- Title
-
Vegf Signaling Promotes Vascular Endothelial Differentiation by Modulating etv2 Expression
- Authors
- Chetty, S.C., Rost, M.S., Enriquez, J.R., Schumacher, J.A., Baltrunaite, K., Rossi, A., Stainier, D.Y., Sumanas, S.
- Source
- Full text @ Dev. Biol.
|
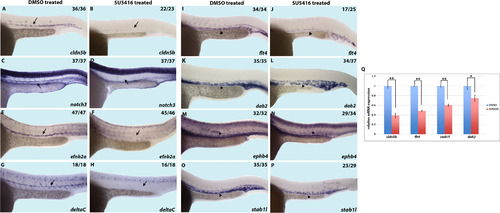
Inhibition of Vegfr signaling by SU5416 chemical treatment decreases arterial and venous marker expression. (A-P) In situ hybridization analysis showing a decrease in arterial and venous vascular endothelial markers upon SU5416 treatment starting at the 50% epiboly stage (5.3 hpf), as compared to DMSO treated control embryos. Arterial markers cldn5b (A and B), notch3 (C and D), efnb2a (E and F), and deltaC (G and H) as well as venous markers flt4 (I and J), dab2 (K and L), ephb4 (M and N), stab1l (O and P), are downregulated in SU5416 treated embryos. (Q) Quantification of selected markers by qRT-PCR. Markers with high expression outside of the vasculature were excluded from qRT-PCR analysis. * indicates p<0.05 and ** indicates p<0.01, �SEM is shown. Arrows indicate the dorsal aorta and arrowheads indicate the posterior cardinal vein. Lateral view of 24hpf embryos, anterior to the left. Numbers of embryos showing displayed phenotype are listed. |
|
Inhibition of Vegfr signaling with a high dose of SU5416 decreases vascular endothelial differentiation while low doses of SU5416 inhibit arterial and promote venous specification. (A-T) In situ hybridization analysis of arterial (cldn5b), venous (stab1l, dab2) and pan-endothelial (etv2) marker expression in embryos treated with 2.5?20 ?M doses of SU5416 starting at the 50% epiboly stage. Note that cldn5b expression is reduced in embryos treated with all doses of SU5416, while stab1l expression appears to be only mildly downregulated or unchanged at the lowest dose of SU5416. In contrast, dab2 expression is expanded into the artery in embryos treated with 2.5?10 ?M doses (G,K,O), and reduced in embryos treated with a 20 ?M dose (S). The expression of etv2 is reduced in embryos treated with medium and high doses of 5?20 ?M SU5416 (L,P,T). Lateral view of 24 hpf embryos, anterior to the left. Arrows indicate the DA and arrowheads indicate the PCV. Numbers of embryos showing the displayed phenotype are listed. (U) qPCR analysis of arterial (cldn5b, aqp8a), venous (flt4, stab1l, dab2, lyve1b, mrc1a) and pan-endothelial (cdh5, etv2) markers in 24hpf-old embryos treated with varied doses of SU5416 starting at the 50% epiboly stage. Note that arterial cldn5b and aqp8a expression is reduced at all doses of treatment; pan-endothelial cdh5 and etv2 as well as venous flt4, stab1l and lyve1b expression is only mildly downregulated or not changed in embryos treated with low 2.5?5 ?M doses of SU5416, and strongly downregulated in embryos treated with higher 10?20 ?M doses; venous dab2 expression is upregulated in embryos treated with low doses, and downregulated only in embryos treated with the highest (20 ?M) SU5416 dose. * indicates p<0.05 and ** indicates p<0.01, �SEM is shown. |
|
Analysis of arterial and venous marker expression in SU5416-treated embryos at 24hpf after commencing treatment at multiple developmental time points. (A-H) Embryos were treated with either 0.1% DMSO or 20 ?M SU5416 beginning at stages ranging from 15 somites to 23 somites and analyzed at 24 hpf by in situ hybridization. (A-D) Arterial marker cldn5b was severely downregulated when treated with SU5416 beginning at the 15-somite stage (16.5 hpf, B) as compared to DMSO treated controls (A). A similar phenotype was observed when treated at the 20-somite stage (19 hpf, C) but was less apparent when treated at the 23 somite stage (20.5 hpf, D). (E-H) Venous marker flt4 was downregulated upon SU5416 inhibition at the 15-somite stage (F) as compared to DMSO treated controls (E). Milder downregulation was observed when treatment was started at the 20-somite and 23-somite stages (G,H) and expansion of flt4 expression into the DA was observed. Arrows indicate DA and arrowheads indicate PCV. Lateral view of 24 hpf embryos, anterior to the left. Numbers of embryos showing the displayed phenotype are listed. (I) qPCR analysis of arterial (cldn5b, aqp8a), venous (flt4, stab1l, dab2, lyve1b, mrc1a) and pan-endothelial (cdh5, etv2) marker expression in SU5416-treated embryos. The treatment was started at the stages indicated and embryos were analyzed at 24 hpf. Note that arterial marker expression is greatly reduced at all stages indicated, while venous marker expression is significantly reduced at the early treatment stages (50%-epiboly and 5-somite), and only slightly reduced or unaffected (flt4, stab1l) or even expanded (dab2, 23-somite) at the later treatment stages. Pan-endothelial markers show strong reduction at early treatment stages and only slight or no reduction at the later treatment stages. * indicates p<0.05 and ** indicates p<0.01,�SEM is shown. |
|
High concentrations of SU5416 result in apoptosis in trunk vasculature at 24hpf and 20-somite stages. (A,B) Caspase 3 immunofluorescent staining of 24hpf embryos treated with either DMSO or 10 ?M SU5416 starting at the 50% epiboly stage. Strong Caspase staining, indicating apoptosis, was observed within trunk and tail vasculature in the 10 ?M SU5416-treated embryos (B) as compared with the DMSO-treated control embryos (A). (C-H) Immunofluorescent staining of apoptotic marker Caspase3 in 20-somite stage embryos treated with either DMSO or 20 ?M SU5416. Embryos treated with SU5416 showed strong Caspase3 staining which co-localized with vascular fli1:GFP fluorescence (F-H) compared to DMSO-treated embryos which did not have Caspase staining in the trunk vascular region (C-E). (I-N) 15-somite stage embryos treated with SU5416 did not appear to have apoptosis in the trunk region (L-N) and had comparable Caspase3 staining to the DMSO-treated controls (I-K). |
|
Analysis of arterial and venous markers in vegfaa- overexpressing embryos. (A-R) In situ hybridization analysis of multiple arterial and venous endothelial markers upon vegfaa121 and vegfaa165 RNA injection as compared to uninjected controls. Arterial markers cldn5b (A and B), deltaC (C and D) and notch3 (E and F), venous markers stab1l (G and H) and flt4 (I and J), and pan-endothelial marker cdh5 (Q and R) are all expanded upon vegfaa RNA injection. In contrast, venous dab2, lyve1b and mrc1a expression is reduced in vegfaa overexpressing embryos (K-P). Note that dab2 and lyve1b expression is mostly confined to the PCV while stab1l and flt4 display some arterial expression in wt embryos (G,I,K,M). Arrows indicate the DA and arrowheads indicate the PCV. Lateral view of 24 hpf embryos, anterior to the left. Numbers of embryos showing the displayed phenotype are listed. (S) qPCR analysis of arterial (cldn5b, aqp8a), venous (flt4, stab1l, dab2, lyve1b, mrc1a) and pan-endothelial (cdh5, etv2) markers in vegfaa and control mRFP overexpressing embryos as compared to uninjected controls (wt). Note the significant increase in arterial and pan-endothelial marker expression and decrease in venous dab2, lyve1b and mrc1a expression in vegfaa overexpressing embryos. *, p<0.05; ** p<0.01 as compared to wt embryos, �SEM is shown. |
|
Vegf signaling upregulates etv2 expression. In situ hybridization analysis showing a decrease or increase in etv2 expression upon Vegfr inhibition or vegfaa overexpression, respectively. (A-D) Chemical inhibition of Vegfr with either SU5416 (A,B) or PTK787 (C,D) treatment starting at the 50% epiboly stage results in a decrease in etv2 expression when analyzed at 24 hpf. (E,F) vegfaa RNA overexpression results in an expansion of etv2 expression. Lateral view of 24 hpf embryos, anterior to the left. Numbers of embryos showing the displayed phenotype are listed. (G) etv2 expression in embryos treated with SU5416 at the 50% epiboly stage and