- Title
-
Nuclear Receptor Subfamily 2 Group F Member 1a (nr2f1a) Is Required for Vascular Development in Zebrafish
- Authors
- Wu, B.J., Chiu, C.C., Chen, C.L., Wang, W.D., Wang, J.H., Wen, Z.H., Liu, W., Chang, H.W., Wu, C.Y.
- Source
- Full text @ PLoS One
|
Spatiotemporal expression of nr2f1a during development. (A) nr2f1a expression can be observed at the 18S stage in the lateral plate mesoderm (lpm), the telencephalon (t), diencephalon (d) and hindbrain (h). (A′) Dorsal view of embryos show that nr2f1a is expressed at lpm and the axial vessels (av). (B, B′) At 24 hpf, nr2f1a is expressed in the telencephalon (t), diencephalon (d), hindbrain (h), as well as in vessels (v), and caudal vein plexus (CVP) of the trunk. B′ is an enlargement of B. (C, D) Cross sections of embryos from B′ show that nr2f1a is expressed in dorsal aorta (da), posterior cardinal vein (pcv), and caudal vein plexus (CVP). (E, E′) At 30 hpf, nr2f1a expression continues in the head, vessels (v), intersegmental vessels (ISV) and caudal vein plexus (CVP) of the trunk. E′ is an enlargement of E. Scale bars in all figures represent 100 µm. EXPRESSION / LABELING:
|
|
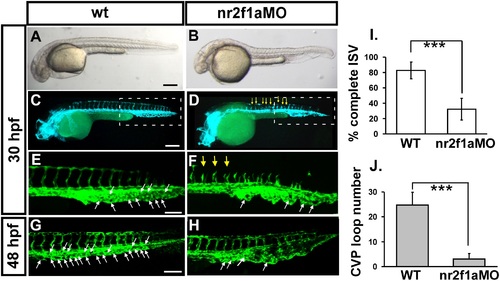
Knockdown of nr2f1a causes defects in zebrafish vascular development. (A-H) Loss of nr2f1a showing ISV growth defect (yellow arrows in F) and mis-pattern plexus at the CVP (caudal vein plexus) (white arrows) compared to wild-type control (E) at 30 hpf. E and F is enlargement of C and D, respectively. At 48 hpf, the swallowed plexus becomes obvious (H). At 30 hpf, in uninjected control embryos, intersegmental vessels (isv) have reached the DLAV at the dorsal aspect of the embryo (C, E) and the caudal vein plexus (cvp) formed honeycomb-like structures at the tail (E, white arrows). At the same stage ISVs are stalled at mid-somite in nr2f1ae2i2 morphants (D, F). (I) Quantification of percentage of completed ISV shows a ~50% increase compared to nr2f1a morphants (n = 26 in wt and n = 34 in nr2f1aMO) at 30 hpf. (J) Quantification of loop formation at CVP shows a 4-fold decreased in nr2f1a morphants (n = 10 in wt and in nr2f1aMO) at 48 hpf. (*** refers to p<0.0001 by an unpaired student′s t-test. Scale bars are 200 µm for A-D, and 100 µm for E-H. |
|
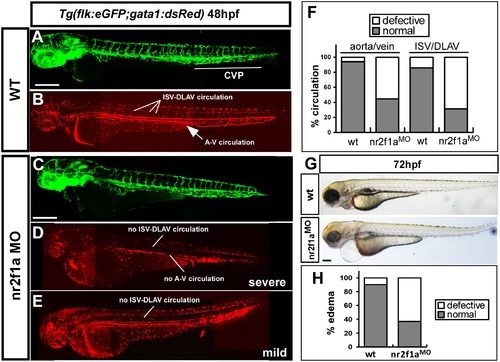
Loss of nr2f1a results in circulation defects and pericardial edema. (A-E) nr2f1ae2i2 MO was injected into transgenic Tg (fli:eGFPy1; gata1:dsRed) embryos with GFP-labeled endothelial cells (A, C) and dsRed-labeled blood cells (B, D, E). Loss of nr2f1a showed a mis-pattern at ISV and at the caudal vein plexus (CVP) and results in circulation defects in severe (D, no circulation and blood stock) or mild (E, blood stock at CVP and shorter circulation) at 52 hpf compared to wild-type fish (B). (F) Circulation defects at the ISV/DLAV (~68% in morphants) and slow to lose axial circulation of the aorta/vein in the trunk region (~58% in morphants) are quantitated in wt (n = 37) and nr2f1aMO (n = 42) at 48-52 hpf. (G, H) Representative edema fish and quantitative results from three independent experiments showed 65% of nr2f1a morphants (n = 69) with mild to severe pericardial edema compared to wt (n = 30). The scale bar in A-E represent 200 µm and in G is 500 µm. PHENOTYPE:
|
|
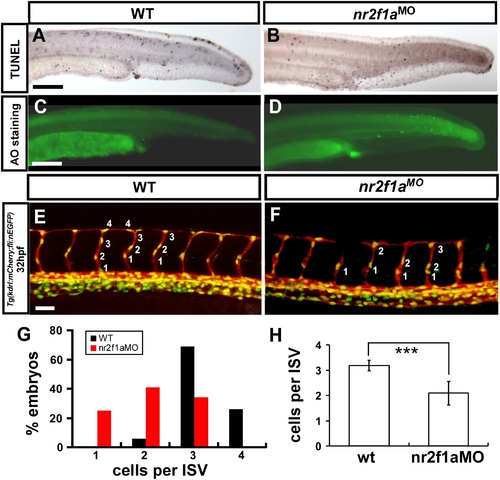
