- Title
-
Vascular Endothelial Growth Factor A and Leptin Expression Associated with Ectopic Proliferation and Retinal Dysplasia in Zebrafish Optic Pathway Tumors
- Authors
- Schultz, L.E., Solin, S.L., Wierson, W.A., Lovan, J.M., Syrkin-Nikolau, J., Lincow, D.E., Severin, A.J., Sakaguchi, D.S., McGrail, M.
- Source
- Full text @ Zebrafish
|
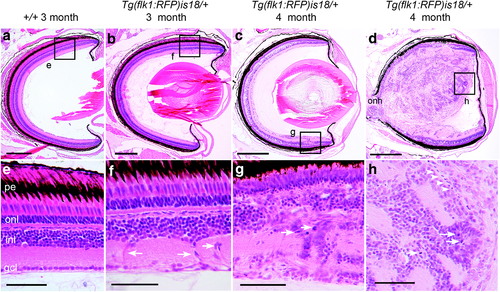
Detection of aberrant cell migration and ectopic proliferation in young adult Tg(flk1:RFP)is18 dysplastic retina. (a, e) Histological staining of retina section from 3-month-old adult wild-type zebrafish showing nuclear layers and organized laminar structure. (b, f) Representative image of section through retina of 3-month-old Tg(flk1:RFP)is18 adult reveals aberrant migration of cells across the inner nuclear layer (arrows, f) (n = 9). (c–h) Representative images of retinal sections from 4-month-old Tg(flk1:RFP)is18 adults (n = 15). (c, g) Retina from 4-month-old Tg(flk1:RFP)is18 adult with dysplasia reveals disruption of inner, outer, and ganglion cell layers with numerous mitotic figures (arrows, g). (d, h) Four-month-old Tg(flk1:RFP)is18 adult with advanced retinal tumor filling the vitreous space. Tumor tissue is composed of fibrous material interspersed with numerous mitotic figures, cells showing heterogeneous nuclear morphology and forming occasional rosettes (arrows, h), and blood vessels (arrowheads, h). pe, pigmented epithelium; onl, outer nuclear layer; inl, inner nuclear layer; gcl, ganglion cell layer. Scale bars (a, b) 200 μm; (c, d) 500 μm; (e–h) 50 μm. |
|
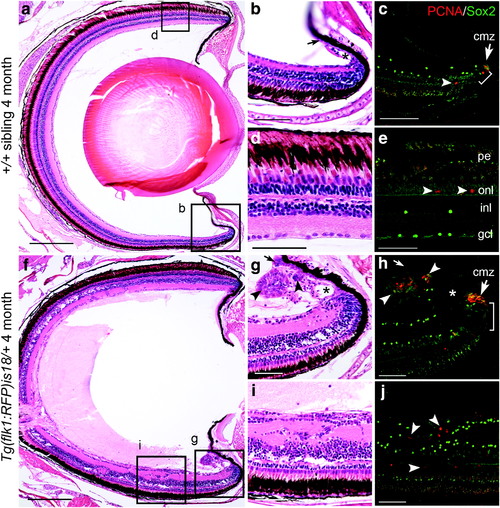
Ectopic proliferation and Sox2 expressing cells in young adult Tg(flk1:RFP)is18 dysplastic retina. (a–e) Wild-type 4-month-old adult (n = 3) shows that PCNA positive cells are restricted to stem/progenitor cells at the ciliary marginal zone (cmz, arrow, c) and rod precursor cells in the photoreceptor outer nuclear layer (arrowheads, e). Sox2-expressing cells overlap with PCNA positive stem cells at the ciliary marginal zone (bracket, c) and are present in amacrine/displaced amacrine cells in the inner nuclear and ganglion cell layers (c, e). (f–j) Retina from 4-month-old Tg(flk1:RFP)is18 adult (n = 3) shows numerous regions of proliferation distributed throughout the neural retina (f). The ciliary marginal zone (g, h) and ventral ciliary circumferential artery (asterisk, g) are expanded. Small masses of PCNA and Sox2 positive cells (arrowheads, g, h) are present in the normally single-cell layered nonpigmented epithelium extending from the ciliary marginal zone (small arrow, g, h–compare to small arrow in wild type b). Ectopic regions of proliferation in the neural retina contain disorganized cells expressing PCNA and Sox2 (arrowheads, j). cmz, ciliary marginal zone; gcl, ganglion cell layer; inl, inner nuclear layer; onl, outer nuclear layer; pe, pigmented epithelium. Scale bars (a, f) 200 μm; all other panels 50 μm. |
|
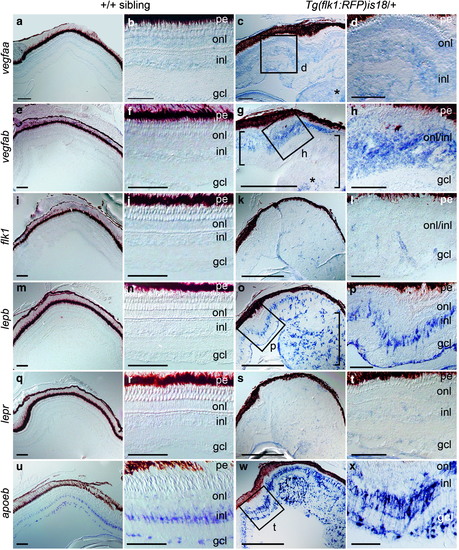
