- Title
-
Apc1-Mediated Antagonism of Wnt/beta-Catenin Signaling Is Required for Retino-Tectal Pathfinding in the Zebrafish
- Authors
- Paridaen, J.T., Danesin, C., Elas, A.T., van de Water, S., Houart, C., and Zivkovic, D.
- Source
- Full text @ Zebrafish
|
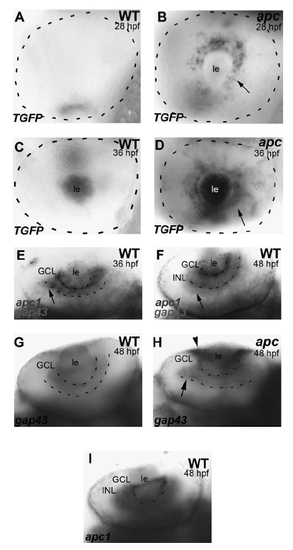
The apcCA50a/CA50a mutation results in aberrant LEF/β-catenin transcription. (A, B) In wild-type embryos at 28 hpf (A), TGFP is absent from the retina. In apc mutants (B), TGFP is expressed in the retinal epithelium surrounding the lens (arrow). (C, D) At 36 hpf, TGFP is upregulated in the mutant lens and RGC layer (arrow in D). (E, F) Double in situ hybridization to apc1 and gap43, which marks differentiating neurons developing axons at 36 (E) and 48 hpf (F), shows that apc1 is expressed in areas of the retina that undergo differentiation. Dashed line indicates eye circumference (A– D) Lateral view, anterior to the left. (E, F) Ventral view, anterior to the left. (G, H) gap43 Expression in WT (G) and apc mutants (H) at 48 hpf shows disorganization of the GCL in the mutants. (I) apc1 Expression in the GCL and INL of the WT retina at 48 hpf. GCL, ganglion cell layer; INL, inner nuclear layer; le, lens; RGC, retinal ganglion cell. Scale bar = 50 μm. |
|
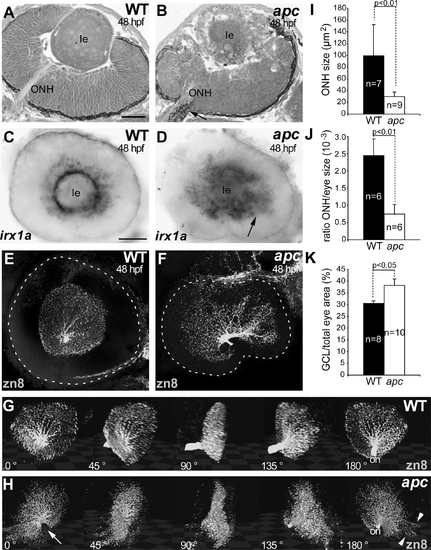
Loss of Apc function leads to mispatterning of the retina. (A, B) Transverse toluidine blue stained sections of wild-type (A) and apc mutant (B) eyes. apc Retina is abnormal, with ectopic retinal pigment around the ONH (arrow). Ventral to the left. (C, D) Expression of irx1a, a marker for RGCs, is expanded in apc mutants (arrow in D). (E, F) Lateral view of wild-type (E) and mutant (F) eye labeled with α-zn8 antibody. Dashed line indicates eye outline as observed by DAPI. The RGC layer in apc mutants (F) is irregular and disorganized. (G, H) Snapshots of a 3D reconstruction of wild-type (G) and apc (H) GCL as observed using zn8 immunolabeling. The eye is displayed at frontal view in the first frame and is rotated by 45° in each consecutive frame. apc Mutant RGC layer is disorganized, with asymmetric distribution of RGC around the lens and RGCs that are not contained within the RGC layer (H). At the choroid fissure (arrow), RGCs are lacking. Widest diameter of RGC layer is 170 μm. (I–K) The mutant ONH and eyes are significantly smaller as determined by measuring ONH area (I) and the ratio of ONH size to eye size ( J; Student’s t-test p<0.01). (K) The percentage of the area covered by RGCs relative to the entire eye area shows that in apc mutants, there is a significant increase in the GCL area size as compared to wild types ( p<0.05). (C–H) Lateral view, anterior to the left. Apc, adenomatous polyposis coli; le, lens; ONH, optic nerve head. Error bars represent standard error of the mean (SEM). Scale bar = 50 μm. |
|
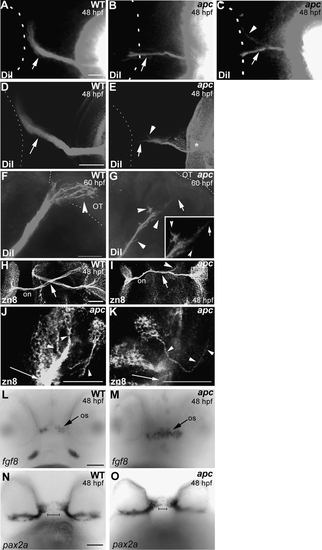
Axon pathfinding defects in apc mutants. (A–E) Whole-eye fills with DiI at 48 hpf show that in most apc mutants, the ON does not cross the OC properly. In 25% of embryos, the ON does not cross at the OC and displays branching axons (arrow in B). In 50% of mutants, a portion of the ON projects toward the optic tract (arrowhead in C), while the remaining axons project into the contralateral ON (arrow in C). In some of the apc mutants with misprojecting ON into the contralateral ON, the ON from the opposite eye appears to initiate correct projection to the OT (arrowhead in E). In addition, axons regularly branch off after exiting the eye (asterisk in E). Ventral view. The outline of the contralateral eye is indicated with dashed lines. (F, G) Lateral view of embryos with DiI-labeled ON in wild-type (F) and apc mutant (G) at 60 