Fig. 5
- ID
- ZDB-FIG-111129-28
- Publication
- Goudevenou et al., 2011 - Def6 Is Required for Convergent Extension Movements during Zebrafish Gastrulation Downstream of Wnt5b Signaling
- Other Figures
- All Figure Page
- Back to All Figure Page
|
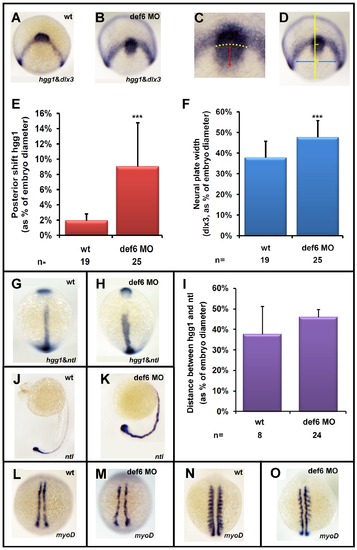
The def6 MO-mediated knockdown phenotype induces CE movement defects. Uninjected (wt) or embryos injected with def6 MO were fixed at tail-bud stage and in situ hybridisations were carried out with probes to hgg1 and dlx3 (A,B). ImageJ software was utilised to analyse the staining patterns, measuring the posterior shift of the hgg1 staining (red double-headed arrow) in relation to the arc formed by dlx3 expression (yellow dotted arc) (C), and measuring the width of the dlx3 staining (blue double-headed arrow) at a constant distance (1/4 of the embryo width) from the dlx3 arc when the embryo was positioned dorsally (D). (E and F) The measured distances were plotted as the average posterior shift (E) or width (F) as a percentage of the total width of the embryo. Two-tailed Student′s t-tests were carried out between groups indicated, and were of statistical significance (p<0.001; three asterisks). This experiment has been repeated at least three times; a representative experiment is depicted here. Zebrafish embryos uninjected (wt) or injected with def6 MO (2.5 ng) were also stained for hgg1/ntl (G, H; statistical analysis shown in Panel I), ntl (J, K) and myoD (L-O-) expression. Images G, H, L, M are embryos at tail-bud stage. Images J. K show embryos at 24 hpf. Images N, O show embryos at the 10-somite stage. G, H, L-O are dorsal views with anterior to the top; J, K are lateral views with anterior to the left. |
| Genes: | |
|---|---|
| Fish: | |
| Knockdown Reagent: | |
| Anatomical Terms: | |
| Stage Range: | Bud to Prim-5 |
| Fish: | |
|---|---|
| Knockdown Reagent: | |
| Observed In: | |
| Stage Range: | Bud to Prim-5 |