nr2f1a is required for the growth of ISV cells. (A-D) TUNEL labeling and AO staining was used to detect apoptotic cells in wild type, and nr2f1ae2i2 morphants. Although some apoptotic cells were observed on the skin, cell death in vascular regions was not elevated compared to wild type controls. (E-H) The number of cells forming each ISV counted in wild type control Tg(kdrl:mCherry; fli1a:negfp)y7 (E) and nr2f1a morphant embryos (F) at 32 hpf. (G) Proportional distribution of ISVs containing 1-5 cells and (H) average ISV cells per ISV counted in both wild type and nr2f1a morphant. (*** refers to p<0.0001 by an unpaired student’s t-test). PHENOTYPE:
|
|
nr2f1a modulates the expression of vascular markers. (A-H) Compared to wild type controls (A, C, E, G), expression of the venous markers mrc1 (D) and flt4 (F) were reduced in the trunk of nr2f1a morphants at 24 hpf. In addition, expression of the arterial marker ephrinb2 (H) and pan-vascular marker stabilin decreased (B). (I) Quantification of the relative expression level by qPCR assay showed a ~25% to 50% reduced expression in vascular markers flt4 (0.66±0.14), EphB4 (0.46±0.08), gridlock (0.73±0.06) and ephrinb2 (0.59±0.05) in nr2f1a morphants. *** refers to p<0.0001, ** refers to p<0.001 and * refers to p<0.05 by an unpaired student’s t-test. Data are represented as means ± SEM. Scale bars represent 100 µm in A-H. EXPRESSION / LABELING:
|
|
Regulatory relationships between nr2f1a and Notch. (A-B) nr2f1a expression is downregulated in rbpsuh morphants (B) as compared to untreated control embryos (A). (D-E) nr2f1a expression is downregulated after treatment with DAPT (E) as compared to DMSO control embryos (D). (C, F) Quantification of the relative nr2f1a expression level by real-time qPCR assay shows a ~50% reduced expression in rbpsuh morphants (C) and DAPT-treated embryos (F) compared to uninjected embryos and DMSO-treated controls. Scale bars represent 100 µm in A, B, D and E. EXPRESSION / LABELING:
|
|
Knockdown of nr2f1a causes defects in vascular development in zebrafish by using translational block ATG morpholino (A-D) Loss of nr2f1a showed ISV growth defect (yellow arrows in B and D) and mis-pattern plexus at CVP (caudal vein plexus) (white arrows in D) at 30hpf and 52hpf. At 30 hpf, in uninjected control embryos, intersegmental vessels (ISV) have reached the DLAV of the embryo (A) and caudal vein plexus were formed honeycomb-like structures at the tail (white arrows in C). At the same stage ISVs are stalled mid-somite in nr2f1aATG morphants (B, D). (G) The number of cells forming each ISV were counted from wild type control Tg(kdrl:mCherry; fli1a:negfp)y7 and nr2f1a morphant embryos at 30 hpf, showing a significant decrease in nr2f1a morphant. (H) Quantification of percentage of completed ISV shows a ~70% increase compare to nr2f1a ATG morphants (n=24 in wt and n=20 in nr2f1aMO) at 30 hpf. (I) Quantification of loop formation at CVP at 48-52hpf shows a ~4-fold decreased in nr2f1a morphants (n=8 in wt and n=12 in nr2f1aMO). In addition, loss of nr2f1a results in circulation defects (blood stock at vessel/CVP or no circulation at ISV/DLAV region) (D′) and pericardial edema (arrowhead in F) shown at 52hpf. (J) Quantitative results showed ~80% of nr2f1a morphants (n=18) with pericardial edema compared to wt (n=20). (*** refers to p< 0.0001 by an unpaired student′s t-test. Scale bars are 50 µm for A-B, 100 mm for C-D, C′-D′, and 200 µm for E-F. |