- Title
-
chokh/rx3 specifies the retinal pigment epithelium fate independently of eye morphogenesis
- Authors
- Rojas-Munoz, A., Dahm, R., and Nüsslein-Volhard, C.
- Source
- Full text @ Dev. Biol.
|
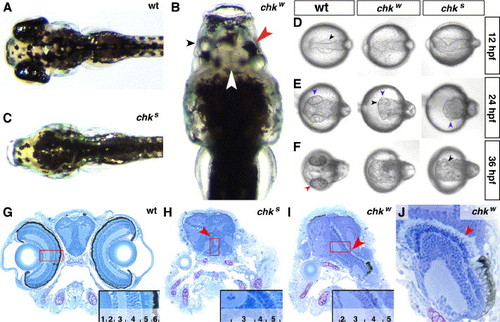
Mutations in the chokh locus affect the development of the RPE. Dorsal view of wt (A), chkw (B) and chks (C) larvae at 5 dpf. (B) In chkw, the RPE is reduced (red arrowhead), the NRs are fused at the midline (white arrowhead) and the lenses are present but misaligned (black arrowhead). Cross-sections through the head of wt (G), chks (H) and chkw (I, J) larvae at 5 dpf. At this stage, the eye is laminated in the wt (inset in panel G). The NRs are fused in both chk alleles (red arrowheads in panels H and I) but undergo lamination and differentiation even in the complete absence of the RPE. (J) The PRCL (red arrowhead) maintains a similar morphology with or without associated RPE. (D–F) Dorsal views of wt, chkw and chks under DIC optics at 12 hpf, 24 hpf and 36 hpf. The evagination of the optic lobes (black arrowheads in panels D–F) and the differentiation of the lens (blue arrowheads in panel E) are always observed in both alleles. In contrast, a pigmented RPE is observed only in wt embryos (red arrowhead in panel F). 1, lens; 2, ganglion cell layer (GCL); 3, inner plexiform layer (IPL); 4, inner nuclear layer (INL); 5, photoreceptor cell layer (PRCL); 6, retinal pigment epithelium (RPE); wt, wild-type; NR, neural retina. |
|
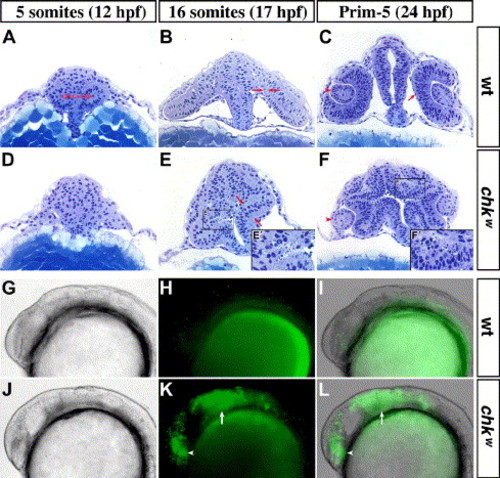
Morphogenetic movements in the eyes are affected and cell death is increased in the optic lobes of chk. Cross-sections of the head of wild-type (A–C) and chkw (D–F) embryos at the 5-somite stage (A, D), 16-somite stage (B, E) and Prim-5 (C, F). At the 5-somite stage, the evagination of the optic lobes is visible in wt embryos (red arrows in panel A) but not in chk (D). At the 16-somite stage, the flattening of the medial layer (delimited by the red arrows in panels B and E) has started in wt embryos (B). Instead, an increase in cell death is observed in the optic lobes of chk (E′). In wt, the RPE is completely flattened and starts differentiating around 24 hpf (arrow in C). In contrast, the lenses are smaller in chk at the Prim-5 stage (arrowheads in C and F), the prospective RPE is not pigmented (F), and the cells in the outer most layer of the optic cup in chk die (F&prime). Similarly, chk embryos (J–L) stained with acridine orange exhibit increased cell death in the eyes (arrowheads) and in the tectum (arrows) at the 20-somite stage, when compared to wt (G–I). In spite of the abnormal morphogenetic movements, extraocular mesenchyme is associated to the optic cups of chk (black arrows in panels B and F). The images are lateral views with the anterior oriented to the left. (I, L) Overlay of the DIC images (G, J) and the fluorescence images (H, K). Prim-5, primordium stage 5. Other abbreviations like in Fig. 1. |
