- Title
-
MYCN Transgenic Zebrafish Model with the Characterization of Acute Myeloid Leukemia and Altered Hematopoiesis
- Authors
- Shen, L.J., Chen, F.Y., Zhang, Y., Cao, L.F., Kuang, Y., Zhong, M., Wang, T., and Zhong, H.
- Source
- Full text @ PLoS One
|
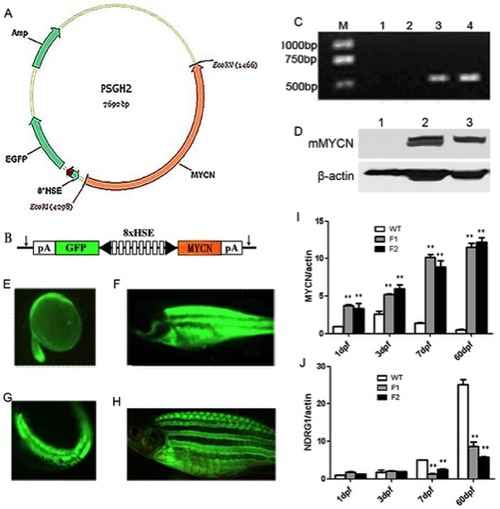
Generation of Tg(MYCN:HSE:EGFP) zebrafish line. (A) Schematic diagram of the structure of PSGH2/MYCN recombinant plasmid. A mouse-MYCN fragment was cloned from HA-MYCN plasmid and inserted into the EcoRI and EcoRV sites of the PSGH2 vector. (B) A schematic presentation of the heat shock element (HSE) promoter. The artificial promoter contains eight multimerized heat shock elements flanked by two minimal promoters in opposed orientation (black arrowhead). EGFP and MYCN are expressed from the bidirectional promoter. The vector is flanked by I-SceI meganuclease sites (arrows). pA, SV40 polyadenylation signal. (C) Transgenic verification by qRT-PCR: M: TAKARA DL2000 marker; lane 1: Blank control (double distilled water); lane 2 and 3: WT and Tg F1 generation embryos at 3 dpf, respectively; lane 4: Positive control (plasmid). (D) Transgenic verification by westernblot: lane 1: WT embryo at 3 dpf; lane 2 and 3: Tg F1 and F2 generation embryos at 3 dpf, respectively. (E–F) EGFP (+) F0 mosaic zebrafish at 24 hours (×50) and 60 days post microinjection (×7.5). (G–H) EGFP (+) F1 Tg zebrafish at 24 hpf (×50) and 60 dpf (×10). (I) Expression of total MYCN (murine exogenous and zebrafish endogenous expression), which was increased gradually in Tg F1, F2 generation fish comparing with that in WT. (J) Expression of NDRG1, which is negative controlled by MYCN in human, was keeping a low lever in Tg F1 and F2 generation. **, P<0.01 EXPRESSION / LABELING:
|
|
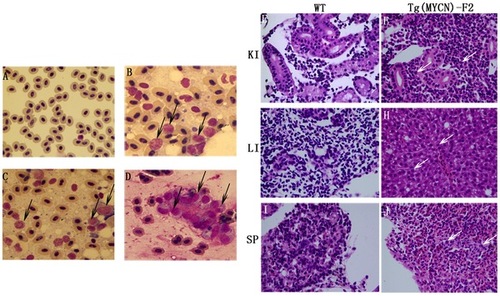
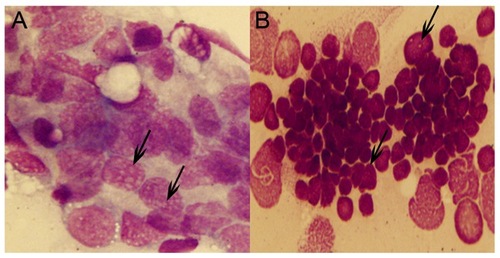
Cytological analysis of Tg(MYCN:HSE:EGFP) zebrafish.Cytology of hematopoietic cells from WT (A) and Tg(MYCN:HSE:EGFP) F0, F1, F2 generation (B, C, D) zebrafish at 60 dpf. The blood cells from WT fish were predominantly erythrocytes, with myeloid cells only occasionally observed. By contrast, erythrocytes were significantly inhibited in Tg fish, enriched for abundant blast-like cells, which are larger than the erythrocytes and have high nuclear to cytoplasmic ratios, containing multiple large nucleoli (black arrow). These blasts were similar to that of human AML peripheral blood. Transverse sections of kidney, liver, and spleen of WT (E, G and I) and Tg(MYCN:HSE:EGFP) F2 generation (F, H and J) zebrafish. Using HE staining, it showed that massive immature hematopoietic cells infiltrated in these organs of Tg fish (white arrow). KI, kidney; LI, liver; SP, spleen. (×1,000) |
