- Title
-
Zebrafish wnt8 and wnt8b share a common activity but are involved in distinct developmental pathways
- Authors
- Kelly, G.M., Greenstein, P., Erezyilmaz, D.F., and Moon, R.T.
- Source
- Full text @ Development
|
RT-PCR analyses of the temporal expression patterns of wnt8, wnt8b and max during normal embryogenesis. RT-PCR was performed on cDNAs synthesized from RNA isolated from select developmental stages. Lanes designated plus or minus refer to the presence or absence (control) of reverse transcriptase in the firststrand cDNA synthesis. (A) wnt8 expression, first detected at the 2- to 4-cell stage and corresponding to the presence of maternal transcripts, continues throughout embryogenesis. (B) wnt8b transcripts are not maternal and, unlike wnt8, they are first detected at a time corresponding to the late gastrula stage. (C) The relative consistency in the max signals throughout embryogenesis indicates that approximate equivalent amounts of cDNAs were being assayed in the wnt8 and wnt8b PCR analyses. |
|
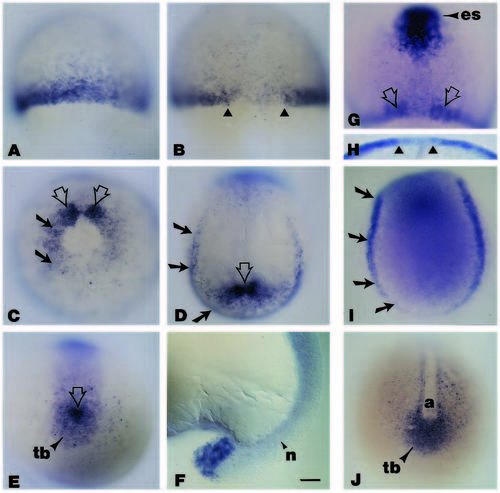
Whole-mount in situ hybridization analyses of the spatial localization of wnt8 during early gastrulation to midsegmentation. (A) Embryo at 50% epiboly illustrating the wnt8 signal in cells at the blastoderm margin; animal pole is to the top. (B) Dorsal view (animal pole to the top), of a whole-mount embryo at approximately 75-80% epiboly. Note the absence of the wnt8 signal in the dorsal midline (triangles). (C) Vegetal pole view of a whole-mount embryo at late gastrulation (90-95% epiboly); dorsal is to the top. The wnt8 signal, no longer evident on the ventral side and downregulated on the lateral sides (black arrows), becomes enriched on either side of the dorsal axial mesoderm (open arrows). (D) During early segmentation (approximately 10-11 hours), the wnt8 signal, seen in this dorsal view of a whole-mount embryo where anterior is to the top, is confined to the lateral and caudal margin of the embryonic shield (black arrows), and in the area surrounding the presumptive tailbud (open arrow). (E) Vegetal pole view illustrating the wnt8 pattern of expression at the posterior end of an embryo at the 1- to 2-somite stage. Note that the signal is now restricted to the tailbud and the cells immediately adjacent to it (open arrow). (F) At 16-18 hours, the wnt8 signal is obvious at the tip of the tail. (G) Dorsal view of a whole-mount embryo at approximately 80% epiboly, illustrating the expression pattern of wnt8 and gsc. The expression of gsc, in cells of the anterior embryonic shield (es), delineates the wnt8- negative area corresponding to the axial hypoblast (open arrows). (H) Higher magnification view from the animal pole clearly demonstrates that by late gastrulation, wnt8 is no longer expressed in cells in the dorsal midline (triangles). (I) Dorsal view with anterior to the top of a whole-mount embryo stained for pax2 expression. Note that the pax2 signals in the lateral and caudal margins of the shield (arrows), are similar to those in a comparable stage embryo stained for wnt8 (Fig. 3D). (J) The expression pattern of snail1 in a whole-mount embryo at a stage comparable to the one in Fig. 2E. Although wnt8 (Fig. 3E) and snail1 (Fig. 3J) are expressed in the tailbud, only snail1 is expressed in the more anterior regions flanking the midline axis. Abbreviations: a, axial mesoderm; n, notochord; tb, tailbud. Scale bar (A-E, G-J), 55 μm; (F), 45 μm. EXPRESSION / LABELING:
|
|
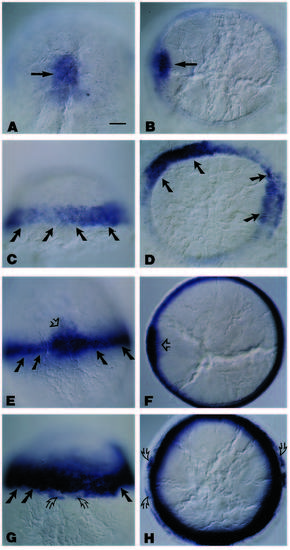