analyzed at 2?3-somite (11 hpf), 10-somite (14 hpf), and 24 hpf stages was quantified by qRT-PCR. Note that etv2 expression in control DMSO treated embryos is increased at the 10-somite and 24 hpf stages compared to the 2?3 somite stage and this upregulation is reduced in SU5416 treated embryos while its initial expression at the 2?3 somite stage is not affected. All expression values are shown relative to etv2 expression at 24 hpf in DMSO treated embryos. (H) Quantification of etv2 expression at 24 hpf by qPCR in PTK787 treated and vegfaa RNA injected embryos. ** denotes p<0.01; * denotes p<0.05, �SEM is shown. Arrows indicate DA and arrowheads indicate PCV. |
|
fli1:etv2 DNA injection rescues arterial and venous marker expression in Vegfr inhibited embryos. (A-H) In situ hybridization analysis showing expression patterns of arterial and venous markers upon Vegfr inhibition and etv2 DNA overexpression. (A,B) Uninjected embryos treated with DMSO displayed normal expression patterns of arterial marker cldn5b (A) and venous marker flt4 (B). (C,D) fli1:etv2 DNA injection does not significantly affect endogenous vascular endothelial marker expression, but did result in small patches of ectopic expression (white arrowheads). (E,F) SU5416 inhibition results in downregulation of arterial cldn5b (E) and venous flt4 (F) marker expression. Embryos injected with fli1:etv2 DNA and treated with SU5416 have increased cldn5b (G) and venous flt4 (H) vascular endothelial marker expression, as compared to SU5416 treated embryos (E,F) demonstrating a partial rescue of Vegfr inhibition phenotype. Arrows indicate DA and black arrowheads indicate PCV. Lateral view of 24 hpf embryos, anterior to the left. Numbers of embryos showing displayed phenotype are listed. (I) qPCR analysis of arterial (cldn5b), venous (flt4, dab2, stab1l) and pan-endothelial (cdh5) marker expression. Note that etv2 overexpression under fli1 promoter partially or completely rescues all marker expression in SU5416 treated embryos. *, p<0.05; ** p<0.01; significance of expression values in fli1:etv2+DMSO embryos was compared to wt+DMSO; �SEM is shown. |
|
Analysis of vascular marker expression in vegfaa mutant embryos. (A-P) In situ hybridization analysis of arterial marker cldn5b (A,B), ephb2a (C,D), notch3 (E,F), pan-endothelial marker etv2 (G,H) and venous marker stab1l (I,J), dab2 (K,L), lyve1b (M,N), and mrc1a (O,P) expression in vegfaa-/- and wild-type sibling embryos at 24 hpf. Note the absence of arterial marker expression in vegfaa mutants (B,D,F) while venous markers are expanded into the arterial region of the single vessel present in vegfaa mutants (J,L,N,P). etv2 expression is reduced in vegfaa mutants and intersegmental vessels are absent due to the failure of angiogenesis (G,H). (Q) qPCR analysis of vascular marker expression in vegfaa-/- embryos compared to their siblings at 24 hpf. Note that all arterial and pan-endothelial markers are strongly downregulated, while flt4 and stab1l are only mildly downregulated. In contrast, venous-specific dab2 and lyve1b expression is increased, while mrc1a expression is unchanged. * p<0.05; ** p<0.01, �SEM is shown. |
|
All vascular endothelial cells acquire venous identity in Vegfaa MO knockdown embryos. (A-C) Vascular endothelial cell numbers counted within a selected region of two-somites in wild-type siblings and vegfaa-/-; fli1:nGFP; kdrl:mCherry embryos at 24 hpf. Only cells in the axial vasculature (not ISVs) were counted in the control embryos. Because vegfaa mutants form only a single vessel, it was not possible to assess arterial and venous cell numbers separately. There was no significant difference in cell numbers between the vegfaa mutants (n=7) and their wildtype siblings (n=8). (D-I) All vascular endothelial cells acquire venous identity in Vegfaa MO-injected stab1l:YFP; kdrl:mCherry embryos, analyzed by confocal microscopy at 48?52 hpf. Note that stab1l:YFP expression is largely restricted to the PCV and is absent from the DA in control uninjected embryos (D-F). In Vegfaa morphants, all vascular endothelial cells exhibit stab1l:YFP fluorescence (G-I). Transverse optical cross section is shown in the insets on the right; the position of cross section is marked with a dashed line (F,I). |