|
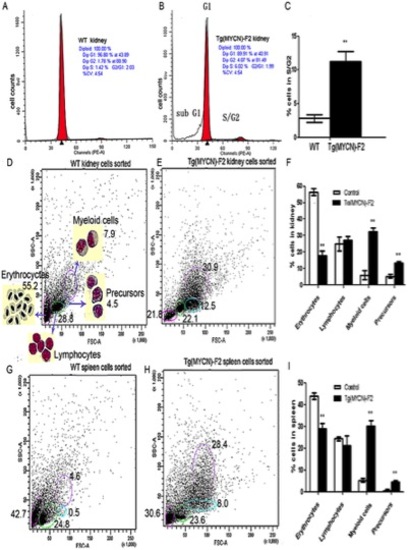
MYCN promoted cell cycle progression and increased the ratios of myeloid cells and its precursors. Single cell suspension from kidney of WT (A) and Tg(MYCN:HSE:EGFP) F2 generation (B) zebrafish was extracted at 60 dpf, treated with red blood cell lysis solution and stained with Propidium Iodide. Bars indicate the postion of modal DNA content peaks corresponding to the indicated G1 or S/G2 cell populations. It showed that MYCN increased the S/G2 ratio, histogram analysis in (C). In addition, elevated apoptosis was also detected in Tg samples as indicated by sub G1 peak (B). FACS analysed blood cells from kidney and spleen of WT (D, G) and Tg (E, H) fish at 60 dpf. Gated populations are as follows: erythrocytes, lymphocytes, myeloid cells, and blood cell precursors (morphologic feature was shown in D). All the detections repeated 3 times and analyzed in the two histograms (F, I) both indicated that myeloid cells and precursors were increased in Tg fish, correspondingly erythrocytes dramatically decreased, meeting the results of peripheral blood smear. **, P<0.01 |
|
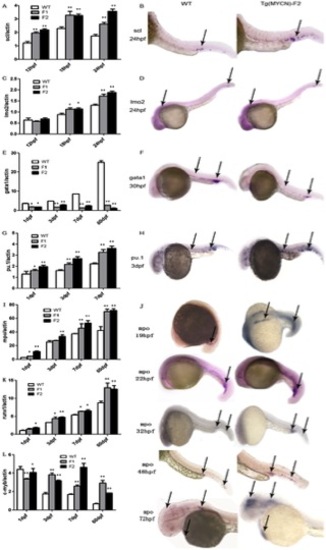
MYCN reprogrammed hematopoiesis by regulating lineage-specific hematopoietic transcription factors. Scl (A) and lmo2 (C) expressed in WT, and Tg(MYCN:HSE:EGFP) F1, F2 generation embryos at 12 hpf, 18 hpf and 24 hpf. In situ hybridization of scl (B) and lmo2 (D) at 24 hpf. MYCN increased the two factors expression. Gata1 (E), pu.1 (G) and mpo (I) were detected by RQ-PCR in WT, Tg F1 and F2 embryos at 1 dpf, 3 dpf, 7 dpf and 60 dpf. In situ hybridization of gata1 (F), pu.1 (H) and mpo (J) in WT and Tg F2 embryos indicated that gata1 was downregulated, however, pu.1 and mpo were increased in Tg group, meaning that myelopoiesis was promoted while erythropoiesis was inhibited. Runx1 (K) and c-myb (L) expression were detected in WT and Tg F1, F2 generation zebrafish at 1 dpf, 3 dpf, 7 dpf and 60 dpf. Runx1, involved in definite hematopoiesis regulation, was upregulated in Tg group. C-myb, predominantly expressed in immature hematopoietic cells, were higher expression in Tg group, which suggests that MYCN overexpression results in accumulation of immature hematopoietic cells in adult fish. *, P<0.05; **, P<0.01 EXPRESSION / LABELING:
PHENOTYPE:
|
|
The examination of transgenic zebrafish persistency. (A) The peripheral blood cells in Tg(MYCN:HSE:EGFP) F1 generation showed no difference in between 2-month and 8-month pf in the amount of cells containing multiple nucleoli. (B)The peripheral blood cells in Tg fish at 13 months pf, after losing the EGFP expression, showed clusters of stripped nucleus. |