- Title
-
Requirement for zebrafish ataxin-7 in differentiation of photoreceptors and cerebellar neurons
- Authors
- Yanicostas, C., Barbieri, E., Hibi, M., Brice, A., Stevanin, G., and Soussi-Yanicostas, N.
- Source
- Full text @ PLoS One
|
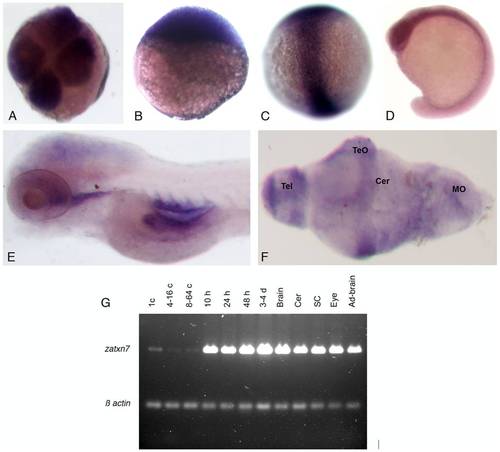
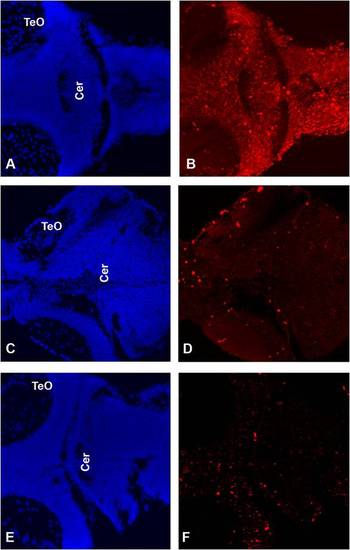
Transcription of the zebrafish atxn7 gene during development. (A-F) In situ detection of zebrafish atxn7 transcripts on either whole mount embryos at the four-cell stage (A), or 3 (B), 8 (C), 16 (D), and 48 hpf (E) or dissected brain of 120 hpf embryos (F). (I) RT-PCR analysis of zebrafish atxn7 transcript accumulation in 1- (1 c), 4- to 16- (4–16 c), and 8- to 64-cell embryos (8–64 c), or 10 (10 h), 24 (24 h), 48 (48 h) and 72 to 96 hpf embryos (3–4 d) and dissected adult brain (Brain), cerebellum (Cer), spinal cord (SC), eye (Eye) and remaining tissues (Ad-brain). RT–PCR for β-actin is shown as a positive control. Abbreviations: Cer, cerebellum; MO, medulla oblongata; TeO, tectum opticum; Tel, telencephalon. |
|
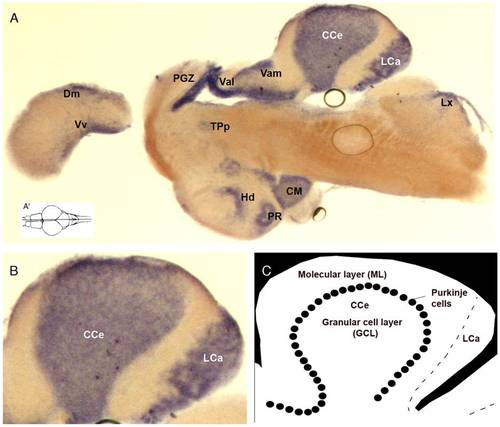
Transcription of the zebrafish atxn7 gene in adult brain sections. In situ detection of zebrafish atxn7 transcripts on midsagittal section of adult brain (A). Dorsal view of the adult zebrafish brain showing the position level of the parasagittal section shown in A (A′). A magnified view of the section showed in panel A (B). Schematic representation of zebrafish cerebellum (C). Anterior is to the left. Abbreviations: CC, crista cerebellaris; CCe, corpus cerebelli; CM, corpus mamillare; Dm, medial zone of dorsal telencephalic area; Hd, dorsal zone of the periventricular hypothalamus; LCa, lobus caudalis cerebelli; LX, lobus vagus; MON, medial octavolateralis nucleus; PGZ, periventricular grey zone; PR, posterior recess of diencephalic ventricle; TPp, periventricular nucleus of the posterior tuberculum; Val, lateral division of valvula cerebelli; Vam, medial division of valvula cerebellaris; Vv, ventral nucleus of the ventral telencephalon. EXPRESSION / LABELING:
|
|
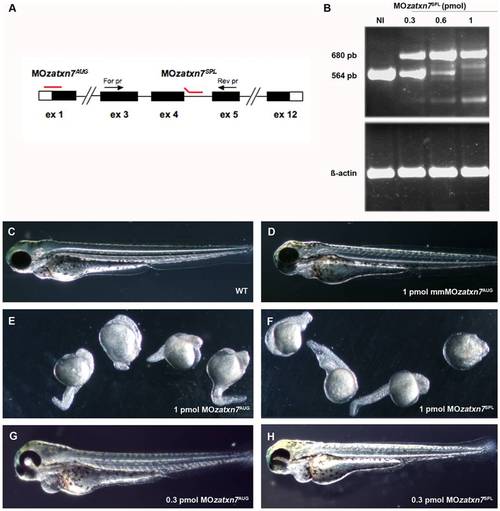
