- Title
-
Unusual Fluorescent Granulomas and Myonecrosis in Danio Rerio Infected by the Microsporidian Pathogen Pseudoloma Neurophilia
- Authors
- West, K., Miles, R., Kent, M.L., Frazer, J.K.
- Source
- Full text @ Zebrafish
|
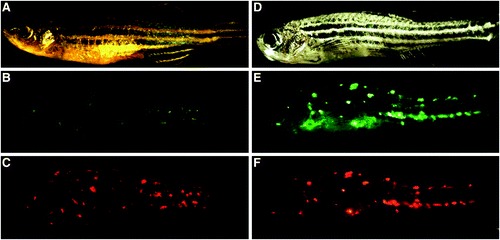
Autofluorescent nodules in Danio rerio. Microscopy demonstrates fluorescent nodules in healthy appearing zebrafish and fish displaying pathologic features. (A) WIK strain fish with brightfield illumination showing normal body contour and pigment and no wasting. (B) Fluorescent microscopy of this fish using green filter settings (emission spectrum 500?550 nm) reveals autofluorescent nodules throughout the animal. (C) Same fish imaged using red filter settings (575?625 nm). Red autofluorescent nodules coincide with locations of green lesions. (D) WIK strain fish shown with brightfield illumination demonstrate abnormal spine curvature, hypopigmentation, and wasting. (E) Identical fish imaged using green filter (500?550 nm) show abundant autofluorescent lesions. (F) Same fish imaged with red filter (575?625 nm) show autofluorescence at identical locations as green fluorescent nodules. |
|
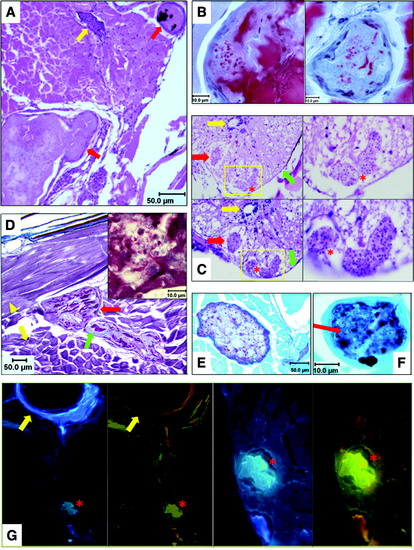
Histopathologic analysis of microsporidia-infected D. rerio. Zebrafish with fluorescent nodules and other signs of infection were analyzed with a panel of histologic stains: (A) H&E stain of fish cross section. Two granulomatous lesions surrounded by inflammatory cells are marked with red arrows. Lesion at upper right contains melanin pigment; yellow arrow denotes a blood vessel. (B) Luna stains of two granulomas. Red spherical objects within lesions are chitin-containing spores stained by Luna. Proteinaceous material stained red by Luna in granulomas is degenerating skeletal muscle. (C) H&E (top) and PAS (bottom) stains of spinal cord demonstrating classic Pseudoloma neurophilia lesions. Low-power images are at left; high-power images of yellow-boxed regions are at right. Spores in lesions appear as punctate basophilic spheres. Red arrows highlight a second lesion outside boxed regions; red asterisks denote two lesions in boxed areas. Yellow and green arrows denote spinal canal and meninges, respectively. (D) Trichrome stain of microsporidial lesions. Skeletal muscle stains pink (yellow arrowhead: longitudinal fibers; yellow arrow: cross-sectioned fibers) and connective tissue is blue. Red arrow identifies two granulomas with dark pink degenerating muscle and blue connective tissue remnants (green arrow). Inset panel displays spores on high power (1000�). (E) PAS stain of a granuloma surrounded by skeletal muscle, counterstained green. (F) GMS stain of a granuloma, highlighting carbohydrates in microsporidial cell walls. Lesion is GMS (+), but spores (red arrow) are GMS (). (G) Fungi-Fluor stain of fish cross section at low (left panels) and high power (right panels). Lesion (red asterisk) is blue with DAPI filter or yellow-green due to Fungi-Fluor staining of chitin in microsporidia with TRITC filter. Yellow arrow denotes a vertebral body adjacent to the peritoneal cavity. DAPI, 4,6-diamidino-2-phenylindole; H&E, hematoxylin and eosin; GMS, Grocott′s methenamine silver; PAS, Periodic acid Schiff; TRITC, tetramethylrhoodamine-5-isothiocyanate. |