|
Arterial marker expression is restored by etv2 overexpression in vegfaa mutant embryos. In situ hybridization analysis of arterial cldn5b expression at 24 hpf. vegfaa mutant embryos and wild-type siblings were injected with fli1a:etv2-2A-mCherry DNA overexpression construct together with tol2 mRNA. Note the absent cldn5b expression in vegfaa-/- embryos (C) and restored and expanded cldn5b expression in (D). ISV sprouting was not rescued in fli1:etv2-mCherry; vegfaa-/- embryos. |
|
PTK787 chemical inhibition decreases arterial and venous marker expression and overall endothelial differentiation. (A-F) In situ hybridization analysis for arterial cldn5b and venous flt4 and stab1l expression at 24 hpf. Embryos were treated with 200 �M PTK787 beginning at the 50% epiboly stage. Note the downregulation of cldn5b and flt4 expression in PTK787 treated embryos. Arrows indicate DA and arrowheads indicate PCV. Numbers of embryos showing the displayed phenotype are listed. (G) qPCR analysis of arterial (c/dnSb), venous (flt4, stab 1 /, dab2) and pan-endothelial (cdh5, etv2) marker expression in PTK787 treated embryos at 24 hpf. Note that arterial marker, pan-endothelial marker and venous flt4 expression is reduced, while stab1l and dab2 expression is not changed significantly.** denotes p<0.01; * denotes p<0.05; NS, not significant. � SEM is shown. |
|
Analysis of marker expression and apoptosis in 28hpf embryos treated with SU5416 beginning at the 23-somite stage (20.5 hpf). (A) qRT-PCR analysis shows a significant decrease of all vascular markers in SU5416-treated embryos compared to DMSO-treated controls at 28hpf, after commencing treatment at the 23-somite stage. (B-G) Double immunofluorescent staining of active Caspase3 and GFP at 28hpf in fli1:GFP embryos treated with either DMSO or 20�M SU5416 (beginning at the 23-somite stage). Note the absence of Caspase3 staining in the trunk region of both the DMSO- and SU5416-treated embryos (C,F), indicating a lack of apoptosis in the trunk vasculature. |
|
Transient fli1:etv2-2A-mCherry expression in the vasculature does not affect the number of vascular endothelial cells. (A-C) Confocal images of trunk vasculature of a fli1:GFP embryo injected with a fli1:etv2-2A-mCherry construct at 24 hpf. Note that mCherry is expressed in a mosaic fashion within the vasculature. (D-F) Confocal images of trunk axial vasculature in a control uninjected fli1:nGFP embryo (D) and a fli1:etv2-2A-mCherry injected embryo (E,F) at 24 hpf. (G) Quantification of vascular endothelial cell number in uninjected fli1:nGFP embryos (n=6) and fli1:nGFP embryos injected with fli1:etv2-2A-mCherry construct (n=8) at 24 hpf. No significant difference in endothelial cell number was observed between control and injected embryos. The cells were quantified in the axial vasculature in a two somite region using lmaris software. |
|
vegfaa mutants do not display apoptosis in trunk vasculature at 24hpf. (A-F) Double immunofluorescent staining of Caspase3 and mCherry in vegfaa mutants (in a kdrl:mCherry background). mCherry staining marks vasculature, while Caspase staining indicates cells undergoing apoptosis. vegfaa mutants did not show Caspase staining in the trunk vasculature (E) and were similar to their wild type siblings (B). |
Reprinted from Developmental Biology, 424(2), Chetty, S.C., Rost, M.S., Enriquez, J.R., Schumacher, J.A., Baltrunaite, K., Rossi, A., Stainier, D.Y., Sumanas, S., Vegf Signaling Promotes Vascular Endothelial Differentiation by Modulating etv2 Expression, 147-161, Copyright (2017) with permission from Elsevier. Full text @ Dev. Biol.