The spatial localization of wnt8b transcripts during normal zebrafish embryogenesis. All embryos are oriented anterior to the top and dorsal facing up, except C, which is a lateral view of the embryo in D. (A) At approximately 90% epiboly, wnt8b signals appear in two wing-like patterns flanking the dorsal midline. (B) At the 2- to 3- somite stage, the wnt8b signal has joined over the dorsal midline (large arrow), and is continuous with signals at the lateral edge of the body axis (small arrows). (C) Lateral view of an embryo at approximately 16 hours, illustrating the wnt8b signals at the future midbrain-hindbrain boundary, the epiphysis, the ventral floor of the forebrain (arrow from asterisk), and in rhombomeres (r) r1, 3 and 5. (D) Dorsal view of the whole mount in C, demonstrates that wnt8b expression is contiguous over the entire width of the rhombomere. wnt8b expression in the forebrain is not obvious in C due to the curvature of the embryo, but close examination in these more rostral regions (E) reveals that wnt8b is expressed at the forebrain-midbrain border and in a discrete layer of the eye (arrows). (F) At approximately 19 hours, wnt8b signals are also detected in a limited region of the optic stalk and in the ventral forebrain (arrow from asterisk). (G) Whole-mount embryo having been hybridized to both the wnt8b and krox20 antisense DIG-labeled RNA probes. The wnt8b signals are confined to the prospective rostral neuroepithelium, in the area corresponding to the future midbrain-hindbrain boundary, and the two thin stripes denoted by the open arrows represent the krox20 expression in prospective r3. (H) To compare directly the relationship between the expression of wnt8b and krox20 in specific rhombomeres, embryos were hybridized simultaneously to wnt8b and krox20 antisense probes. Only the characteristically weak wnt8b signal in r1 can be detected in the double in situ; signals in r3 and r5 are not resolved due to the robust krox20 signal. (I) Dorsal view of a whole-mount embryo at a stage comparable to the embryo in F, illustrating the prominent pax2 expression in the optic stalk and at the midbrain-hindbrain boundary. Abbreviations: e, epiphysis; fmb, forebrain-midbrain boundary; mhb, midbrain-hindbrain boundary; os, optic stalk; r, rhombomeres. Scale bar (A,B), 55 μm; (C-F, H-I), 45 mm; (G), 65 μm. EXPRESSION / LABELING:
|
|
Expression pattern of wnt8 (A) and snail1 (B) in the tailbud of spadetail mutant embryos, and wnt8b in the rostral CNS of wild-type (C) and cyclopsb16 mutant (D) embryos. Anterior is to the left and dorsal is to the top in all photomicrographs. (A) For wnt8, the increase in the relative intensity of the signal compared with that in a wild-type embryo (Fig. 3F), can be attributed to the snail1-positive cells (Fig. 5B) that failed to converge properly during gastrulation. (C) In wild-type embryos at approximately 24 hours postfertilization, wnt8b is expressed in the epiphysis (top arrow), the optic stalk (lower arrow), the ventral floor of the forebrain, just caudal to the optic stalk signal, and at the midbrain and hindbrain junction. (D) In homozygous cyc-/- mutants, wnt8b expression is unaffected in the epiphysis (top arrow) and at the boundary behind midbrain and hindbrain. Note the loss of the signal in the optic stalk (arrow from asterisk) and in the ventral floor of the forebrain just caudal to it. Abbreviations: h, hindbrain; m, midbrain; n, notochord. Scale bar (A,B), 45 μm; (C,D), 55 μm. EXPRESSION / LABELING:
|
|
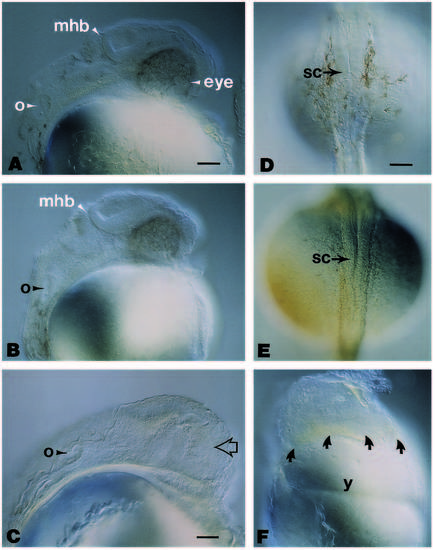