hpf. Overlay of fluorescence and transmission images. Anterior to the left. In wild-type embryos (F), the ON terminates in the OT (arrowhead). In apc mutants (G), some RGC axons branch off the ON (arrowheads) and the ON does not reach the tectum (arrow). Inset ON magnification. Dashed lines indicate boundary between ventral midbrain and OT. (H–K) Dorsal flatmount view of RGCs and their projections labeled with α-zn8 antibody at 48 hpf shows pathfinding defects at the OC (arrow), whereas some axons continue along the optic tract (arrowhead). Box in (I) shows magnified area in (J, K). (J, K) Single confocal planes of RGCs and their axons within the apc retina. (J) Most disorganized groups of RGCs are able to extend axons (arrowheads) toward the ONH (arrow). (K) In rare cases, RGC misproject within the retina (arrowheads) and do not contribute to the ON (arrow). Anterior is up. (L, M) Expanded fgf8 expression in the os region of apc mutants (arrow). (N, O) In apc mutants, pax2a expression is expanded toward the diencephalic midline as indicated by the bars. (L, M) Ventral view. ce, Cerebellum; ON, optic nerve; os, optic stalk; OT, optic tectum. Scale bar = 50 μm. |
|
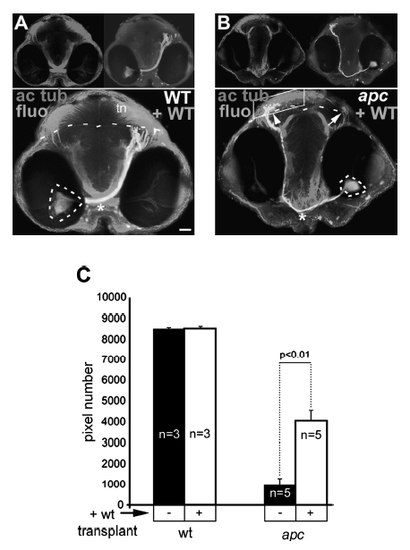
Axons from transplanted wild-type cells within the apc-/- retina are able to reach the tectum. (A, B) Transplantation of wild-type cells in retina of wild-type (A) or mutant (B) embryos. Frontal view showing location of fluorescein-labeled transplanted wild-type cells (circled with dashed lines) and their axons projecting to tectum (right panel inset) and acetylated tubulin labeling (left panel inset). The boundary between OT and tegmentum is indicated with dashed lines. In mutants (A), wild-type axons are able to cross at the OC (asterisk) and project to the mutant tectum (arrow), similarly to the axons of wild-type cells transplanted into wildtype embryos (arrow). Acetylated tubulin labeling in the OT neuropil is increased. Note the more ventral location of the OC (asterisk) in transplanted mutant versus wild-type embryos. (C) Quantification of acetylated tubulin labeling in tectum neuropil (boxed area in B) shows 4.2-fold increase of the axonal termination zone in apc mutants upon transplantation with wild-type cells into the retina (Student’s t-test p<0.01). Error bars represent standard deviation (SD). tn, Tectal neuropil. Scale bar = 50 μm. |
|
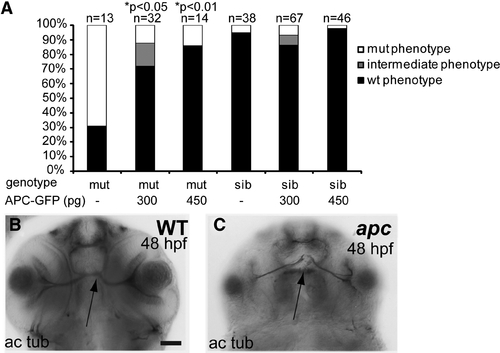
Apc axon phenotype is mainly the result of β-catenin stabilization. (A) Percentage of genotyped mutant, wildtype, and heterozygous sibling embryos, showing mutant (white), intermediate (gray), or wild-type (black) phenotypes upon injection with 300 or 450 pg apc-GFP mRNA. The number of injected embryos is indicated above the bars. Injection of apc-GFP mRNA partially rescues the axonogenesis defects as demonstrated by acetylated tubulin labeling as scored over all affected brain domains. Uninjected mutant controls show variable penetrance of axon pathfinding defects (69%; n = 13). Injection with 300 pg apc-GFP causes rescue of the mutant phenotype (72%; n = 32; p<0.05). Injecting 450 pg rescues 86% of injected mutant embryos (n = 14; p<0.01). About 5% of the sibling embryos were erroneously scored as mutants. Statistics: two-tailed Fisher’s exact probability test for a table of frequency data. p-Values are indicated above the bars. (B, C) Example of a wild-type (B) and an apc mutant embryo labeled with α-acetylated tubulin antibody showing a normal and a defective ON crossing (arrow). Acetylated tubulin labeling of fully rescued apc mutants is indistinguishable from wild-type controls. (B, C) Ventral view. Scale bar = 50 μm. PHENOTYPE:
|

Unillustrated author statements PHENOTYPE:
|