- Title
-
Sodium pump activity in the yolk syncytial layer regulates zebrafish heart tube morphogenesis
- Authors
- Langenbacher, A.D., Huang, J., Chen, Y., and Chen, J.N.
- Source
- Full text @ Dev. Biol.
|
Sodium pump function is important for cardiomyocyte migration and heart tube elongation. (A–D) Time-lapse images showing a dorsal view (anterior to top) of heart morphogenesis in a wild type Tg(myl7:EGFP) embryo. After arriving at the midline, wild type cardiomyocytes rapidly migrate anteriorly and leftward between 18.5 and 26 hpf, generating the heart tube. (E–H) Time-lapse images showing a dorsal view (anterior to top) of heart morphogenesis in a atp1a1m883 mutant Tg(myl7:EGFP) embryo. Cardiomyocytes migrate to the midline normally in the majority of atp1a1m883 mutants (E), but only exhibit limited migration from 18.5 to 26 hpf. At 26 hpf, atp1a1m883 mutants have a clump of cardiomyocytes at the midline (H). PHENOTYPE:
|
|
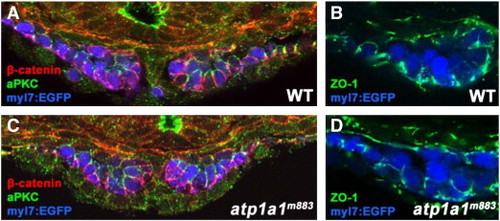
Cell polarity and tight junctions are not disrupted by loss of Atp1a1 function. (A–D) Transverse vibratome sections of 20S stage wild type and atp1a1m883 Tg(myl7:EGFP) embryos. (A,C) The polarized localizations of β-catenin (red, basolateral) and aPKC (green, apicolateral) are indistinguishable between wild type (A) and atp1a1m883 mutant (C) embryos at the 20S stage. (B,D) Tight junctions are present basolaterally in both wild type (B) and atp1a1m883 mutant (D) embryos based on the localization of the tight junction protein ZO-1 (green) at the 20S stage. PHENOTYPE:
|
|
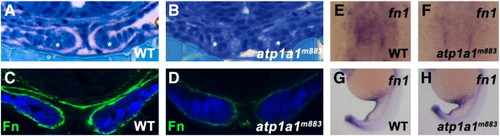
Sodium pump activity is required for extracellular matrix deposition around the cardiac cone. (A,B) Transverse microtome sections of 20S stage wild type and atp1a1m883 embryos stained with methylene blue. In wild type embryos, sulfated proteoglycans are stained pink by methylene blue, allowing visualization of the abundant extracellular matrix present around the cardiomyocytes of the cardiac cone (*) at the 20S stage (A). In atp1a1m883 mutants, the cardiac cone forms normally (*), but thickened extracellular matrix is completely absent (B). (C,D) Transverse vibratome sections of 20S stage wild type and atp1a1m883 Tg(myl7:EGFP) embryos. Wild type embryos (C) have Fibronectin protein (green) surrounding the cardiomyocytes of the cardiac cone (blue), while atp1a1m883 mutants (D) have extremely reduced Fibronectin protein deposition. (E,F) Dorsal views of fn1 expression in 20S stage wild type and atp1a1m883 embryos. Fn1 is expressed in the cardiac region of wild type embryos (E), but is dramatically reduced in atp1a1m883 mutants (F). (G,H) Lateral views of fn1 expression in 20S stage wild type and atp1a1m883 embryos. Both wild type (G) and atp1a1m883 mutant (H) embryos exhibit fn1 expression in posterior structures including the yolk extension and tail. EXPRESSION / LABELING:
PHENOTYPE:
|
|
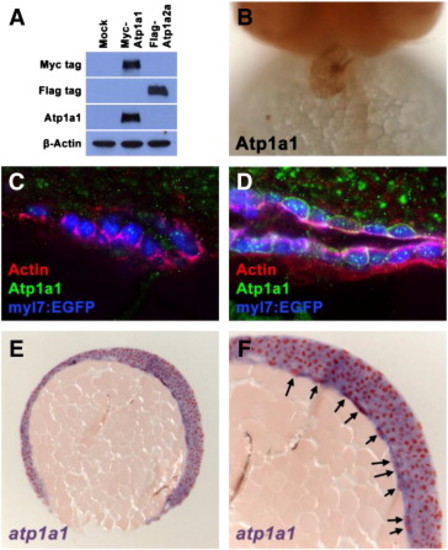
Endogenous Atp1a1 protein is not detected in the heart at stages when morphogenesis defects are first observed in atp1a1 mutants. (A) Western blot visualizing Myc-tagged, Flag-tagged, Atp1a1, and β-Actin proteins in HEK293T cell lysates. Mock transfected cell lysates have no reactivity with Myc, Flag, or Atp1a1 antibodies. Lysates from cells transfected with a Myc-tagged zebrafish Atp1a1 construct contain a protein that reacts with both Myc and Atpa1a antibodies. Lysates from cells transfected with a Flag-tagged zebrafish Atp1a2a construct contain a protein that reacts only with the Flag antibody. The presence of β-Actin protein in mock, Myc-Atp1a1, and Flag-Atp1a2a lysates demonstrates equal loading of total protein. (B) Ventral view of Atp1a1 antibody staining on 48 hpf wild type embryo. Atp1a1 protein is strongly detected in the heart. (C,D) Transverse vibratome sections of 20S stage (C) and 24 