- Title
-
Tailbud-derived Bmp4 drives proliferation and inhibits maturation of zebrafish chordamesoderm
- Authors
- Esterberg, R., Delalande, J.M., and Fritz, A.
- Source
- Full text @ Development
|
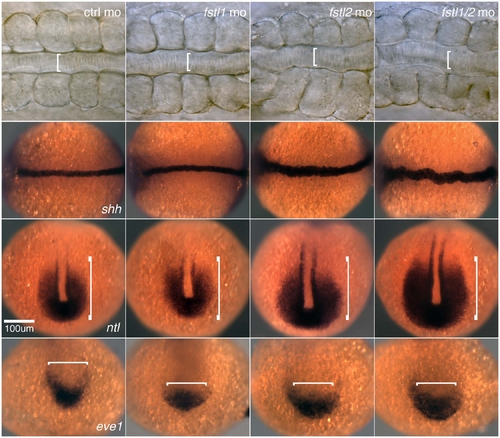
Expansion of the tailbud in Fstl1/2 morphant embryos. (A-T) Expression of the tailbud markers spt (A-D), bmp4 (E-H), PSMAD1/5/8 (I-L) and eve1 (M-P), and the posterior axial marker admp (Q-T) in six-somite embryos injected with control morpholino (A,E,I,M,Q), fstl1/2 morpholino (B,F,J,N,R), fstl2 mRNA (C,G,K,O,S), and fstl1/2 morpholino and fstl2 mRNA (D,H,L,P,T). (U) Levels of PSMAD1/5/8, but not PSMAD2, are increased in Fstl1/2 morphants beginning at late gastrulation. All views except E-L are dorsal views, with anterior towards the top. (E-L) Lateral views, with anterior towards the left. Brackets illustrate the expression domain observed in control embryos. Arrows indicate the tailbud. Insets in E-H are views of the tailbud. EXPRESSION / LABELING:
|
|
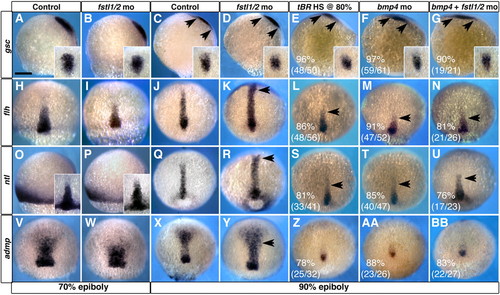
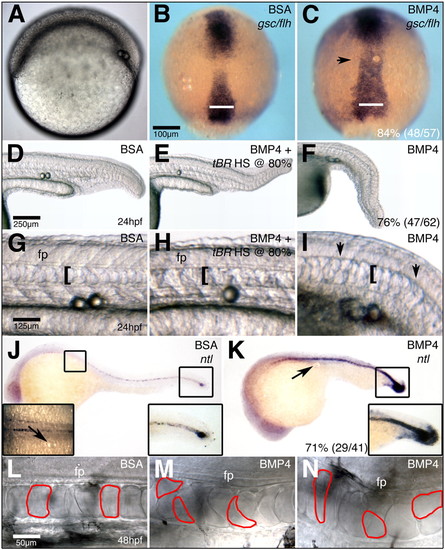
Bmp4 is required in the establishment of chordamesoderm during late gastrulation. (A,B,H,I,O,P,V,W) Morpholino knockdown of Fstl1 and Fstl2 has no effect on the development of axial structures prior to 70% epiboly. (C-F,J-M,Q-T,X-AA) At 90% epiboly, alteration of BMP signaling reveals a requirement for BMP activity in patterning of chordamesoderm. (G,N,U,BB) The chordamesoderm phenotype observed when Fstl1/2 and Bmp4 are knocked down in concert resembles that of Bmp4 morphants. Arrows in C-G and K-N,R-U,Y indicate the extent of prechordal plate and chordamesoderm domains, respectively. gsc (C-G) expression in prechordal plate, and flh (J-N), ntl (Q-U) and admp (X-BB) expression in chordamesoderm. (A-G,O,P) Views are lateral, with ventral towards the left. Insets in A-G are dorsal views of the prechordal plate domain. Insets in O,P are dorsal views of chordamesodermal domain. All other views are dorsal views, with anterior towards the top. Scale bar in A: 100 μm in all panels. EXPRESSION / LABELING:
PHENOTYPE:
|
|
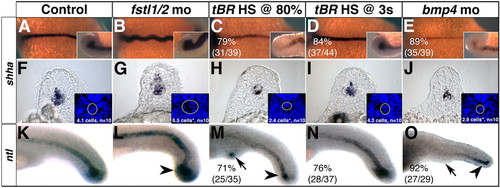
Sizes of the notochord and chordoneural hinge (CNH) are influenced by Bmp4 activity during late gastrulation. (A-E) shha expression in 14-somite stage embryos. Insets are lateral views of the CNH. (F-J) shha expression in sections through somites 5-10 of 18-somite stage embryos. Insets are cropped images of the same section stained with DAPI. The mean number of DAPI-stained cells from 10 experimental embryos was subjected to one-way ANOVA, followed by a two-tailed, equal variance t-test. (G,H,J) Asterisks indicate significance (P<0.001). (K-O) ntl expression in 24 hpf embryos. CNH domains are indicated with arrowheads, arrows indicate to ectopic tail formation. (A-E) Dorsal views, with anterior towards the left. (K-O) Lateral views, with anterior towards the left. EXPRESSION / LABELING:
PHENOTYPE:
|
|
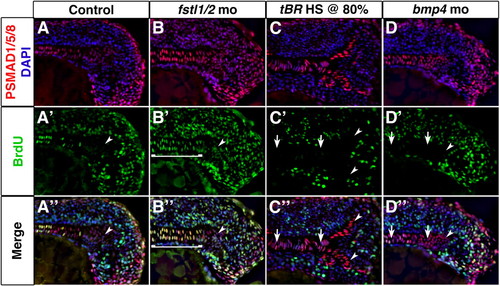