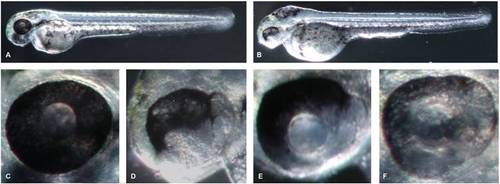
Morpholino-mediated inactivation of zebrafish atxn7 impairs embryonic development. Schematic representation of the zebrafish atxn7 gene showing exons 1 (ex 1), 3 to 5 (ex 3-5) and 12 (ex 12) (black boxes), location of MOzatxn7AUG and MOzatxn7SPL (red lines) and position of oligonucleotides (black arrows) used for RT-PCR analysis of MOzatxn7SPL-mediated inhibition of zebrafish atxn7 intron 4 splicing (black arrows) (A). Untranslated exonic regions and intronic sequences are depicted as empty boxes and single lines, respectively. RT-PCR analysis of zebrafish atxn7 intron 4 splicing in non-injected (NI) and morphant embryos that had received 0.3, 0.6 and 1 pmol MOzatxn7SPL (B). RT-PCR for β-actin is shown as a positive control. Phenotypes of 48 hpf wild-type embryo (C), and age-matched 1 pmol mmMOzatxn7AUG (D), 0.3 pmol MOzatxn7AUG (G) and 0.3 pmol MOzatxn7SPL morphants (H). Phenotypes of 24 hpf 1 pmol MOzatxn7AUG (E) and 1 pmol MOzatxn7SPL morphants (F). PHENOTYPE:
|
|
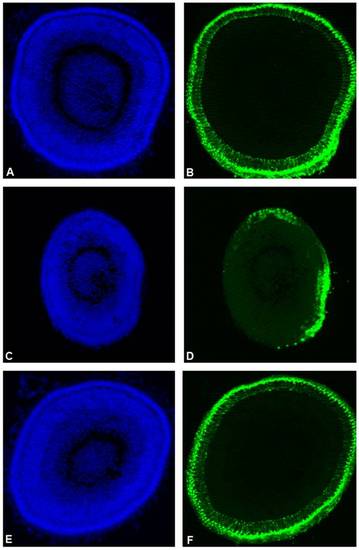
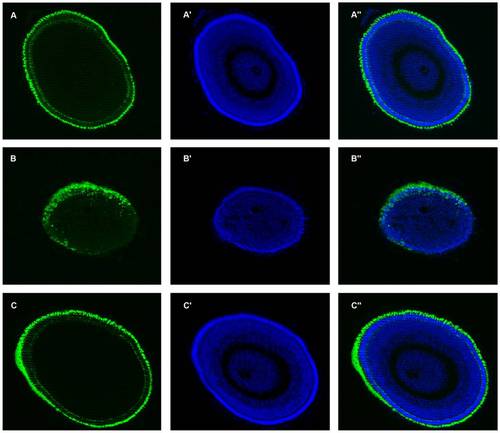
Partial zebrafish atxn7 depletion impairs photoreceptor differentiation. DAPI staining (A, C, and E) and rhodopsin immunostaining (B, D, and F) of eye cryosections of 48 hpf wild type embryo (A and B) and age-matched 0.3 pmol MOzatxn7SPL (C and D) and 1 pmol mmMOzatxn7AUG morphant embryos (E and F). EXPRESSION / LABELING:
PHENOTYPE:
|
|
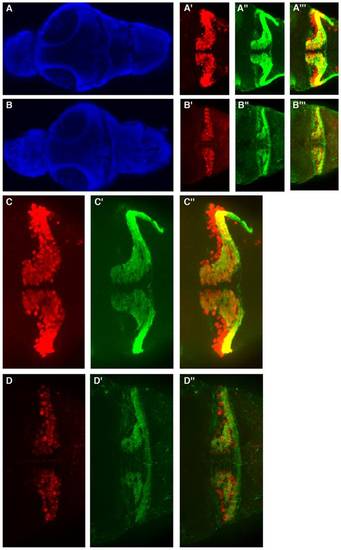
Moderate zebrafish atxn7 depletion impairs the differentiation of cerebellar neurons. Dorsal views of dissected brains from 5 dpf 1 pmol mmMOzatxnAUG (A, A′, A′′, A′′′, C, C′ and C′′) and 0.3 pmol MOzatxn7SPL morphants (B, B′, B′′, B′′′, D, D′ and D′′). DAPI staining (A and B), Pav7 immunostaining of Purkinje cells (A′, B′, C and D) and Vglut1 immunostaining of granule cells (A′′, B′′, C′ and D′). Anterior is to the left. Enlarged views of the brains showed in A′ (C), A′′ (C′), A′′′ (C′′), B′ (D), B′′ (D′), and B′′′ (D′′). Merge images of the photographs A′ and A′′ (A′′′), B′ and B′′ (B′′′), C and C′ (C′′), and D and D′ (D′′). |
