- Title
-
Neuronal Ndrg4 Is Essential for Nodes of Ranvier Organization in Zebrafish
- Authors
- Fontenas, L., De Santis, F., Di Donato, V., Degerny, C., Chambraud, B., Del Bene, F., Tawk, M.
- Source
- Full text @ PLoS Genet.
|
Characterization of the ndrg4 mutant. (A) Schematic representation of the ndrg4 genomic locus. The extended region on the exon 2 represents the sequence targeted by the CRISPR/Cas9 system. Red: sgRNA binding site. Blue: PAM sequence. ndrg4+ corresponds to the wild-type allele; ndrg4 *31 and ndrg4 *34 are the loss-of-function alleles used in this study. (B) Schematic of ndrg4 protein product. In ndrg4 *31 and ndrg4 *34 mutant fish, the deletions result in a frameshift generating a premature STOP codon at the level of the amino acids 31 and 34 (of 352) in the ndr family domain. Lateral views of a control (C) and a ndrg4 mutant (D) embryos at 72 hpf. The arrows point to the heart, note the pronounced heart edema (white asterisk) observed in the ndrg4 mutant. Lateral view of ndrg4 mRNA expression in a control (E) and a ndrg4-/- embryo (F) at 48hpf. Note the absence of ndrg4 expression in the mutant. Scale bar = 200 μm. EXPRESSION / LABELING:
PHENOTYPE:
|
|
ndrg4 is required for sodium channel and neurofascin clustering in the peripheral nervous system. (A-C) Lateral view of mbp RNA expression in control (A), ndrg4 mutant (B) and morphant (C) embryos at 4 dpf. Arrows indicate mbp-expressing cells along the PLLn. Scale bars = 200μm. (D-L) Acetylated tubulin (ac tub; red) and sodium channels (NaCh; green) immunohistochemistry of a (D-F) control, ndrg4 mutant (G-I) and ndrg4 morphant (J-L) PLLn at 4 dpf. Scale bars = 5μm. (M) High magnification of three nodes of Ranvier (arrowheads) from a control nerve. Scale bar = 100nm. (N) A significant decrease in the number of sodium channels clusters is observed in ndrg4 mutants and morphants in comparison to controls (p<0.001). Acetylated tubulin (ac tub; red) and FIGQY (green) immunohistochemistry of a (D’-F’) control, ndrg4 mutant (G’-I’) and ndrg4 morphant (J’-L’) PLLn at 4dpf. Scale bars = 5 μm. (O,P) A significant decrease in the number of FIGQY labeled clusters is observed in ndrg4 mutants and morphants in comparison to controls (p<0.001). |
|
ndrg4 is not required for axonal outgrowth or early Schwann cell development. Acetylated tubulin expression in control (A), ndrg4 mutant (C) and morphant (B) embryos at 4 dpf showing the PLLn nerve. Scale bar = 45μm. (d-F) Whole mount in situ hybridization of a (d) control embryo, ndrg4 mutant (F) and ndrg4 morphant (E) showing sox10 expression in PLLn SCs (arrows) at 3 dpf. Scale bar = 200μm. Lateral view of a control foxd3::GFP embryo (G), a ndrg4 morphant (H) at 3 dpf showing SCs (arrows) along the PLLn. Transmission electron micrographs showing cross-section through (I) control and ndrg4 mutant (J). Control PLLn shows an average of 10.7 myelinated axons (blue asterisks). (J) An average of 5.36 myelinated axons (blue asterisks) is observed in the ndrg4 mutant’s PLLn. (S: Schwann cell). Scale bars = 0.5μm. (K,L) Quantification of the total number of axons and the number myelinated axons in controls, ndrg4 mutants and ndrg4 morphants. NS: Non Significant. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Chimeric embryos show evidence of ndrg4 requirement in neurons for sodium channel clustering. (A) ndrg4MO mCherry labeled PLL neurons shown by arrows. (b,e) ndrg4MO mCherry labeled axons of the PLLn in two different chimeric embryos; (c,f) sodium channels along the PLLn of ndrg4MO mCherry labeled (arrow) and of control (arrowheads) axons; (d,g) merge of the two labelings. Sodium channel clustering is absent in ndrg4MO axons (mCherry labeled) while control ones in the same PLLn show normal clustering. (H) Control mCherry labeled neurons indicated by arrows. The dashed line indicates the margin of the PLLg. (I) Control mCherry labeled axons (arrows). (J) Sodium channel clusters along the PLLn in control labeled (arrows) and other non-labeled (arrowheads) axons. (K) Merge of the two labelings. For (a, H) scale bars = 5μm. For (b-G; I-K) scale bars = 10μm. (L-N) WT mCherry labeled axons in ndrg4 morphant embryos are shown in (L, arrow) and the corresponding sodium channels in (M, arrows). Note the clustering of the nodes in the WT labeled axons while the other ndrg4 deficient axons show no sign of sodium channel clustering. (N) Merge of the two labelings. Scale bar = 7 μm. (O-Q) WT mcherry labeled axons in ndrg4-/- are shown in (O) and the corresponding sodium channels in (P, arrows). (Q) Merge of the two labelings showing clustered sodium channels along the WT axons (arrows). Scale bar = 5 μm. |
|
ndrg4 function is required for the expression of several genes that are essential for vesicle docking. (A) qPCR showing a significant decrease in the expression of nsfa, syt1a, stxbp1b but not vamp2 in ndrg4 mutants with mRNA relative expression to ef1a. Controls from 3 different experiments have deliberately been set to 100 per cent. (n = 3 independent experiments of 30 embryos each). (B-D) Lateral views showing Snap25 expression in the PLLn axons at 4 dpf. Snap25 is visible all along the PLLn (arrows) in controls (B), but detected to a lesser extent in the ndrg4 mutants (C) and morphants (D) PLLn. (E-G) Lateral views of SV2 immunostaining, labeling synaptic vesicles at 4 dpf. (E) Synaptic vesicles are regularly distributed in the control PLLn (arrows) whereas they form agglomerates (arrowheads) in ndrg4 mutants (F) and morphants (G). Scale bars = 10μm. (H-K) Western blots showing a sharp decrease in Snap25 protein expression in ndrg4 mutants and morphants. Snap25 expression is significantly decreased in ndrg4 morphants (H,J) and mutants (I,K) with protein relative expression to β-actin. Controls from 3 different experiments have deliberately been set to 100 per cent. (n = 3 independent experiments of 30 embryos each). For the mutants study, the average of three groups of controls has been set to 100 per cent. EXPRESSION / LABELING:
PHENOTYPE:
|
|
snap25 can regulate the clustering of sodium channels and neurofascin along the PLLn and increase the clustering of sodium channels in ndrg4 mutants. (A-C) lateral views of control (A), snap25 morphant (B) and snap25 MO+snap25b mRNA (C) embryos at 72hpf. (D,F,H) lateral views of acetylated tubulin staining showing the PLLn in control (D), snap25 morphant (F) and snap25 MO+snap25b mRNA (H) embryos at 4dpf. (E-I”) lateral views of sodium channels and acetylated tubulin staining along the PLLn of control (E-E”), snap25 morphant (G-G”) and snap25MO+ snap25b mRNA (I-I”) embryos at 4dpf. (J,K) Quantification of the sodium channel and neurofascin clustering, data are represented as mean±sem. Scale bars = 200 μm, 60 μm and 5 μm in (C), (F) and (I,I”) respectively. Transmission electron micrographs showing cross-section through (L) control and (M) snap25 morphant’s PLLn at 4 dpf. (L) Control PLLn shows an average of 10.7 myelinated axons (blue asterisks). (M) An average of 9.13 myelinated axons (blue asterisks) is also observed in the snap25 morphant embryo’s PLLn. (S: Schwann cell). Scale bars = 0.5μm. (N) Quantification of the number of nodes seen in the PLLn in controls, ndrg4 morphants and ndrg4 morphants injected with 150pg of snap25b mRNA. (O) Quantification of the number of nodes seen in the PLLn in controls, ndrg4 mutants and ndrg4 mutants injected with 150pg of snap25b mRNA. PHENOTYPE:
|
|
Tetanus toxin injection does not impair sodium channel clustering and myelination in the PNS. (A) Lateral view of a control foxd3::GFP embryo at 3 dpf. Arrows indicate SCs along the PLLn. (A') Acetylated tubulin expression in the same control embryo at 3 dpf. Arrows show the PLLn axons. (A'') Merge of (A) and (A’). (C) Lateral view of a TeNT injected foxd3::GFP embryo at 3 dpf. Arrows indicate SCs along the PLLn. (C') Acetylated tubulin expression in the same TeNT injected embryo at 3 dpf. Arrows indicate the PLLn axons. (C'') Merge of (C) and (C'). Scale bars = 50μm. Sodium channel labeling in control (B) and TeNT injected embryo (D) and their corresponding axons of the PLLn, (B') and (D') respectively. (B'') merge of (B) and (B'), (D'') merge of (D) and (D’). Scale bars = 10μm. (E,F) Transmission electron micrographs showing cross-section through (E) control and (F) TeNT injected embryos’ PLLn at 4 dpf. (E) Control PLLn shows an average of 6 myelinated axons (blue asterisks). (F) An average of 6 myelinated axons (blue asterisks) is also observed in the TeNT injected embryo’s PLLn. (S: Schwann cell). Scale bars = 0.5μm. (J) Quantification of the number of nodes seen in the PLLn shows no significant difference between controls and TeNT injected embryos. |
|
ndrg4 morphant and mutant phenotype. (A-C) Overall morphology of a control embryo (A), ndrg4 morphant embryo (B), ndrg4 MO+ndrg4 mRNA co-injected embryo (C), at 48hpf. (B) ndrg4 morphant embryo displays smaller head and eyes and is slightly thinner. (C) Co-injection of ndrg4 MO and mRNA rescue this phenotype. Scale bars = 500μm. (D-F) Acetylated tubulin and sodium channels staining of a ndrg4 MO+mRNA co-injected embryo showing clustered sodium channels along the PLLn (34.2 ±3.2; n = 12 embryos) similar to controls. Scale bar = 5μm. (G) Sodium channels staining in sox10::mRFP transgenic line in controls and ndrg4 morphants. Arrowheads indicate the clustering of sodium channels while arrows point to nodal gaps. Note the absence of sodium channels clustering at the nodes in ndrg4 morphants. Scale bar = 5 μm. (H-J) Snap25 immunostaining in HuC::GFP larvae at 3dpf. (H) Control embryos show Snap25 expression in PLLg neurons (arrows). (I) A reduced Snap25 expression was observed within the PLLg in ndrg4 morphants. (J) Rescue of Snap25 expression in the PLLg in ndrg4MO+mRNA co-injected embryos. Scale bars = 5μm. (K) Snap25 expression in the PLLg of control and ndrg4-/- embryos. Note the significant decrease in the expression of Snap25 within the PLLg of ndrg4-/- in comparison to controls. EXPRESSION / LABELING:
|
|
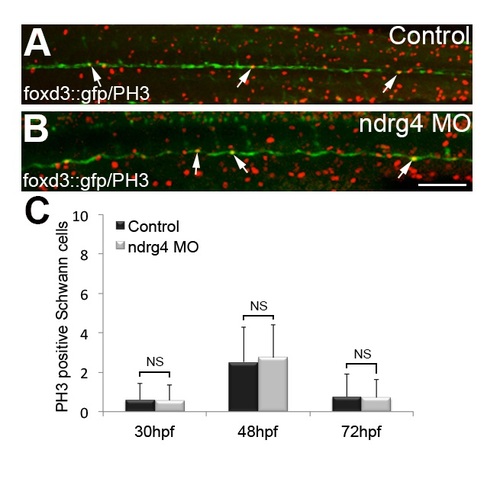
ndrg4 is not required for SC proliferation. PH3 immunohistochemistry in (A) control and (B) ndrg4 morphant foxd3::GFP embryos at 48 hpf. Arrows indicate dividing PLLn SCs. Scale bar = 100μm. (C) Quantification of PH3 positive SCs in controls and ndrg4 morphants shows no significant differences between the two groups at 30 hpf, 48 hpf and 72 hpf. PHENOTYPE:
|
|
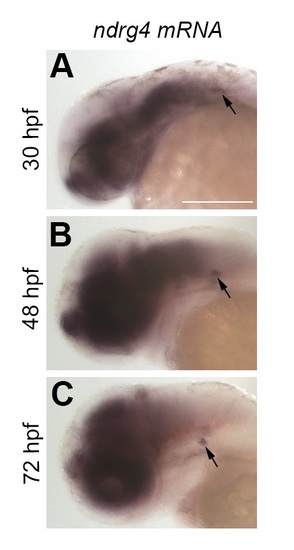
ndrg4 mRNA is expressed in the PLL ganglion and not in SCs. (A-C) In situ hybridization showing ndrg4 mRNA expression in the brain, eye and in the PLL ganglion (arrow) at 30 hpf (A), 48 hpf (B) and 72 hpf (C). Scale bar = 200μm. EXPRESSION / LABELING:
|
|
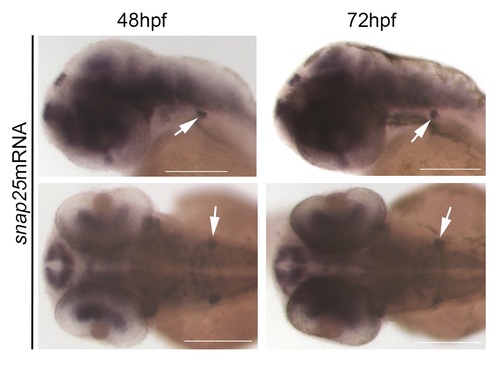
Snap25 mRNA is expressed in the PLL ganglion. In situ hybridization showing snap25b mRNA expression in the brain, eye and in the PLL ganglion (arrow) at 48 hpf and 72 hpf. Scale bar = 200μm. EXPRESSION / LABELING:
|

Unillustrated author statements PHENOTYPE:
|