BMP activity establishes proliferation in axial mesoderm. (A-D″) Longitudinal sections through the tailbud of 14-somite embryos exposed to PSMAD1/5/8 (red) and BrdU (green) antibodies reveal that axial cells undergoing proliferation are responding to BMP activity. Cell proliferation is increased in axial tissue of Fstl1/2 morphants (B-B″), and reduced in tBR embryos heat-shocked at 80% epiboly (C-C″) and Bmp4 morphants (D-D″). Arrowheads indicate the CNH. Anterior is towards the left in all views. Brackets and arrows indicate the expanse and absence of proliferation in chordamesoderm, respectively. EXPRESSION / LABELING:
PHENOTYPE:
|
|
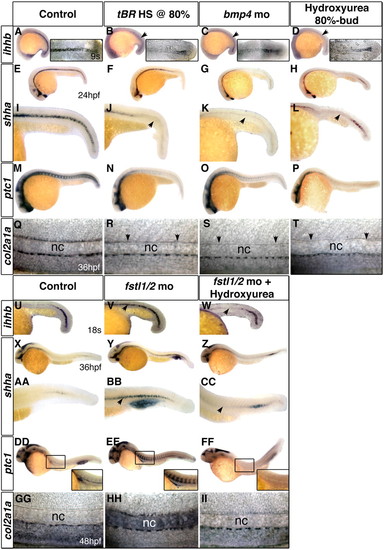
The temporal state of the notochord changes with the alteration of BMP activity. The expression of chordamesoderm markers in embryos with reduced BMP activity is strongly reduced, resembling older embryos. Fstl1/2 morphant embryos maintain expression of these genes well past equivalently staged controls, resembling the expression patterns observed in younger embryos. (A-D,U-W) ihhb expression in 9-somite (A-D) or 18-somite stage (U-W) embryos. (E-L,X-CC) shha expression in 24 hpf (E-L) and 36 hpf (X-CC) embryos. (I-L,AA-CC) High-magnification view of the trunk. (M-P,DD-FF) ptc1 expression in 24 hpf (M-P) and 36 hpf (DD-FF) embryos. (Q-T,GG-II) col2a1a expression in 36 hpf (Q-T) and 48 hpf (GG-II) embryos; arrows in R-T indicate absence of floorplate expression; arrows elsewhere indicate the shift in chordamesodermal gene expression relative to controls. nc, notochord. All views are lateral, with anterior towards the left. Insets in A-D are flat-mount dorsal views. (Q-T,GG-II) Lateral views of the trunk. Insets in DD-FF indicate higher magnification views of the boxed areas. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Disruption of the temporal state of the notochord can be observed through notochord cell morphology. (A,C,D) 24 hpf control embryos; notochord cells differentiate in an anterior to posterior manner. High magnification view of notochord cells at the level of the hind yolk extension (C) and at the level of the cloaca (D). (B,E,F) 24 hpf Fstl1/2 morphants. High magnification view of notochord cells at the level of the hind yolk extension (E) and at the level of the cloaca (F). (G-L) 24 hpf tBR embryos heat-shocked at 80% epiboly (G,H) and Bmp4 morphants (I,J). (K-N) Application of cell cycle inhibitors in wild-type (K,L) and Fstl1/2 morphants (M,N). (O) Cell transplantation procedure. See text for details. (P,Q) Wild-type donor cells (green) populating the notochord of tBR embryos do not enlarge and vacuolate prematurely (Q). (R,S) tBR donor cells (green) consistently fail to populate the notochord of wild-type hosts; instead, they contribute to tissues in paraxial mesoderm. All views except R are lateral views of 24 hpf embryos with anterior towards the left. (R) Dorsal view with anterior towards the left. Red outlines indicate notochord cell morphology. PHENOTYPE:
|
|
A requirement for Bmp4 on the dorsal side of the gastrula. (A) Bead implantation on the dorsal side of the gastrula. (B,C) gsc and flh expression in 90% epiboly embryos that have had BSA- (B) or Bmp4- (C) soaked beads implanted into axial tissue. (D,F) The notochord of 24 hpf embryos implanted with BSA- (D) or Bmp4- (F) soaked beads. (E) tBR embryos implanted with Bmp4-soaked beads and heat-shocked at 80% epiboly. (G-I) High magnification images of the notochord in D-F. (J,K) ntl expression in 27 hpf embryos implanted with BSA- (J) or Bmp4- (K) soaked beads. (L-N) High-magnification images of the notochord at the level of the hind yolk extension of 48 hpf embryos that were implanted with BSA- (L) or Bmp4- (M,N) soaked beads. Floor plate is out of the focal plane in N. (B,C) Dorsal views, with anterior towards the top. Bars in B,C indicate the width of chordamesodermal domain at its widest point in the control embryo in B. All other views are lateral views, with anterior towards the left. Arrows in C,J,K indicate the position of the implanted bead. Brackets and arrows in G-I indicate the width of the notochord and absence of floorplate, respectively. Red outlines in L-N indicate notochord cell morphology. |
|
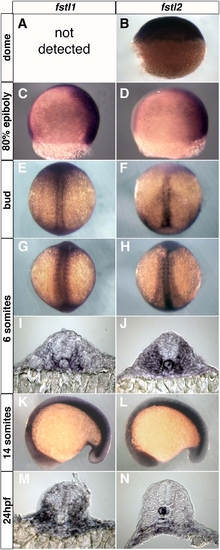
Expression of follistatin-like 1 and follistatin-like 2. (A) Expression of these genes was not present. (B) Lateral view, animal pole at the top. (C,D) Lateral views, ventral to the left. (E-H) Dorsal views, with anterior to the top. (I,J) Transverse sections through the trunk of six-somite embryos. (K,L) Lateral views, with anterior towards the left. (M,N) Transverse sections of 24 hpf embryos at the level of somites 10-15. |
|
Phenotype of translation-blocking fstl1/2 morpholinos. The size of the notochord and tailbud are increased in embryos injected with a single fstl morpholino, and to a greater extent when fstl1/2 morpholinos are injected in combination. Brackets illustrate the domain observed in control embryos. |
|
BMP attenuation in tBR embryos heat-shocked during late gastrulation. Wild-type and tBR embryos were heat-shocked during late gastrulation and fixed at 30-minute intervals. Antibodies against PSMAD1/5/8 (red) labels cells responding to BMP activity at the times and backgrounds indicated. |
|
Ventral margin extirpation. (A) Model of the procedure. Approximately 100 cells were removed from the ventral margin (red) of 80% epiboly embryos. All images shown are of embryos with their ventral margins removed. (B-D) flh (B), ntl (C) and admp (D) expression in embryos at 90% epiboly. (E-E″) Antibodies against PSMAD1/5/8 (red) and BrdU (green) in longitudinal sections of the tail of 14-somite stage embryos. (F) High-magnification view of notochord cells of 24 hpf embryos at the level of the cloaca. (G,H) shha and ptc1 expression in 24 hpf. |
|
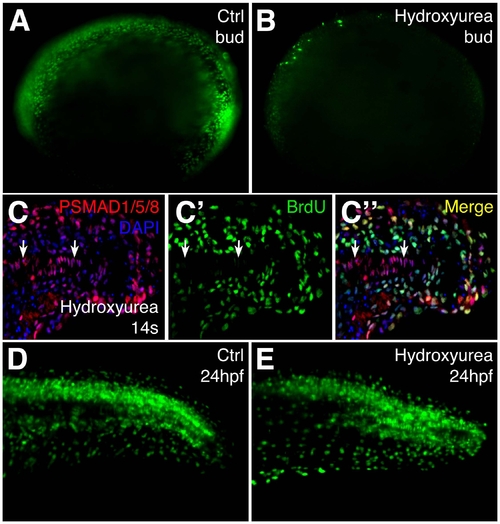