Morphological comparisons between uninjected, β-galactosidase-RNA-injected and wnt8-RNA-injected zebrafish embryos at approximately 30 hours postfertilization. Embryos in (A-C) are oriented anterior to the top and dorsal to the left. (A) The prominent eye, otocyst and midbrain-hindbrain boundary are evident in this lateral view of an uninjected embryo. (B) Note the apparent normal morphology of these structures in a lateral view of an embryo having been injected with approximately 1 ng of b-galactosidase RNA. (C) In embryos injected with approximately 0.1 ng of wnt8 RNA, the region where the eye should have formed (arrow) and the regions flanking the midbrain-hindbrain boundary are noticeably affected, but the otocyst appears to have developed normally. (D) Dorsal view of an uninjected embryo at approximately 30 hours, illustrating the spinal cord oriented in an anterior (top) to posterior (bottom) direction. (E) Dorsal view of an embryo having been injected with approximately 0.2 ng of wnt8 RNA, illustrating that a single axis forms even though there were defects apparent in the mid- and forebrain similar to those seen in C, but out of the plane of focus. The spinal cord in these wnt8-injected embryos is morphologically similar to that seen in the uninjected control (D). (F) Lateral view of an embryo having been injected with approximately 0.2 ng of wnt8 RNA and examined at 18 hours of development. The majority of this embryo has developed as an amorphous mass above the yolk (arrows). Despite the few anatomically visible landmarks, and although it is out of the plane of focus, there is a morphologically recognizable notochord exhibiting the characteristic vacuolated arrangement (see Westerfield, 1989). Abbreviations: mhb, midbrain-hindbrain boundary; o, otocyst; sc, spinal cord; y, yolk. Scale bar (A, B), 55 μm; (C), 40 μm; (D-F), 65 μm. |
|
The spatial localization of gsc and ntl transcripts in b-galactosidase-RNA-injected and wnt8-RNA-injected embryos at 55-60% epiboly. Embryos in A, C, E and G are oriented animal pole to the top, whereas embryos in B, D, F and H are viewed from the animal pole. (A,B) gsc expression in embryos injected with 1 ng of b-galactosidase RNA. gsc expression is restricted to the shield (arrow) in these embryos. (C,D) gsc expression in embryos injected with approximately 0.5 ng of wnt8 RNA. Note that the gsc signal has expanded in thickness at the germ ring (arrows) and there is no readily identifiable shield. The increased gsc signal in the germ ring can occur in a uniform manner around the entire circumference as in C, or as seen in a view from the animal pole of another early gastrula embryo (D), it may appear intermittently in a random fashion (arrows). (E,F) The spatial pattern of ntl expression in embryos injected with approximately 1 ng of b- galactosidase RNA. The ntl signals are detected in the germ ring (arrows) and in cells that have involuted in the embryonic shield (open arrow). (G,H) The spatial pattern of ntl expression in embryos injected with approximately 0.5 ng of wnt8 RNA, illustrating the increase in the width of the ntl expression in the germ ring (arrows). In addition, the ntl signal in cells ventral to the germ ring in the enveloping layer (open arrows), has increased over those cells in b- galactosidase injected (F) or uninjected controls (data not shown). This apparent increase in the signal is not restricted to any one region since there are numerous cells around the entire germ ring expressing ntl (open arrows, H). Scale bar (A-H), 65 μm. |
|
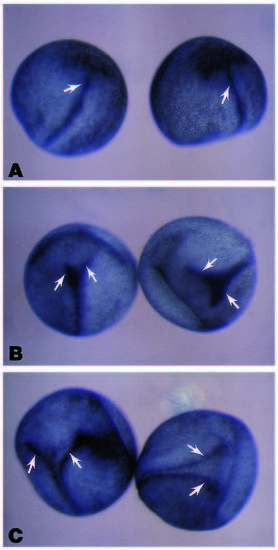
Ectopic expression of prolactin, zebrafish wnt8 and wnt8b in Xenopus embryos. (A) Embryos injected with prolactin RNA appear normal with a single axis (arrow), whereas those injected with either wnt8 (B) or wnt8b (C) exhibit two axes (arrows). |

Unillustrated author statements EXPRESSION / LABELING:
|