
|
Defects in cranial development in Class I meis morphants. A: Lateral view of a 5dpf larva injected with 5 ng meis1 ATG Morpholino. B: Ventral view of the craniofacial skeleton of an uninjected larva at 5dpf. C: Ventral view of a meis1 ATG morphant showing a fusion between the Meckel′s and palatoquadrate cartilages and the ceratohyal and first ceratobranchial (asterisk). Ectopic cartilage is indicated by an arrowhead. D: Lateral view of a flat mount showing a fused Meckel′s and palatoquadrate cartilage and the hyosymplectic. E: Ventral view of flat mount showing ectopic cartilage (arrowhead) and fused ceratobranchials (asterisk). F: Ventral view of a larval skeleton 5 days after injection with 20 ng meis1 SPL Morpholino. G: Ventral view of a larval skeleton after co-injection with 20 ng meis1 SPL Morpholino and 2 ng meis1 ATG Morpholino. Ectopic cartilage is indicated by an arrowhead and the fusion between Meckel′s and the palatoquadrate is indicated by the asterisk. H: Lateral view of a 5dpf larva injected with 10 ng meis2a Morpholino. I: Ventral view of the skeleton of a meis2a ATG morphant. J: Flat mount of a meis2a morphant (5 ng ATG MO) indicating a fusion between Meckel′s and palatoquadrate (asterisk) and ectopic cartilage (arrowhead). Abbreviations as in Figure 1. PHENOTYPE:
|
|
Neurocranium defects elicited by knock-down of wu:fb16h09. A?D: In situ hybridization for expression of wu:fb16h09. (A) Lateral view and (B) dorsal view of a 24hpf embryo showing expression of wu:fb16h09 in neural tissues of the head (g; ganglia) and trunk hypochord, middle of the somite, and ventral spinal cord (arrow). (C) Lateral view and (D) ventral view of a 48hpf zebrafish showing expression of wu:fb16h09 in the forebrain (f), trigeminal, and other cranial ganglia (g) and regions flanking the presumptive mouth (s, stomodeum). E: Lateral view of a 5dpf larva after injection with 10 ng wu:fb16h09 Morpholino. Arrowhead indicates collapse of the head rostral to the eye. F: Ventral view of the craniofacial skeleton of an uninjected larva at 5dpf. G: Ventral view of 5dpf skeleton of a wu:fb16h09 SPL morphant showing loss of the ethmoid plate and fusion of the trabeculae. (Note that we do not observe cartilage fusions in the viscerocranium with this Morpholino.) H: Flat mount of the neurocranium of wu:fb16h09 ATG morphant after injection with 10 ng ATG Morpholino. Asterisk indicates reduced/absent ethmoid plate and fused trabeculae. Other abbreviations as in Figure 1. |
|
Analysis of macc1 expression and function in the larval zebrafish head. A: Microarray analysis of Macc1 expression in the embryonic mouse face. Expression increases in the mandibular (MdP), maxillary (MxP), and frontonasal (FNP) prominences between E10.5 to E12.5. The x-axis represents the five time points for each prominence. The y-axis shows the relative expression level on a log2 scale, i.e., every integer represents a doubling in the level of expression from the preceding number. The dashed black line indicates the average expression level for all probes. B: In situ hybridization for macc1 at 48hpf. Lateral view shows macc1 expression in the otic vesicle (ov) and developing mouth (s). B′: Ventral view after removal of the yolk to more clearly show macc1 expression in the roof of the stomodeum (s). C: Lateral view of 5dpf zebrafish after injection of 7.5 ng macc1 SPL1. The head and eyes of macc1 morphants are hypoplastic. D: Ventral view of the craniofacial skeleton of an uninjected larva at 5dpf. E: Ventral view of mild phenotypes associated with injection of 5 ng macc1 SPL1 Morpholino. E′: Ventral view of severe phenotypes associated with injection of 10 ng SPL1 macc1 Morpholino. F: Ventral view of mild phenotypes associated with injection of 10 ng macc1 SPL2 Morpholino. F′: Ventral view of severe phenotypes associated with injection of 20 ng SPL2 macc1 Morpholino. Defects in the ceratobranchials (asterisk), the ceratohyals (arrowhead), and Meckel′s and palatoquadrate cartilages resulting in a recessed lower jaw (arrow) are indicated. Other abbreviations as in Figure 1. |
