- Title
-
Spatial Regionalization and Heterochrony in the Formation of Adult Pallial Neural Stem Cells
- Authors
- Dirian, L., Galant, S., Coolen, M., Chen, W., Bedu, S., Houart, C., Bally-Cuif, L., Foucher, I.
- Source
- Full text @ Dev. Cell
|
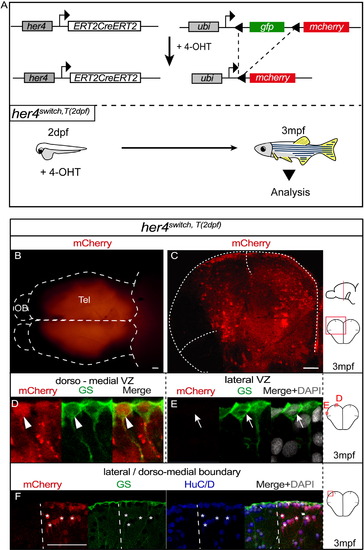
her4-Expressing Progenitors at 2 dpf Generate Adult NSCs of the Dorsomedial Pallium (A) Genetic strategy used for the time-controlled fate mapping of her4-expressing progenitors: 4-hydroxy-tamoxifen (4-OHT) triggers ERT2CreERT2 activation in her4+ progenitors allowing GFP excision and permanent mcherry expression (top). Experimental design to map the adult fate of early her4+ progenitors: 4-OHT is applied at 2 dpf (her4switch,T(2 dpf)) and recombined animals are analyzed at 3mpf (bottom). (B) Dorsal view (whole-mount, anterior left) of a her4switchT(2 dpf) adult telencephalon showing regionalized mCherry expression. Dotted lines delineate the telencephalon (Tel) and the olfactory bulb (OB). (C) Cross-section of the telencephalon in a her4switch,T(2 dpf) adult, focusing on the pallium and stained as indicated. Dotted lines delineate pallial boundaries with the medial and lateral pallial sulci; one hemisphere is shown. (D and E) High magnification of the dorsomedial (D) or lateral (E) NSCs. mCherry is expressed only in dorsomedial RGCs expressing GS (D, arrowheads) and not in lateral RGCs (E, arrows). (F) Magnification of the boundary (dotted line) between the dorsomedial and lateral pallial domains, showing the segregation of mCherry+ and mCherry NSCs and neurons. Asterisks indicate some mCherry+ neurons. See also Figure S1. |
|
A Restricted Number of Progenitors at 2 dpf Generate the Lateral aNSCs Following Massive Postembryonic Amplification (A) Experimental design to analyze the morphology of aNSCs polyclones generated from embryonic progenitors in the lateral pallium. (B) Dorsal (top) and lateral views (bottom) of ubiswitch,T(2 dpf) telencephali (whole mount of one hemisphere, anterior left). Representative lateral clone types 1, 2, and 3 are shown (dotted lines). Asterisks highlight medial pallial clones. (C) Cross-sections of the telencephali shown in (B; section plane: yellow) and stained as indicated. (D) Experimental design to map the adult fate of individual early pallial progenitors in single brains. (E) Lateral projections of the adult pallium (whole-mount) in hsp70zebrabow,HS(2 dpf) fish showing expression of CFP/YFP (top) or CFP/YFP/dTomato (bottom). (F) Cross-section of the telencephalon of hsp70zebrabow,HS(2 dpf) adults showing expression of CFP/YFP (left) or CFP/YFP/dTomato (right). (G) Scheme depicting the typical morphology of lateral pallial clones after a recombination as in (A) and (D), and cross-sections at anterior, medial, and posterior levels. Triangles represent the RGCs, colored domains represent the neurons generated from these progenitors, and arrows represent the progression of neurogenesis. See also Figure S2. |
|
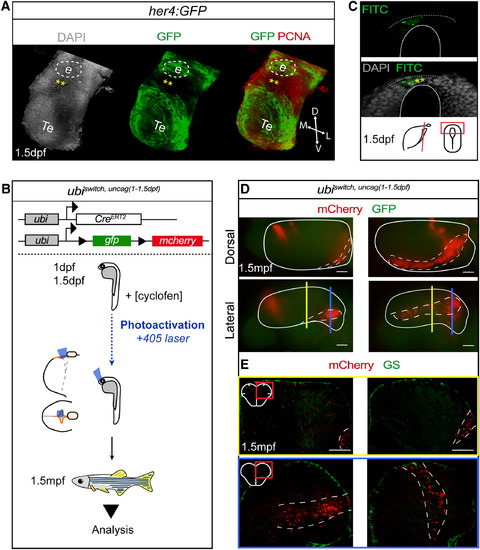