|
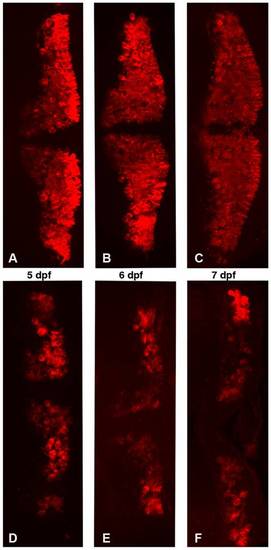
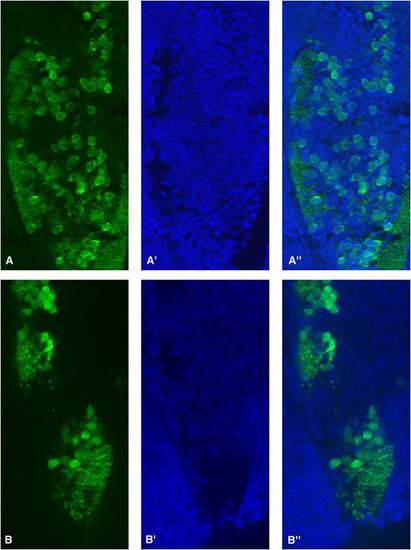
Partial zebrafish atxn7 depletion impairs the differentiation of cerebellar neurons. Dorsal views of dissected brains from 5 (A and D), 6 (B and E), and 7 dpf (C and F) 1 pmol mmMOzatxn7AUG (A, B, and C) and 0.3 pmol MOzatxn7SPL morphants (D, E, and F) immunostained with an anti-zebrin II antibody, which specifically labels Purkinje cells. |
|
[1–232] N-terminal fragment of human atxn7 can rescue photoreceptor differentiation defect in 0.3 pmol MOzatxn7SPL morphant Rhodopsin immunostaining (A, B, and C) and DAPI staining (A′, B′, and C′) of eye cryosections of 48 hpf 1 pmol mmMOzatxn7AUG (A, A′ and A′′) and 0.3 pmol MOzatxn7SPL morphants (B, B′ and B′′) and age matched 0.3 pmol MOzatxn7SPL morphant co-injected with 2 fmol human atxn7 mRNA (C, C′ and C′′). Merge images of the photographs A and A′ (A′′), B and B′ (B′′), and C and C′ (C′′). |
|
[1–232] human atxn7 fragment can rescue differentiation defects of cerebellar neurons in 0.3 pmol MOzatxn7SPL morphant. Dorsal views of dissected brains from 5 dpf 1 pmol mmMOzatxnAUG (A, A′ and A′′) and 0.3 pmol MOzatxn7SPL morphants (B, B′ and B′′) and age-matched 0.3 pmol MOzatxn7SPL morphant co-injected with human atxn7 mRNA (2 fmol) (C, C′ and C′′). Pav7 immunostaining of Purkinje cells (A, B and C) and Vglut1 immunostaining of granule cells (A′, B′ and C′). Anterior is to the left. Merge images of the photographs A and A′ (A′′), B and B′ (B′′), and C and C′ (C′′). |
|
Identification and sequence of the zebrafish atxn7 gene. Molecular phylogeny of the human (hatxn7 or ENSG0000 hatxn7L2 or ENSG00000162650; hatxn7L3 or ENSG00000087152; and hatxn7L3B or ENSG00000253719) and zebra zatxn7l2a or ENSDARG00000055300; zatxn7l2b or ENSDARG00000056268; and zatxn7l3 or ENSDARG0000002933 transcription of the zatxn7l2a, zatxn7l2b and zatxn7l3 genes in zebrafish embryos aged 24, 48 and 72 hpf (B). Seque ENSG00000163635), mouse (M.m., ENSMUSG00000021738), and zebrafish (D.r., ENSDARG00000074804) atxn7 p