|
Progressive and dose-dependent loss of the neural crest?derived cranial skeleton in Class III morphants. A, D?E′: vgll4l morphants. F?H′: tshz2 morphants. I?K′: pkdcc morphants. A, F, I, Lateral view of 5dpf zebrafish after injection with gene-specific Morpholino. B and C, Ventral view of uninjected larva at 5dpf. D, E, G, H, J, and K, Ventral view of mild morphant phenotypes at 5dpf. D′, E′, G′, H′, J′, and K′, Ventral view of severe morphant phenotypes. Bottom: Magnified images of craniofacial skeletons. Dose and Morpholino are indicated in the figure. The arrowheads in G′, J′, and K′ indicate the remnants of the viscerocranial cartilages. Asterisk indicates fused trabecula and reduction of the ethmoid plate. Scale bars = 500 μm in A, B, and C. Abbreviations as in Figure 1. PHENOTYPE:
|
|
Absence of p53 alleviates the cartilage phenotypes present in wu:fb16h09 and pkdcc morphants. A: Flat-mounted neurocranium of wu:fb16h09 morphant in wildtype zebrafish after injection of 10 ng ATG Morpholino. B: Flat-mounted neurocranium of wu:fb16h09 morphant in p53 null zebrafish after injection of 10 ng ATG Morpholino. (Note that the wu:fb16h09 ATG Morpholino only generates neurocranial defects and so we were not able to assess the influence of p53 on viscerocranial defects in this experiment.) C: Flat-mounted cranial cartilages of pkdcc morphant in wildtype zebrafish after injection of 20 ng ATG Morpholino. D: Flat-mounted cranial cartilages of pkdcc morphant in p53 null zebrafish after injection of 20 ng ATG Morpholino. Abbreviations as in Figure 1. PHENOTYPE:
|
|
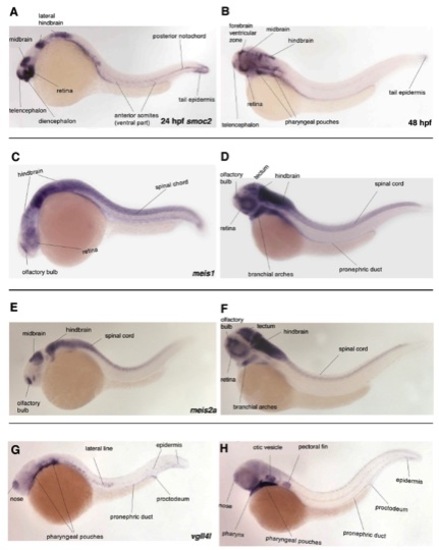
Expression of smoc2, meis1, meis2a, and vgll4l at 24 and 48hrs of zebrafish development. All images are lateral views, anterior to the left. A, C, E, G are 24 hpf and B, D, F, H are 48 hpf. A, B) At 24 hpf, smoc2 is expressed in specific brain regions as well as tail mesoderm, including somites and notochord. At 48 hpf, smoc2 expression resolves to the brain and pharyngeal pouches. C, D) meis1 is expressed in the hindbrain and spinal cord as well as the olfactory bulb and retina at 24hpf. Expression is also observed in the retina and branchial arches at 48hpf. E, F) meis2a is expressed in the midbrain, hindbrain and spinal cord as well as the olfactory bulb at 24 hpf. By 48 hpf, expression expands to include the branchial arches. G, H) At 24-48 hpf, vgll4l is expressed in the nose, pharyngeal pouches, lateral line, epidermis and pronephric duct. (All images taken from ZFIN with permission from Thisse et al., 2001 - Expression of the zebrafish genome during embryogenesis (NIH R01 RR15402). ZFIN Direct Data Submission). EXPRESSION / LABELING:
|
|
Cartilage rotation and fusion defects in meis1 and meis2a morphants. A, Ventral view of viscerocranial cartilages of a 5dpf uninjected larva. A′, Magnified view of A showing position of palatoquadrate (arrowhead). B, Ventral view of viscerocranial cartilages of a 5dpf meis1 ATG morphant larva. B′, Magnified view of B showing cartilage fusions in morphants as well as an apparent rotation of the palatoquadrate cartilage (arrowhead). C, Ventral view of viscerocranial cartilages of a 5pf morphant larva co-injected with both meis1 ATG and meis1 SPL MOs. C′, Magnified view of C showing cartilage fusions in morphants and the apparent rotation of the palatoquadrate cartilage (arrowhead). D, Ventral view of viscerocranial cartilages of a 5dpf meis2a ATG morphant larva. D′, Magnified view of D showing cartilage fusions (asterisks) in morphants. E, Ventral view of viscerocranial cartilages that were dissected and flat-mounted from a 5dpf meis2a ATG morphant larva showing cartilage fusions (asterisks). Arrow indicates where a cartilage fusion was broken during dissection. PHENOTYPE:
|
|
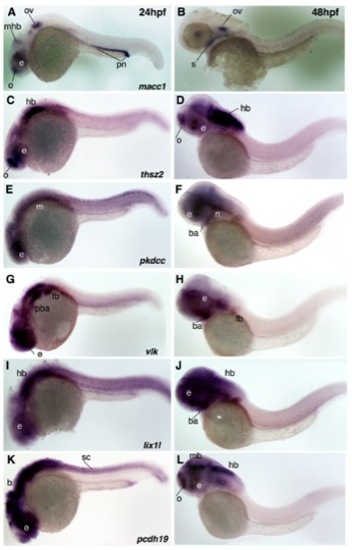
Expression of macc1, thsz2, pkdcc (LOC565254), vlk, lix1l, and pcdh19 at 24 and 48hrs of zebrafish development. All images are lateral views, anterior to the left. A, C, E, G, I and K are 24 hpf and B, D, F, H, J and L are 48hpf. A, B) macc1 expression at 24hpf is observed in the olfactory bulb, eye, midbrainhindbrain boundary, otic vesicle, and pronephros. At 48hpf, macc1 expression continues in the otic vesicle and becomes apparent in the roof of the stomodeum. C, D) thsz2 is expressed in the olfactory bulb, eye and hindbrain at both 24 and 48hpf. Weak expression also occurs in the branchial arches. E, F) pkdcc (LOC565254) is expressed diffusely in the eye and anterior and posterior mesoderm at 24hpf and resolves to the eye, branchial arches, and the region of the forming neurocranium at 48hpf. G, H) vlk expression at 24hpf is observed in the eye, posterior branchial arches and fin bud. By 48hpf, expression expands to the more anterior branchial arches. I, J) Expression of lix1l is observed in the eye and hindbrain at 24 and 48hpf as well as more diffusely in the branchial arch region at 48hpf. K, L) pcdh19 is expressed in all CNS structures at 24hpf and resolves to the olfactory bulb, eye, midbrain, and hindbrain at 48hpf. Abbreviations: b, brain; ba, branchial arches; e, eye; fb, fin bud; hb, hindbrain; m, mesoderm; mb, midbrain; mhb midbrain-hindbrain boundary; n, neurocranium; o, olfactory bulb; ov, otic vesicle; pba, posterior branchial arches; pn, pronephros; s, stomodeum; sc, spinal cord. |
|
Skeletal phenotypes obtained from other potential Class I-III morphants. A, ventral view of 5dpf skeleton of a lix1l ATG morphant showing inversion of the ceratohyal and loss of ceratobranchials. A′, ventrolateral view of the same larva in (A). B, ventral view of a 5dpf skeleton of a pcdh19 ATG morphant showing reduction of the ethmoid plate. B′, flat mount of the neurocranium of pcdh19 ATG morphant showing reduction of ethmoid plate and fusion of trabeculae. C and C′, ventral view of a 5dpf skeleton of a vlk SPL morphant showing mild (C) and severe (C′) loss of neural crest-derived cartilages. PHENOTYPE:
|
|
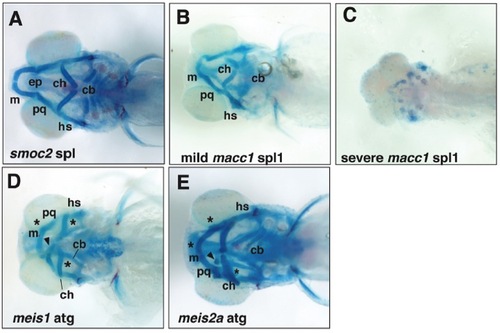
Examples of morphant phenotypes maintained in the absence of p53. All images are of the morphant phenotypes seen in p53 null larvae. A, Ventral view of viscerocranial cartilages of a 5dpf smoc2 SPL morphant showing inversion of the ceratohyal, and a reduction in ceratobranchial size and number. Figure 2 shows similar morphant phenotype obtained in wild-type larva. B, C, Compare with macc1 morphant phenotypes obtained in wild-type larva shown in Figure 5. B, Ventral view of viscerocranial cartilages of a 5dpf macc1 spl1 morphant larva presenting with a mild phenotype - loss of ceratobranchials, inversion of the angle between the paired ceratohyal and reduction of Meckel?s cartilage. C, Ventral view of viscerocranial cartilages of a 5dpf macc1 SPL1 morphant presenting with a severe phenotype ? almost complete aplasia of all head cartilages. D, E, Compare with meis1 and meis2a morphant phenotypes obtained in wild-type larva shown in Figure 3. D, Ventral view of viscerocranial cartilages of a 5dpf meis1 ATG morphant larva showing cartilage fusions between Meckel?s and palatoquadrate cartilages and ceratohyal and ceratobranchial cartilages. E, Ventral view of viscerocranial cartilages of 5dpf meis2a ATG morphant larva showing cartilage fusions between paired Meckel?s cartilages, Meckel?s and palatoquadrate, and ceratohyal and ceratobranchial cartilages. Both meis morphants also show ectopic cartilages (arrowhead) and cartilage fusions (*). Abbreviations: cb, ceratobranchials; ch, ceratohyal; ep, ethmoid plate; hs, hyosymplectic; m, Meckel?s; and pq, palatoquadrate. |
|
Analysis of Adap1 expression and function in the larval zebrafish. |