|
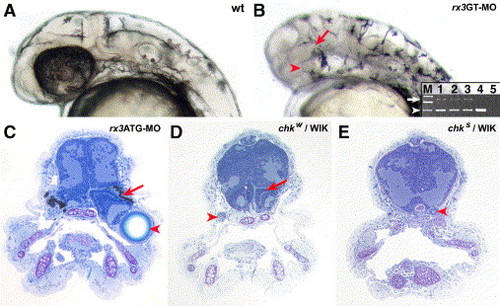
Phenocopy of the chk phenotype by morpholinos directed against rx3. The phenotype upon injection of the morpholinos rx3GT-MO (B) or rx3ATG-MO (C) into wt embryos phenocopies the chk phenotype, compared to buffer-injected controls (A). (A, B) 40 hpf embryos, (C) 9 dpf larvae. The highest effective doses were 200 μM for both rx3ATG-MO and rx3GT-MO. At these concentrations, the RPE is reduced or absent in the morpholino-injected embryos, but the NRs and the lenses (red arrows and arrowheads in panels E and G, respectively) are present. (B) A splicing defect in the mRNA of rx3 (white arrow) is observed upon rx3GT-MO injection as scored by RT-PCR. The aberrantly spliced product is 906 bp long, and the properly spliced mRNA (white arrowhead) is 514 bp long. M, molecular weight markers; 1, 2 and 3, RT-PCR products from a single embryo at 24 hpf, 30 hpf and 40 hpf respectively; 4, injected control at 40 hpf; 5, control without reverse transcriptase. The products in line 3 were obtained from the embryo in the photo. (D, E) chk phenotypes in the WIK background. The red arrows and arrowheads indicate the NR and the lenses, respectively. ATG-MO, morpholino designed against the translation initiation region; GT-MO, morpholino against one of the donor splice sites. See Fig. 1 for other abbreviations. |
|
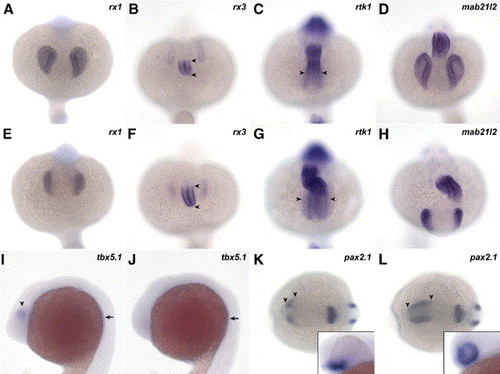
The patterning of the eye is affected at the level of the RPE. Markers for the different axis of the eye were scored at 20 somites (A–J), 10 somites (K, L) or 30 hpf (insets in panels K, L) in wt (A–D, I, K) and chkw embryos (E–H, J, L). At the 20-somite stage, the expression domain of rx1 (A, E), rx3 (B, F) and mab21l2 (D, H) is reduced in the NR of chk. In contrast, the expression in the diencephalon of rtk1 is expanded into the NR compared to wt (arrowheads in panels G and C, respectively). Similarly, the expression pattern of rx3 is expanded in the hypothalamus (arrowheads in B and F). The ventral marker pax2.1 (K, L) is expanded into dorsal and posterior regions of the eye (arrowheads in K and L) at 10 somites. At 30 hpf, the expression domain of pax2.1 includes the entire NR in chk, while in wt it is restricted to the optic stalk region (insets in panels K, L). The expression of tbx5.1 is observed in the dorsal region of the eye (arrowhead in I), but it is lost in mutant embryos (J) at 20 somites. However, tbx5.1 is still present in the rudiments of the pectoral fins (arrows in I and J). The images in the figure are dorsal view, anterior downward (A–H); lateral view, anterior to the left (I, J); dorsal view, anterior to the left (K, L); and lateral view of a first plane of the eye (insets in panels K and L). Abbreviations like in Fig. 1. EXPRESSION / LABELING:
|
|
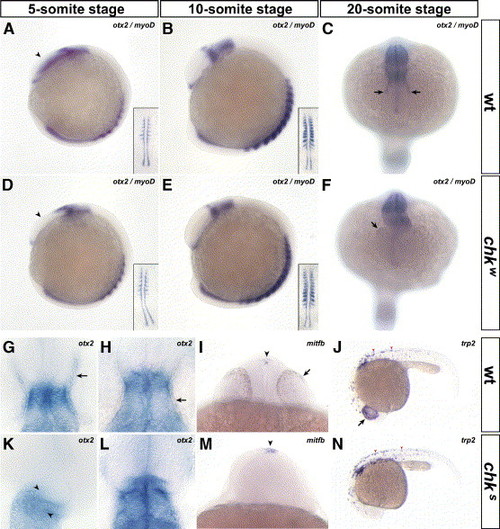