Lateral aNSCs-Fated Embryonic Progenitors Are Located in the Posterior Telencephalic Roof Plate (A) Three-dimensional view of the prosencephalon at 1.5 dpf in a her4:GFP embryo immunostained as indicated. Dashed line represents the position of the epiphysis (e). Posterior part of the telencephalic roof plate. Te, telencephalon. (B) Experimental design to fate map embryonic progenitors located at the posterior telencephalic roof plate: caged-cyclofen (cyclofen) was locally photoactivated in ubi:creErt2;ubi:switch embryos at 1–1.5 dpf using a 405 nm laser beam (blue box represents the laser-activated area). Recombined animals (ubiswitch,uncag(1-1.5 dpf)) were analyzed at 1.5 mpf. (C) Cross-section focusing on the posterior telencephalic roof plate of an embryo injected with caged-FITC and analyzed immediately after uncaging. The uncaged area (asterisks) is limited to the roof plate. (D) Dorsal (top) and lateral (bottom) whole-mount views of ubiswitch,uncag(1-1.5 dpf) telencephali. Dotted lines surround the pallium. (E) Cross-sections at the medial level (top: yellow section plane in D) and at the posterior level (bottom: blue section plane in D) of ubiswitch,uncag(1-1.5 dpf) lateral telencephali stained as indicated. See also Figure S3. |
|
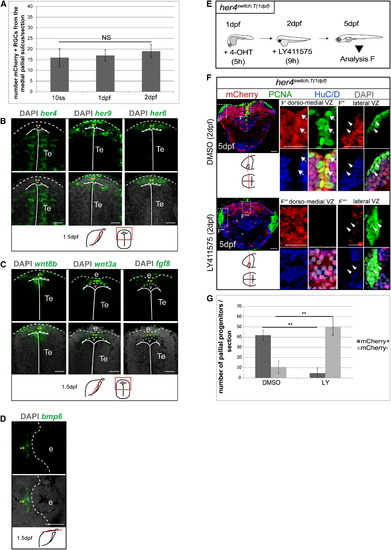
The her4-Negative Cells at the Origin of Lateral Pallial aNSCs Belong to the “Progenitor Pool” Subclass of Embryonic Neural Progenitors (A) Number of mCherry+ RGCs per adult telencephalic section, from the sulcus ypsiloniformis up to the edge of the VZ, after recombination at 1–10 somites, 1 dpf or 2 dpf of her4switch embryos. Values are presented as mean ± 95% CI (ANOVA). (B–D) Compared expression of her4, her9, her6, wnt8b, wnt3a, fgf8, and bmp6 along the posterior telencephalic roof plate at 1.5 dpf, revealed with fluorescence in situ hybridization without/with 42,6-diamidino-2-phenylindole. Frontal (B and C) or horizontal (D) single confocal planes are shown. Dashed line represents roof plate of the neural tube (or epiphysis) and plain lines represent the ventricle. Roof plate. e, epiphysis; Te, telencephalon. (E) Experimental design to assess Notch sensitivity of pallial progenitors at 2 dpf. (F) Medial cross-sections of the telencephalon in her4switchT(1 dpf) larvae treated with DMSO or LY411575. Magnification of the dorsomedial VZ (F′ and F′′′) and lateral VZ (F′′ and F′′′′), immunostained as indicated. Arrows and arrowheads highlight, respectively, the dorsomedial progenitors (mCherry+/PCNA+ cells) and the lateral progenitors (mCherry/PCNA+ cells). (G) Compared number of pallial dorsomedial progenitors (dark gray) and of lateral progenitors (light gray) in control (DMSO) and treated (LY411575) conditions. Values are presented as mean ± 95%CI (ANOVA, p<0.05). See also Figure S4. |
|
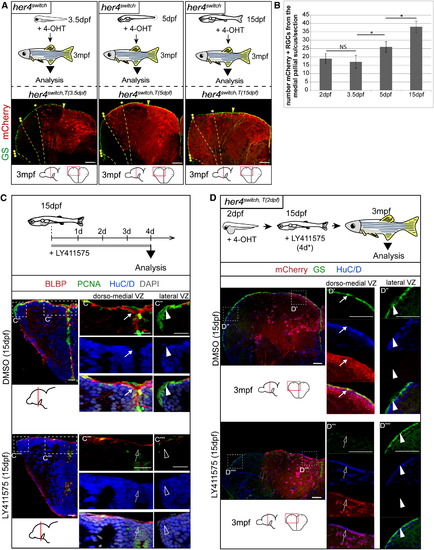
Pallium Development Occurs in Two Heterochronic Waves (A) Experimental design. (B–D) Cross-sections of the telencephalon in her4switch,T(2 dpf) animals at 5 dpf (B), 15 dpf (C), and 1.5 mpf (D) stained as indicated. Arrows and asterisks highlight, respectively, the PCNA+/mCherry- progenitors and the first neurons of the lateral pallium. Arrowheads indicate the lateral pallial sulcus. See also Figure S5. |
|