obtained from the Ensembl data base (http://www.ensembl.org). Molecular phylogeny was determined using ClustalW Peptidic sequences were aligned using Align (http://www.ebi.ac.uk/Tools/msa/clustalw2/). Colour code for amino acids acids, blue. Abbreviations: Homo sapiens, H.s.; Mus musculus, M.m.; Danio rerio, D.r. EXPRESSION / LABELING:
|
|
Mild zebrafish atxn7 depletion impairs retina differentiation. 48 hpf 1 pmol mmMOzatxn7AUG (A) and 0.3 pmol MOzatxn7AUG morphant embryos (B). Eye of a 48 hpf 1 pmol mmMOzatxn7AUG morphant (C) and partially depigmented retinas of 48 hpf 0.3 pmol MOzatxn7AUG (D) and 0.3 pmol MOzatxn7SPL (E and F) morphant embryos. |
|
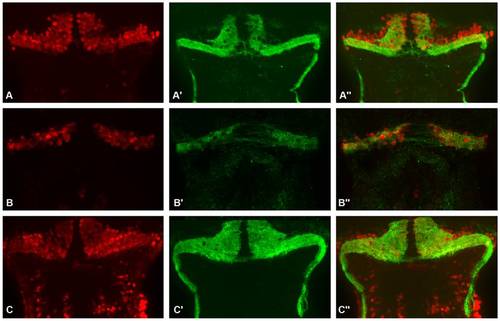
Partial zebrafish atxn7 depletion impairs Purkinje cell differentiation. Frontal sections of dissected brains of 5 dpf 1 pmol mmMOzatxn7AUG (A-A′′) and 0.3 pmol MOzatxn7SPL morphant embryos (B-B′′). RORα immunostaining of Purkinje cells (A and B) and DAPI staining (A′ and B′). Merge images of the photographs A and A′ (A′′) and B and B′ (B′′). PHENOTYPE:
|
|
|
|
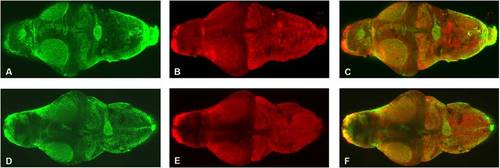
Mild zebrafish atxn7 depletion does not impair overall brain organization. Dorsal view of dissected brains from 5 dpf 1 pmol mmMOzatxn7AUG (A-C) and 0.3 pmol MOzatxn7SPL morphant embryos (D-F). Anterior is to the left. GFAP immunostaining of glial cells (A and D) and HuC immunostaining of neuronal cells (B and E). Merge images of the photographs A and B (C) and D and E (F). |
|
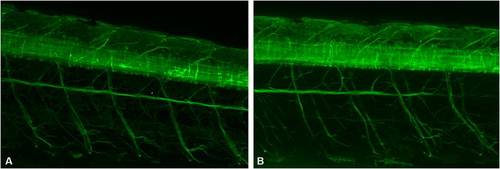
Partial zebrafish atxn7 depletion does not impair spinal cord differentiation. Lateral views of 48 hpf Tg[NBT:MAPT-GFP]zc1 transgenic embryos following injection of 1 pmol mmMOzatxn7AUG (A) and 0.3 pmol MOzatxn7SPL (B). Anterior is to the left. |
|
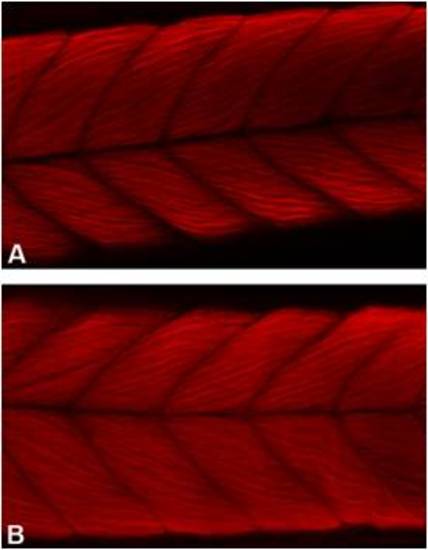
Moderate zebrafish atxn7 depletion does not impair the differentiation of trunk muscles. Lateral views of 48 hpf 1 pmol mmMOzatxn7AUG (A) and 0.3 pmol MOzatxn7SPL morphant embryos (B) following rhodamine-coupled phalloidin labelling of muscle F-actin. Anterior is to the left. |