- Title
-
Transcript Analysis of Zebrafish GLUT3 Genes, slc2a3a and slc2a3b, Define Overlapping as Well as Distinct Expression Domains in the Zebrafish (Danio rerio) Central Nervous System
- Authors
- Lechermeier, C.G., Zimmer, F., Lüffe, T.M., Lesch, K.P., Romanos, M., Lillesaar, C., Drepper, C.
- Source
- Full text @ Front. Mol. Neurosci.

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |
|
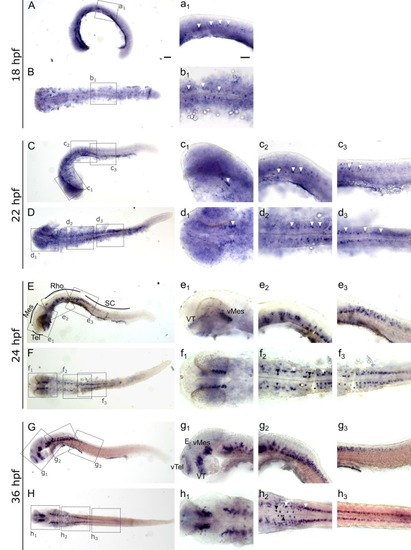
Whole mount RNA EXPRESSION / LABELING:
|
|
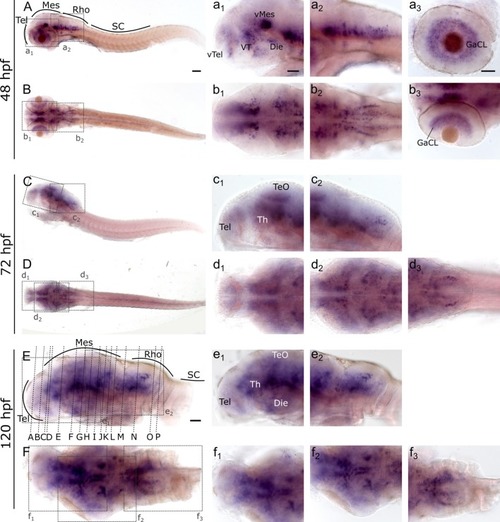
Whole mount RNA |
|
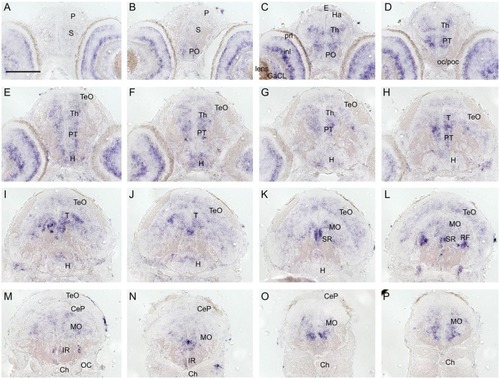
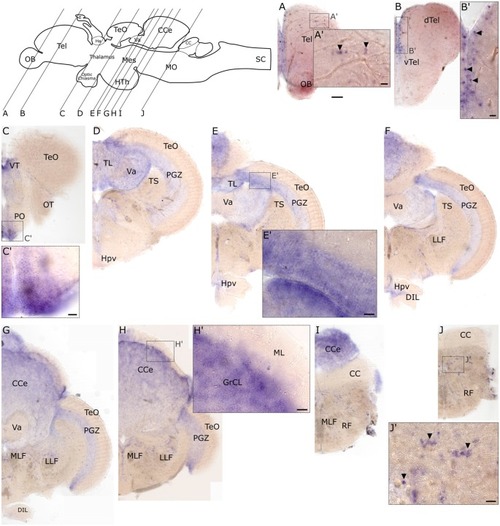
Cryosections (20 μm) of 120 hpf embryo processed for RNA EXPRESSION / LABELING:
|
|
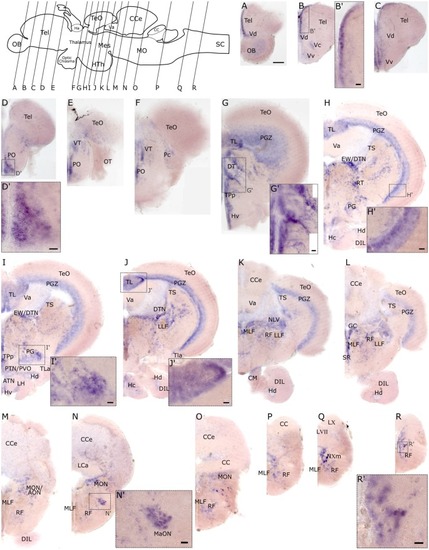
Whole mount RNA EXPRESSION / LABELING:
|
|
RNA EXPRESSION / LABELING:
|
|
RNA |