BrdU incorporation of embryos treated with the cell cycle inhibitors hydroxyurea and aphidicolin. Embryos were treated from 80% epiboly to bud stage, then inhibitors were removed by washing. (A,B) BrdU incorporation of DMSO- (A) and hydroxyurea/aphidicolin-treated (B) bud stage embryos. (C-C″) Antibodies against PSMAD1/5/8 (red) and BrdU (green) in longitudinal sections of the tail of 14-somite hydroxyurea/aphidicolin treated embryos. (D,E) BrdU incorporation of DMSO- (D) and hydroxyurea/aphidicolin-treated (E) 24 hpf embryos. |
|
Bmp2/7 do not affect notochord morphology. (A,B) gsc and flh expression in 90% epiboly embryos that have had BSA- (A) or Bmp2/7- (B) soaked beads implanted into axial tissue. (C,D) The notochord of 24 hpf embryos implanted with BSA- (C) or Bmp2/7- (D) soaked beads. Beads are located anterior to the region shown in D. (E,F) ntl expression in 27 hpf embryos implanted with BSA- (E) or Bmp2/7- (F) soaked beads. (G,H) High-magnification images of the notochord at the level of the hind yolk extension of 48 hpf embryos that were implanted with BSA- (G) or Bmp2/7- (H) soaked beads. fp, floorplate (A,B) Dorsal views, with anterior towards the top. Bars in A,B indicate the width of chordamesodermal domain at its widest point in the control embryo in A. All other views are lateral views, with anterior towards the left. |
|
Myotome patterning as a proxy of notochord maturity. (A-C) myod1 expression in 9-somite stage embryos is increased in adaxial and medial fast muscle cells of Fstl1/2 morphants (B), and is reduced in the medial fast muscle domain of tBR embryos heat-shocked at 80% epiboly (C). (D-F) Immunostaining against Myod1 and F-actin reveals an increase in Myod1-positive cells of the medial fast muscle domain of Fstl1/2 morphants (E), and a decrease in Myod1-positive cells in tBR embryos (F). Images are of the anterior-most three somites. The mean number of Myod1-stained cells from five experimental embryos was subjected to one-way ANOVA, followed by a two-tailed, equal variance t-test. Asterisks indicate significance (P<0.001). (G-I) Immunostaining with the F59 antibody, which recognizes mainly slow muscle, reveals that the horizontal myoseptum is increased in 24 hpf Fstl1/2 morphant embryos (H). Muscle fibers are disorganized in tBR embryos (I). (J-L) Immunostaining with antibodies against 4D9, which labels Engrailed-positive cells (red), and Prox1, which labels slow muscle cells (green), reveal an increase in medial fast muscle (red) and slow muscle pioneers (yellow) in 24 hpf Fstl1/2 morphants (K), and a reduction of Engrailed positive cells in tBR embryos (L). (M-O) Sections through the trunk level of 24 hpf embryos confirm the results in J-L. Antibodies against PSMAD1/5/8 (red) and Engrailed (green) reveal an increase in PSMAD1/5/8-positive chondrocytes as well as medial fast muscle fibers and slow muscle pioneers (green) in Fstl1/2 morphants (N). Chondrocytes do not form in tBR embryos, and the domain of medial fast muscle fibers and slow muscle precursors is reduced (O). |