Lateral Progenitors Progressively Express her4 and Become Notch-Sensitive at Juvenile Stages but Maintain a Cryptic Boundary with the Dorsomedial VZ (A) Adult fate of progenitors expressing her4 at 3.5 dpf (her4switchT(3.5 dpf)), 5 dpf (her4switchT(5 dpf)), or 15 dpf (her4switch,T(15 dpf)): experimental design and respective cross-sections of adult telencephali immunostained as indicated. Yellow stars and dotted lines indicate the mCherry+ boundary observed after recombination at different stages (single star: recombination at 2 dpf; double stars: recombination at 5 dpf; triple stars: recombination at 15 dpf). Yellow arrowhead points to the sulcus ypsiloniformis. (B) Numbers of mCherry+ RGCs after recombination at 2 dpf, 3.5 dpf, 5 dpf, and 15 dpf in her4switch fish counted from the sulcus ypsiloniformis up to the lateral edge of the VZ. mCherry+/GS+ RGCs comprise 33% of the VZ at 3.5 dpf, and 85% after a recombination at 15 dpf. Values are presented as mean ± 95%CI (ANOVA, p < 0.05). (C) Notch sensitivity of pallial progenitors at juvenile stages. Experimental design and cross-sections of the telencephalon in control (top) or LY411575-treated fish (bottom) immunostained as indicated. Magnification of the dorsomedial VZ (C′ and C′′′) and lateral VZ (C′′ and C′′′′), immunostained as indicated. Arrows point to medial progenitors, arrowheads to lateral progenitors, empty arrows/arrowheads indicate when these progenitors are depleted. (D) Adult analysis of the telencephalon of fish treated with a Notch inhibitor at juvenile stage. Experimental design and cross-sections of the telencephalon in control (top) or LY411575-treated fish (bottom) immunostained as indicated. Arrows point to medial (mCherry+) progenitors, arrowheads to lateral (mCherry) progenitors, empty arrows/arrowheads indicate when these progenitors are depleted. See also Figure S6. |
|
Dual Embryonic Origin of Pallial aNSCs and Persistence of Adult NE Cells (A) Posterior cross-sections showing the lateral edge of the adult VZ in her4:GFP fish (left) or WT fish (right) stained as indicated. Arrows point to the her4/PCNA+ NE pool of progenitors. (B) Scheme of the lateral edge of the adult posterior pallial VZ (as shown in A) depicting RGCs (blue triangles), NE cells (blue), her4 expression (yellow), and wnt3a/her9 expression (pink). The her4 NE pool contains wnt3a+ and/or her9+ proliferating progenitors at the junction between the tela-choroïdea and the posterior pallial VZ edge. (C) Summary of pallium formation: the dorsomedial pallial NSCs and neurons (green) derive from embryonic neurogenic progenitors. The lateral pallial NSCs and neurons (blue) derive from few NE “progenitor pool” (dark blue) that are first amplified and become neurogenic only from 5 dpf onward and persist lifelong. Both aNSC populations remain segregated in space. See also Figure S7. |
Reprinted from Developmental Cell, 30(2), Dirian, L., Galant, S., Coolen, M., Chen, W., Bedu, S., Houart, C., Bally-Cuif, L., Foucher, I., Spatial Regionalization and Heterochrony in the Formation of Adult Pallial Neural Stem Cells, 123-36, Copyright (2014) with permission from Elsevier. Full text @ Dev. Cell