- Title
-
The yolk syncytial layer regulates myocardial migration by influencing extracellular matrix assembly in zebrafish
- Authors
- Sakaguchi, T., Kikuchi, Y., Kuroiwa, A., Takeda, H., and Stainier, D.Y.
- Source
- Full text @ Development
|
The YSL regulates myocardial migration. Embryos injected with a 4 bp mismatch mtx1 control MO (A,C,G), mtx1 MO(A) (B,D-F,H,N-O,Q), mtx1 MO(B) (I) or mtx1 MO(C) (J,K), into the YSL at the 1000-cell stage (3 hpf), embryos injected with mtx1 MO(C) into the YSL at the 1000-cell stage and mtx1 mRNA into the YSL at dome stage (L), and uninjected control embryos (M,P). (A-F) Live embryos at 34 hpf. (A,B) Lateral views, anterior to the left. mtx1 MO(A)-injected embryos exhibit cardiac edema (arrowhead) and a flattened hindbrain ventricle (arrow) (B). (C-F) Frontal views reveal the cardiac phenotype of control and mtx1 MO(A)-injected embryos. Approximately 30% of mtx1 MO(A)-injected embryos showed a clear cardia bifida phenotype at 34 hpf (D). Rhodamine Dextran was co-injected with mtx1 MO(A) to visualize the distribution of the injected solution, and this method revealed that it was mostly restricted to the YSL. Bright field (D) and fluorescent views (E) are merged in F. White arrowheads point to the cardiac tissues. Expression of the myocardial-specific markers, cmlc2 (G-J,P,Q) and Tg(cmlc2:GFP) (K-O), in embryos injected with mtx1 MOs into the YSL. (G-O) Dorsal views, anterior to the top. (P,Q) Frontal views, arrowheads point to the myocardial clusters. At the 21-somite stage (19.5 hpf), myocardial cells have fused at the midline and form a shallow cardiac cone in control MO-injected embryos (G), whereas they remained bilateral in mtx1 MO-injected embryos (H-K). (H-K) Three independent mtx1 MOs, MO(A) (H), MO(B) (I), and MO(C) (J,K), caused the same phenotype in a fully penetrant manner when examined at the 21-somite stage (19.5 hpf). (L) mtx1 mRNA injection into the YSL at dome stage rescued the myocardial migration defect of mtx1 MO(C)-injected embryos (21-somite stage, 19.5 hpf, shown). At 24 hpf, myocardial cells have fused at the midline and begun to form a tube that extends toward the left anterior part of control uninjected embryos (M), whereas myocardial cells have just fused (N) or not yet fused (O) in mtx1 MO(A)-injected embryos. At 38 hpf, myocardial cells form a single tubular functioning heart in uninjected control embryos (P), but these cells formed two clearly distinct structures in approximately 30% of mtx1 MO(A)-injected embryos (Q). EXPRESSION / LABELING:
|
|
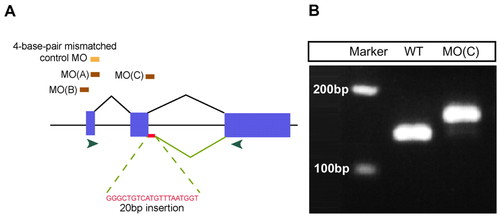
Specificity of mtx1 MOs. (A) Schematic diagram of mtx1 genomic structure and position of mtx1 MOs. mtx1 has three exons (blue boxes) and two introns. Two independent ATG MOs, MO(A) and MO(B), and a splice donor MO, MO(C), caused essentially the same phenotype when injected into the YSL at the 1000-cell stage. The splice donor MO, mtx1 MO(C), leads to a 20 bp insertion in mtx1 mRNA, which leads to a frame shift and premature termination. Green arrowheads indicate the position of the primers used for the RT-PCR analysis shown in B. (B) RT-PCR analysis shows that an additional 20 bp are integrated into mtx1 mRNA in mtx1 MO(C)-injected embryos. EXPRESSION / LABELING:
|
|
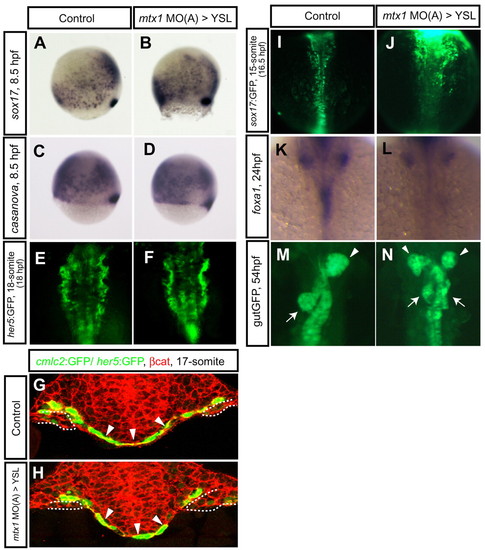
Endoderm development in mtx1 MO-injected embryos. Endoderm differentiation and morphology in wild-type embryos (A,C,E,G,I,K,M) and embryos injected with mtx1 MO(A) into the YSL (B,D,F,H,J,L,N). Expression of early endoderm markers, sox17 (A,B) and casanova/sox32 (C,D), at 80% epiboly (8.5 hpf) did not appear to be affected in mtx1 MO(A)-injected embryos. (E,F) Anterior views of her5:GFP transgenic embryos at the 18-somite stage show unaffected pharyngeal endoderm development. (G,H) The pharyngeal endoderm was also examined in transverse confocal images (dorsal at the top) of Tg(cmlc2:GFP); Tg(her5:GFP)ne2067 embryos counterstained for β-catenin (red). Arrowheads point to the pharyngeal endoderm and dashed lines outline the myocardial cells in the lateral plate mesoderm. (I,J) Dorsal views of the mid-trunk region of sox17:GFP transgenic embryos at the 15-somite stage. Anterior to the top. sox17:GFP-positive endodermal cells in the future foregut region are coalescing toward the midline in control embryos (I), whereas they appeared to be delayed in their migration in mtx1 MO(A)-injected embryos (J). (K,L) Dorsal views of foxa1 expression in the digestive organ-forming region at 24 hpf. By 24 hpf, endodermal cells have already coalesced at the midline and formed a rod in wild-type embryos (K), whereas they remained as a sheet in mtx1 MO(A)-injected embryos (L). (M,N) Ventral views of gutGFP expression at 54 hpf. Approximately 30% of mtx1 MO-injected embryos showed duplicated hepatic (white arrowheads) and pancreatic (white arrows) buds at this stage (N), further illustrating the endoderm morphogenesis defects. EXPRESSION / LABELING:
|
|
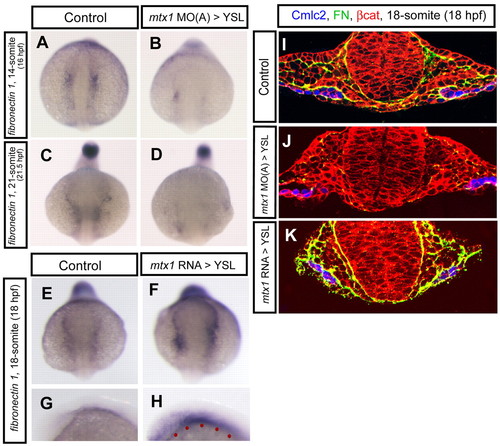
fibronectin 1 expression is regulated by mtx1. fibronectin 1 gene expression and Fibronectin protein localization around the migrating myocardial cells were examined in embryos injected into the YSL with mtx1 MO(A) or mtx1 mRNA. (A-F) Anterior views, (G,H) Lateral views in the head region. (A-H) fibronectin 1 expression at the 14-(A,B; 16 hpf), 18-(E-H; 18 hpf), and 21-somite stages (C,D; 21.5 hpf). (A-D) fibronectin 1 expression appeared to be reduced in mtx1 MO(A)-injected embryos. (E-H) Conversely, fibronectin 1 expression appeared to be increased in mtx1 mRNA-injected embryos. (F,H) fibronectin 1 expression was upregulated mostly in embryonic tissues, even though mtx1 mRNA was injected into the YSL (red dots in H), indicating that mtx1 regulates fibronectin 1 expression in a non-cell-autonomous manner. (I-K) Transverse sections of cmlc2:GFP (false colored blue) transgenic embryos immunostained for β-catenin (red) and Fibronectin (green). Fibronectin deposition appeared to be decreased in mtx1 MO-injected embryos (J), whereas it appeared to be increased in mtx1 mRNA-injected embryos (K) specifically in embryonic tissues. EXPRESSION / LABELING:
|
|
Genetic interaction between natter/fibronectin 1 and mtx1. (A-D) Lateral views of bright field images at 34 hpf, anterior to the left. (A) natter heterozygote, (C) natter homozygous mutant, (B) natter heterozygote injected with mtx1 MO(A) into the YSL and (D) natter homozygous mutant injected with mtx1 MO(A) into the YSL. The partial or complete reduction of fibronectin 1 in mtx1 MO-injected embryos uncovered novel phenotypes. |
|
Morphogenesis of mtx1 MO-injected heterozygous natter embryos. (A-E) Wild-type embryo; same embryo is shown at all stages. (F-J) heterozygous natter embryo injected with mtx1 MO(A) into the YSL at the 1000-cell stage; same embryo is shown at all stages. Lateral views, animal pole up and dorsal to the right (A-C,F-H); dorsal views, animal pole up (D,I), lateral views, anterior to the left and dorsal up (E,J). mtx1 MO-injected heterozygous natter embryo showed no apparent defect at the tailbud stage (A,F). A phenotype first appeared at the 6-somite stage at the hindbrain level as the YSL got darker (G). At the 17-somite stage, the head ventricle was almost collapsed onto the YSL (I). At 24 hpf, somite boundaries are clearly visible in wild-type embryos (E), whereas they were harder to visualize in mtx1 MO-injected heterozygous natter embryos (J), especially in the anterior part. |
|
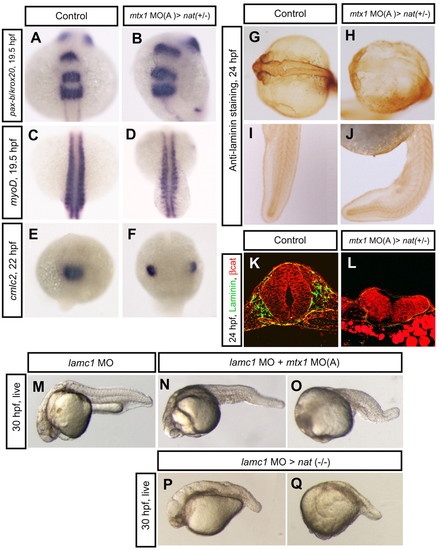
Regional specification and ECM formation in mtx1 MO-injected heterozygous natter embryos. (A,B) Dorsal views of hindbrain, (C,D) somite and (E,F) heart regions. pax-b and krox20 expression in the midbrain-hindbrain boundary and rhombomeres 3 and 5 at 19.5 hpf (A,B), and myod expression in the segmented somites and adaxial cells at 19.5 hpf (C,D), appeared unaffected in mtx1 MO-injected heterozygous natter embryos. (E,F) cmlc2 expression in the myocardial cells at 22 hpf reveals that mtx1 MO-injected heterozygous natter embryos displayed cardia bifida. (G-L) Laminin immunostaining at 24 hpf. Laminin deposition appeared to be greatly reduced in the hindbrain region of mtx1 MO-injected natter heterozygous embryos (H), but not in the trunk and tail (J). (K,L) Transverse sections of embryos (anterior region) immunostained for β-catenin (red) and Laminin (green). Laminin deposition was greatly downregulated and the head structure collapsed in mtx1 MO-injected heterozygous natter embryos (L). (M-Q) Lateral views of bright field images at 30 hpf, anterior to the left. Embryos injected with laminin c1 MO at the 1-cell stage exhibited shortened body axis and defects in notochord differentiation (M). Embryos injected with lamimin c1 MO at the 1-cell stage and mtx1 MO(A) into the YSL at the 1000-cell stage showed enhanced phenotypes in the hindbrain region (N) or entire head region (O). Homozygous natter mutant embryos injected with laminin c1 MO (P,Q). EXPRESSION / LABELING:
|