- Title
-
Mutations in zebrafish genes affecting the formation of the boundary between midbrain and hindbrain
- Authors
- Brand, M., Heisenberg, C.P., Jiang, Y.J., Beuchle, D., Lun, K., Furutani-Seiki, M., Granato, M., Haffter, P., Hammerschmidt, M., Kane, D.A., Kelsh, R.N., Mullins, M.C., Odenthal, J., van Eeden, F.J., and Nüsslein-Volhard, C.
- Source
- Full text @ Development
|
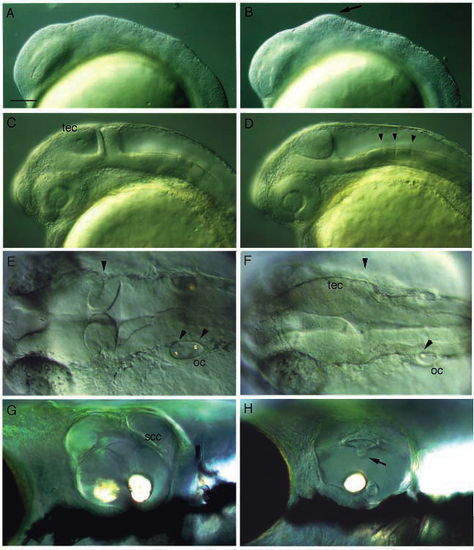
Lateral views of living noi and ace mutant embryos. (A) Wild-type embryo at 36 hours of development. (B) Sibling of the embryo in A that is homozygous mutant for a strong allele of noi (noith44). Absence of the tectum, cerebellum and MHB is indicated by an asterisk. (C) Embryo homozygous mutant for a weak allele of noi (noity31a). A partially formed tectum, but no cerebellum or MHB, is observed. (D) Embryo homozygous mutant for acerebellar (aceti282a). An enlarged tectum is present, but no cerebellum or MHB are seen (see also Fig. 7). (E) Pronephric duct (pn) in a wild type and (F) homozygous mutant embryo for noith44. In the mutant embryos, no pronephric duct is found; the arrow points to the few cells in the mutant that may represent partially developed pronephric duct cells; consistent with the presence of a normal duct during earlier stages of development. (G) Blood streaming through the common cardinal vein over the yolk in a 36 h wild-type embryo (bracketed by arrows). (H) Blood cells accumulate in a dent on the ventral yolk sac in a homozygous mutant embryo for noith44. No such accumulation is seen in wild-type embryos. Embryos in A and B and E-F were placed in PTU at about 26 h to suppress formation of melanin. cb, cerebellum; ey, eye; tec, tectum; ov, otic vesicle; pn, pronephric duct. Scale bar, 150 µm (A-D), 60 µm (E-H). PHENOTYPE:
|
|
Apoptosis in the tectum of noi mutant embryos. Shown is a comparison of Nomarski (A-C) and fluourescence (D-F) images of the same embryos, stained with acridine orange to detect apoptotic cell death. (A,D) Wild-type embryo at 22 h of development; (B,E) homozygous mutant noitu29a embryo at 22 h, stained with acridine orange. (C,F) Homozygous mutant noity22b embryo at 26 h, stained with acridine orange. Large numbers of stained cells are observed in the tectum of mutant embryos in E and F (arrows) that are absent in the wild type. Degenerating, more refractile cells are also seen with Nomarski optics in C (between arrowheads). Scale bar, 120 µm. PHENOTYPE:
|
|
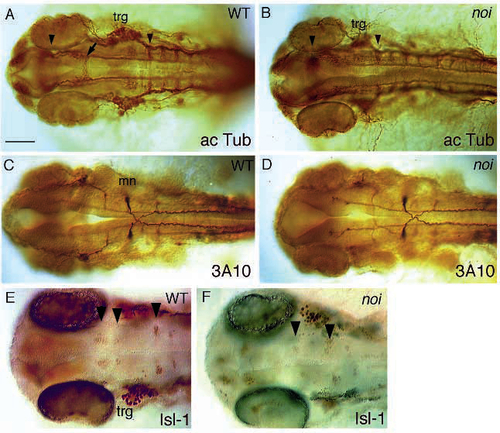
Overall organisation of the CNS in noi mutant embryos. Shown are dorsal views of wild type and mutant embryos stained with antibodies to reveal axonal organisation and formation of neuronal nuclei. (A,B) Anti-acetylated-tubulin staining in 30 h wild-type and mutant noity22b embryos. Arrowheads point to the position of the tract of the posterior commissure, and to the first hindbrain commissure. Notice the reduced distance. The arrow in A points to the ventral tegmental commissure that is absent in the mutant noi embryo in B. (C,D) mAb 3A10 staining in 30 h wild-type and mutant noith44 embryos. The Mauthner neuron (mn) and the other T reticular interneurons stained by this antibody develop normally, indicating that hindbrain development is largely normal in noi embryos. (E,F) Isl staining of 32 h wild-type and mutant noith44 embryos. Arrowheads point to bilateral pairs of clustered neuronal nuclei stained by this antibody in the midbrain and hindbrain (see text). Whereas two clusters are seen in the midbrain of the wild-type embryo in E (probably corresponding to the nucleus of the medial longitudinal fascicle), only one such pair of reduced size is seen in the mutant. Notice the reduced distance to the rhombomere 2 cluster. A group of 2-3 nuclei in rhombomere 1 is also unaffected in the mutant, but not in focus. trg, trigeminal ganglion. Scale bar, 100 µm (A-D), 75 µm (E, F). |
|
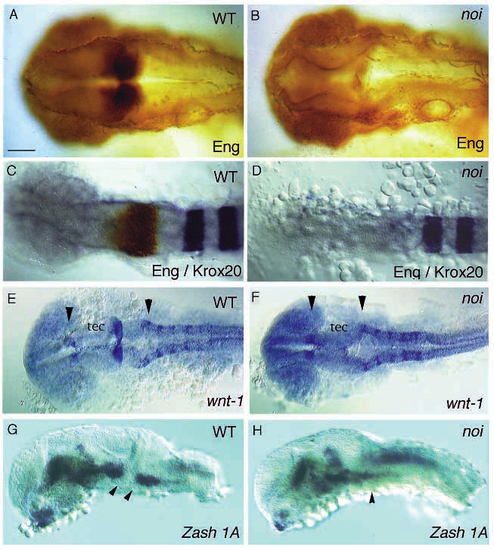
