- Title
-
Partitioning of gene expression among zebrafish photoreceptor subtypes
- Authors
- Ogawa, Y., Corbo, J.C.
- Source
- Full text @ Sci. Rep.
|
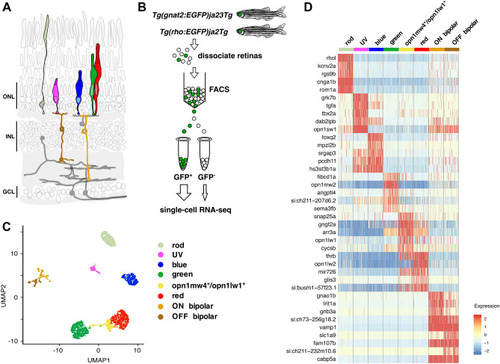
Transcriptome profiles of adult zebrafish photoreceptor subtypes. ( |
|
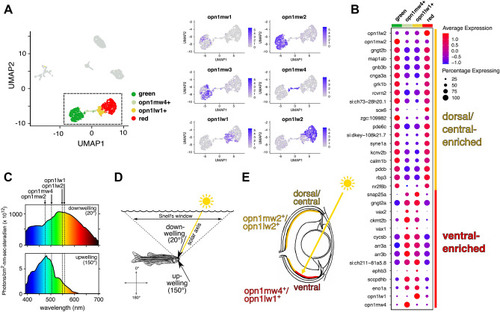
A distinctive subpopulation of red and green cones in the ventral retina. ( |
|
Expression partitioning of paralogous phototransduction genes. ( |
|
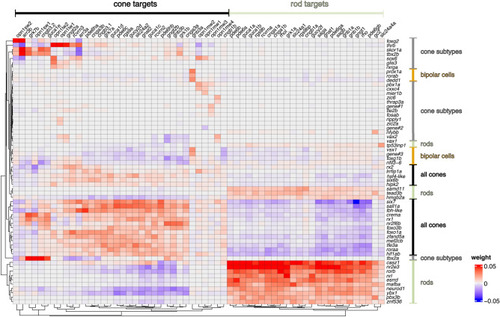
Candidate transcriptional regulators responsible for expression of phototransduction genes. Heatmap showing positive (red) and negative (blue) associations between transcriptional regulators (transcription factors and cofactors) and differentially expressed phototransduction genes (target genes) calculated by GENIE3 algorithm in SCENIC. Rows and columns are arranged according to divisive hierarchical clustering (dividing clusters in a top-down manner). The (dis)similarity of observations was calculated using Euclidean distances. Cell type expression patterns of the transcriptional regulators are presented in Fig. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. EXPRESSION / LABELING:
|