hpf (D) wild type embryos. At the 20S stage (C), Actin (red) is primarily basolateral around the cardiomyocytes of the cardiac cone (blue), whereas Atp1a1 protein (green) is not detectable in the cardiomyocyte membranes. By 24 hpf (D), polymerized Actin (red) is present on the ventral and lumenal surfaces of the primitive heart tube. Atp1a1 protein (green) is strongly localized to the membrane of cardiomyocytes in a punctate manner by this stage. (E,F) Microtome section of atp1a1 expression in 90% epiboly wild type embryo. (E) At 90% epiboly, atp1a1 is expressed ubiquitously. (F) Magnified portion of image in (E) shows that yolk syncytial layer nuclei strongly express atp1a1 (arrows). EXPRESSION / LABELING:
|
|
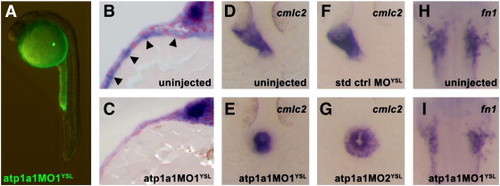
Atp1a1 is required in the yolk syncytial layer for heart tube elongation. (A) Embryos injected at the 512 to 1000-cell stage with fluorescein-tagged atp1a1MO1 exhibit a yolk-specific fluorescence pattern at 24 hpf, indicating that the morpholino′s effects are restricted to the yolk syncytial layer. (B,C) Microtome sections of atp1a1 expression in 12S stage wild type and atp1a1MO1YSL embryos. Strong expression of atp1a1 is visible in the YSL of wild type embryos (arrowheads, B), whereas atp1a1MO1YSL embryos display a specific loss of YSL expression of atp1a1 while maintaining normal embryonic expression (C). (D–G) Dorsal views of cmlc2 expression in 24 hpf uninjected and YSL-morphant embryos. Uninjected and standard control oligo YSL-injected (std ctrl MOYSL) embryos have an elongated heart tube at 24 hpf visualized by cmlc2 expression (D,F) whereas most atp1a1MO1YSL and atp1a1MO2YSL embryos have a clump of cardiomyocytes at the midline at 24 hpf (E,G). (H,I) Dorsal views of fn1 expression in the cardiac region of 15S stage wild type and YSL-morphant embryos. Expression of fn1 is reduced in atp1a1MO1YSL embryos (I) compared to wild type embryos (H). EXPRESSION / LABELING:
PHENOTYPE:
|
|
Atp1a1 expression by the YSL is regulated by mxtx1. (A–D) Microtome sections of atp1a1 expression in 95% epiboly (A,B) and 19S stage (C,D) wild type and mxtx1MOYSL embryos. Nuclei are labeled red by neutral red staining. Expression of atp1a1 (purple staining, arrowheads) is present in the YSL of wild type embryos at 95% epiboly (A) and the 19S stage (C). The expression of atp1a1 in the YSL is strongly reduced in mxtx1MOYSL embryos at the same stages (B,D). (G) Summary of Atp1a1 function. Atp1a1 expression in the YSL is regulated by mxtx1, and is required for the deposition of Fn/ECM around the developing heart. This ECM deposition is critical for normal heart tube elongation. Atp1a1 also has a cell autonomous role within cardiomyocytes to regulate the contractility of the heart. EXPRESSION / LABELING:
|
|
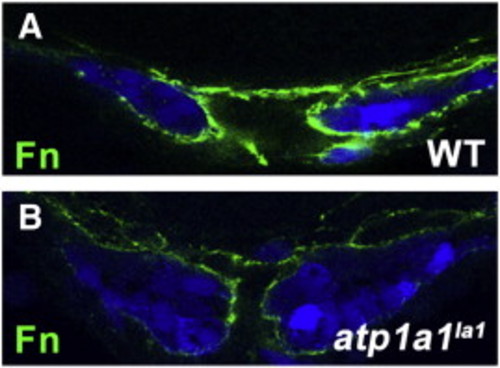
Reduction of Fibronectin protein deposition around atp1a1la1 mutant myocardium. (A,B) Transverse vibratome sections of 20S stage wild type and atp1a1la1 Tg(myl7:EGFP) embryos. Wild type embryos (A) have Fibronectin protein (green) surrounding the cardiomyocytes of the cardiac cone (blue), while atp1a1la1 mutants (B) have reduced Fibronectin protein deposition. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Localization of Atp1a1 protein. (A) Lateral view of Atp1a1 protein in 2 days post fertilization embryo. Atp1a1 protein is detected in the brain (b), heart (h), and pronephros (p). (B) A magnified image of the pronephros expression (p) detected by the anti-Atp1a1 antibody. EXPRESSION / LABELING:
|

Unillustrated author statements PHENOTYPE:
|
Reprinted from Developmental Biology, 362(2), Langenbacher, A.D., Huang, J., Chen, Y., and Chen, J.N., Sodium pump activity in the yolk syncytial layer regulates zebrafish heart tube morphogenesis, 263-270, Copyright (2012) with permission from Elsevier. Full text @ Dev. Biol.