The specification and differentiation of the RPE do not occur in chk. In situ hybridizations with probes for otx2 (A–H, K, L), mitfb (I, M) and trp2 (J, N) performed in mutant (D–F, K–N) and wt embryos (A–C, G–J) indicate that the RPE is specifically affected in chk. At the 5-somite stage, the expression pattern of otx2 is prematurely downregulated in the eye field of chk compared to wt (arrowheads in panels D and A, respectively). This downregulation resembles the expression pattern of oxt2 in wt embryos at the 10-somite stage (panel B compared to panels D and E). At the 20-somite stage, otx2 is expressed in the RPE of wt embryos (arrows in panel C), weakly expressed in chkw (F) and absent in chks (K). The myogenic marker myoD was included to stage the embryos (insets in panels A, B, D and E). Higher magnification images of flat-mounted embryos at the 20-somite stage (G, K) and at 30 hpf (H, L). They show a distortion of the brain at the level of the tectum (arrowheads in K) and the lack of otx2 expression or pigmentation in the RPE of chk (K, L), compared to wt (arrows in panels G, H). The markers for RPE differentiation mitfb (I, M) and trp2 (J, N) are not expressed in the prospective RPE of chk (M, N) compared to wt (arrows in panels I and J) at 24 hpf. However, their expression pattern is maintained in the epiphysis (arrowheads in panels I, M) and in the body melanophores (arrowheads in panels J and N). The orientation of the embryos is: lateral view, anterior to the left (A, B, D, E, J and N); dorsal view, anterior downward (C, F); dorsal view, anterior upward (G, H, K and L); and ventral view (I, M) Abbreviations like in Fig. 1. EXPRESSION / LABELING:
|
|
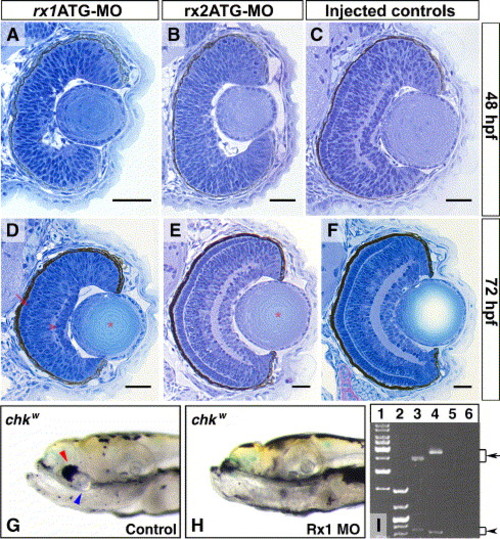
The rx3 paralogs, rx1 and rx2, are not involved in the development of the RPE. (A–F) Cross-sections, dorsal upwards, through the eyes of wt embryos injected with rx1-MOs (A, D), rx2-MOs (B, E) or injection buffer only (C, F). When compared to the controls (C, F), the morphant phenotypes do not affect the proportion of the NR surrounded by RPE or the pigmentation in the RPE cells. However, other specific phenotypes are observed. At 48 hpf, the NRs of the morpholino-injected embryos are not laminated (A, B), and the eyes of rx1-morphants (A) are smaller than those of control embryos (C). At 72 hpf, the eyes of rx1 morphants (D) are still smaller than the control embryos (F), but an incipient PRCL and GCL are visible (arrow and arrowhead in D, respectively). Conversely, the only evident phenotype at 72 hpf in rx2 morphants is a subtle delay in the maturation of the lenses, also present in rx1 morphants (asterisk in panels D and E). (G, H) Lateral images of 9 dpf double-mutant embryos for rx3 and rx1, obtained by injecting rx1ATG-MO into the chkw background. The lenses and the small patches of NR-associated RPE observed in chkw (blue and red arrowheads, respectively in panel G) are absent in the double mutants. Splicing defects in the mRNA of rx2 (arrow in line 3) and rx1 (arrow in line 4) are observed upon rx2GT-MO or rx1GT-MO injections as scored by RT-PCR (I). 1, 1 kbp DNA markers; 2, low molecular weight DNA markers; 5–6, controls without reverse transcriptase for rx2 and rx1, respectively. Scale bars: 20 μm. Refer to Fig. 1 for abbreviations. |