Induction of vegfab and lepb expression in distinct cell populations in Tg(flk1:RFP)is18 dysplastic retina and retinal tumor. In situ hybridization of retina cryosections from +/+ (a, b, e, f, i, j, m, n, q, r, u, v) and Tg(flk1:RFP)is18 (c, d, g, h, k, l, o, p, s, t, w, x) adult siblings. Weak expression of vegfaa is detected in the three nuclear layers of the retina (a, b) and in early dysplastic (c [box], d) and retinal tumor tissue (c, asterisk). Expression of vegfab (e, f), the VEGF receptor flk1 (i, j), lepb (m, n), or the Leptin receptor lepr (q, r) is not detected in wild-type +/+ retina. (g, h) In early dysplastic retina vegfab is detected throughout the inner and outer nuclear layers (g [small bracket], h). In the tumor containing region (large bracket) vegfab is detected in a central region of the lesion (asterisk). (k, l) The VEGF receptor flk1 was present in small groups of cells scattered throughout the tissue that may overlap with microvessels. (o, p) lepb is highly expressed in a subset of cells in the inner nuclear layer and cells in the ganglion cell layer in dysplastic retina and present in many cells evenly distributed in the early tumor (bracket). (s, t) The lepr receptor expression is faint and diffuse in dysplastic retina and tumor tissue. (u, v) In wild-type retina the glial marker apoeb is strongly expressed in inner nuclear layer Müller glia and astrocytes in the ganglion cell layer/nerve fiber layer. (w, x) apoeb expression is highly elevated in dysplastic retina and is detected in projections crossing the inner plexiform layer. Numerous cells throughout the tumor tissue labeled intensely with apoeb. gcl, ganglion cell layer; inl, inner nuclear layer; onl, outer nuclear layer; pe, pigmented epithelium. All scale bars, 100 μm, except panels (g, k, o, s, t) scale bars, 500 μm. |
|
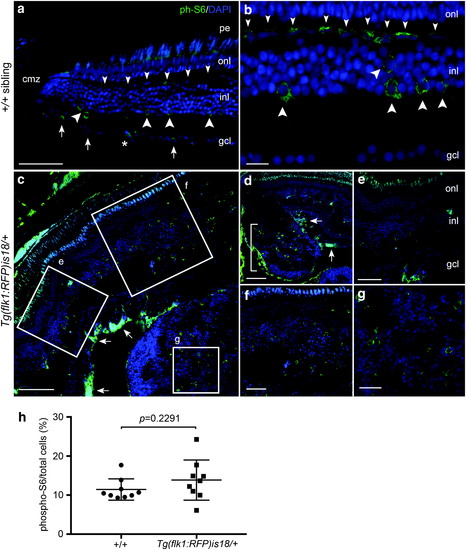
Activated mTOR signaling marker phosphorylated-S6 in adult zebrafish wild-type retina, Tg(flk1:RFP)is18/+ dysplastic retina, and retinal tumor. (a, b) phospho-S6 (green) is detected in putative horizontal cells (small arrowheads), amacrine and/or Müller glia (large arrowheads), and retinal ganglion cells and/or displaced amacrine cells (small arrows) in the mature region of the retina. Nuclei are labeled with 4′,6-diamidino-2-phenylindole. Asterisk marks a blood vessel. (c–g) In Tg(flk1:RFP)is18/+ dysplastic retina phospho-S6 labeling is present in a subset of cells throughout regions of the retina with increased cell number and disorganized retinal layers (c–f). Intense labeling of phospho-S6 was present in the ganglion cell and nerve fiber layers (c, d, arrows). Large lesions contained phospho-S6 positive cells distributed throughout the tumor mass (d [bracket], g). (h) Percentage of phospho-S6 cells/total cells in wild-type retina and Tg(flk1:RFP)is18/+ tumor. +/+ 11.4% ± 0.9 vs. Tg(flk1:RFP)is18/+ 13.9% ± 1.7, p = 0.2291. n = 9 sections, three sections each from three individuals of each genotype. gcl, ganglion cell layer; inl, inner nuclear layer; onl, outer nuclear layer; pe, pigmented epithelium. Scale bars, (a, g, f) 50 μm; (b, c) 20 μm; (d, e) 100 μm; (h) 25 μm. |