Expression of markers of the MHB in noi. Shown are dorsal (A-F) and lateral (G,H) views of wild type and noith44 mutant embryos, stained with markers of the MHB as indicated. (A,B) mAb4D9 staining for eng proteins at 28 h. Staining is eliminated in the MHB of mutant embryos. (C,D) Doubly stained embryos at the 8-somite stage, for krx20 RNA in blue and eng proteins in brown. No eng proteins are detected in the primordium of the MHB of mutant embryos. (E,F) Expression of wnt1 at the 20-somite stage posterior to the tectum (tec) is eliminated in noi, as is the posteriorly adjoining cerebellar fold. Notice the reduced distance between the wnt1-expressing domains in the dorsal forebrain and the first rhombomere (arrowheads). (G,H) zash1A staining at 26 h. A gap of staining (arrowheads) is observed at the MHB between the tegmental and rhombomeric expression domains, which is absent in the mutant embryo. Scale bar, 75 mm (A,B,G,H); 100 µm (C,D); 120 µm (E,F). EXPRESSION / LABELING:
PHENOTYPE:
|
|
Pax-b protein, but not RNA expression, is affected in noi. Shown are dorsal views of wild-type and noi mutant embryos (axial view as optical section in C and D), stained for pax-b protein and RNA, as indicated. (A-D) pax-b antibody staining is eliminated from (A,B) the MHB primordium (asterisks) and otic placode (op) and (C,D) the pronephric anlage (arrow) of mutant embryos at the 5-somite stage. (E,F) pax-b RNA expression in wild type and noith44 mutant embryos. RNA expression is found in all tissues of the mutant, except for the MHB (arrow) and pronephros. Notice the overall reduction of RNA level in the mutant, which is not yet apparent at the 14-somite stage in (G,H): only a trace of RNA can be detected in the MHB of mutant embryos at this stage (arrowhead), whereas RNA expression in the optic stalk (os), otic placode, pronephric anlage and hindbrain neurons is normal. Scale bar, 100 µm (A,B); 50 µm (C,D); 140 µm (E-H). EXPRESSION / LABELING:
|
|
Development of living ace embryos. Shown are lateral or dorsal (E,F) views of whole-mounted living wild type (left) and ace mutant siblings (right) as indicated. (A,B) At the 5-somite stage, mutant embryos show a bulge in the prospective midbrain primordium (arrow). (C,D) At the pharyngula stage, mutant embryos lack a cerebellum and the MHB fold, but show an enlarged tectum. The embryos were placed into PTU to suppress melanization. Arrowheads point to rhombomere boundaries. (E,F) In a dorsal view at 30 h, the absence of the MHB fold and the increased size of the tectum are particularly evident. Arrowheads point to the posterior edge of the tectum, coincident with the MHB fold in the wild type, and to the otoliths in the otic cyst. Notice the reduced size of the otic cyst in the mutant. (G,H) High magnification view of the otic capsule of the free swimming larva (day 6): notice the smaller size, and the truncated semicircular canal (arrow in H). Only one otolith is apparent in the mutant embryo. cb, cerebellum; oc, otic cyst; scc: semicircular canal; tec, tectum. Scale bar, 100 µm (A-D); 75 µm (E,F); 65 µm (G,H). PHENOTYPE:
|
|
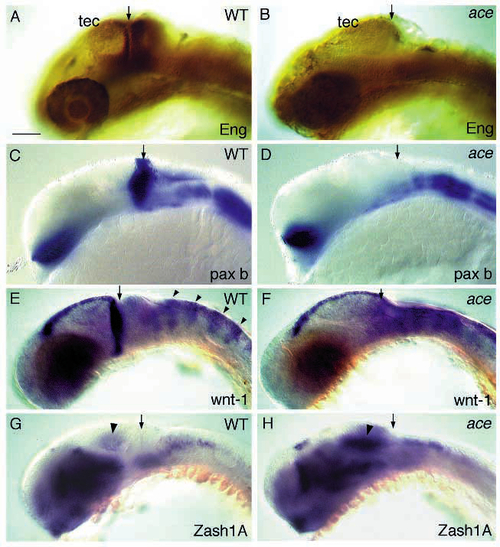
Expression of MHB markers is affected in ace embryos. Shown are lateral views of wild-type (left) and ace mutant (right) embryos. Arrows point to the posterior border of the tectum. (A,B) eng protein (dark brown staining around the MHB) is not detected in the tectum, tegmentum and cerebellum of 28 h ace mutant embryos. (C,D) pax-b expression is missing in the posterior midbrain of 24 h ace mutant embryos, but is unaffected elsewhere. (E,F) wnt1 expression is not found in the posterior tectum of 26 h ace mutant embryos. Notice the extension of the wnt1 stripe in the dorsal tectum to more posterior levels. Expression in the rhombomeres (arrowheads) is not affected. (G,H) Expression of Zash1A in the tectal ventricular zone (arrowhead) is strongly increased in the mutant embryo compared to its sibling at 26 h. Stainings of the embryos in G and H were done in the same vial. Scale bar, 90 µm. |

Unillustrated author statements |