|
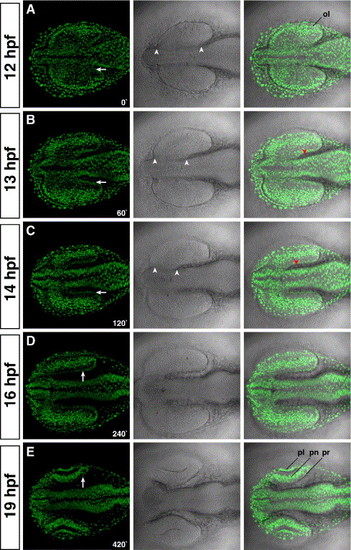
The prospective RPE is formed by the coordinated migration of the midbrain. (A) At 12 hpf, the optic lobes are fully formed and attached along their A/P axis to the diencephalon (arrowheads in the middle panels). (B) At 13 hpf, the furrow that separates the optic lobes from the diencephalon is formed by the massive migration of the midbrain towards the forebrain region. (C) The furrow is almost fully formed at 14 hpf. The size of the furrow is similar between panels C with D. Therefore, from 14 hpf onwards, the velocity of the midbrain migration decreases drastically. (E) At 19 hpf, the optic cups are formed, and the lens start invaginating from the ectoderm. At all times examined, extraocular mesenchyme is closely associated to the prospective RPE (red arrowheads in panels B and C). The time is given in minutes. Left panels, H2A:H2A-EGFP; middle panels, DIC; right panels, overlay. Anterior is to the left. ol; optic lobe; pl, prospective lens; pn; prospective neural retina; pr, prospective retinal pigment epithelium. |
|
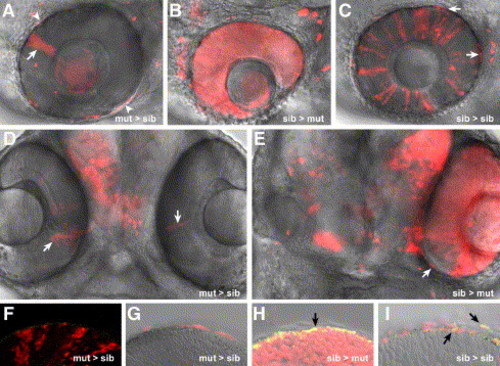
chk/rx3 specifies the prospective RPE. Single confocal images of mosaic eyes generated by transplantation of rhodamine-labeled cells at the late blastula stage. chks mutant cells transplanted into wt embryos form small clones of NR (arrows in panels A and D) but are completely excluded from the RPE. Some cells are false positives as concluded by the analysis of the complete Z stack (asterisks in panel A). Importantly, even donor cells in close proximity to the eye can be precisely considered as excluded from the RPE (arrowheads in panel A). Conversely, wt cells transplanted into chks mutant embryos form NR and RPE, while the host cells are always excluded from the RPE (B, E). Notably, even in a fully rescued eye with mosaic NR, the RPE is of wt origin (arrow in E). Donor cells randomly contribute to the RPE in control transplanted embryos (arrows in panel C). Specific staining of the RPE with anti-RPE65 (green in panels F–I). Overlapping signal is observed in the RPE only in sib > mut (H) or sib > sib (I) mosaics (black arrows). The images correspond to 70 hpf larvae. The orientation of the chimeras is dorsal up, anterior to the left (A, B, C) or ventral view, anterior upward (D, E). |
Reprinted from Developmental Biology, 288(2), Rojas-Munoz, A., Dahm, R., and Nüsslein-Volhard, C., chokh/rx3 specifies the retinal pigment epithelium fate independently of eye morphogenesis, 348-362, Copyright (2005) with permission from Elsevier. Full text @ Dev. Biol.