|
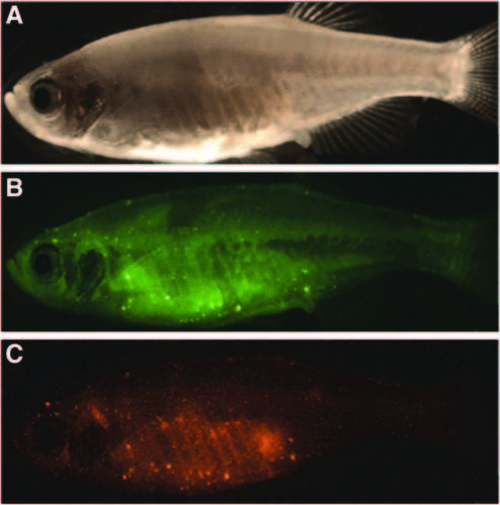
Immunocompetent nacre fish also develop autofluorescent nodules. Nacre fish cohabiting with Danio rerio bearing fluorescent lesions acquire nodules with similar characteristics. (A) Brightfield image of nacre fish exhibiting no apparent signs of infection. (B) Imaging with green filter settings (emission spectrum 500? 550 nm) reveals yellow-green autofluorescent nodules throughout the animal. (C) Imaging using red filter settings (575?625 nm) demonstrates red autofluorescence in the exact locations where green fluorescent nodules were seen. |
|
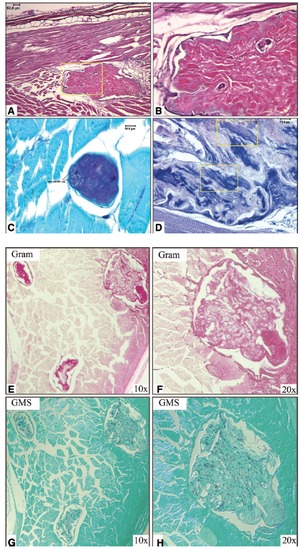
Histopathologic analysis of microsporidia-induced granulomas in D. rerio. Zebrafish with fluorescent nodules were fixed, sectioned, and stained using several methods: (A) Low-power H&E stain of lesion in skeletal muscle. Skin and subcutaneous tissue are seen at the top of the panel. Skeletal muscle runs longitudinally above the lesion and is shown in the cross section surrounding the lesion. Yellowboxed region is shown at higher magnification in panel B. (C) High-power image of a lesion stained by PAS. The lesion itself is PAS + , as are individual spores within the granuloma as denoted by black lines. (D) High-power image of a PTAH-stained lesion showing proteinaceous debris and fibrin deposits within the granuloma. Yellow boxes highlight myonecrosis, with striations of degenerating skeletal muscle still visible. Intact skeletal muscle at the bottom of the image shows striations clearly. (E) The 10X-magnified gram stain of lesions within skeletal muscle. Lesion in upper right corner is shown at 20 � magnification in (F). (G) GMS stain highlighting carbohydrates in the fungal cell wall. Section shown in panel G is from an adjacent section to that shown in panel E. All 3 lesions are GMS+ at 10 � magnification. (H) The 20 � magnification of the GMS+ lesion shown in the upper right corner of panel G. |
|
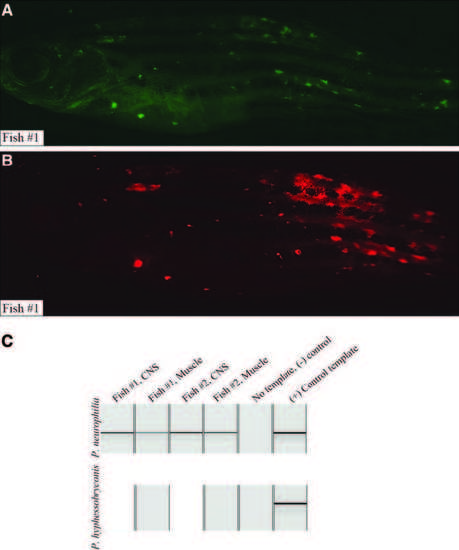
PCR analysis of microsporidia-infected D. rerio. Two fish with fluorescent granulomas were commercially assayed by PCR for microsporidial DNA from Pseudoloma neurophilia and Pleistophora hyphessobryconis (IDEXX RADIL, Columbia, MO). (A) Lck:EGFP transgenic fish with fluorescent nodules, wasting, and abnormal spine curvature imaged using FITC/GFP filter (emission spectrum 500?550 nm). (B) Identical fish imaged using TRITC filter (emission 575?625 nm). Like other animals harboring fluorescent nodules (Fig. 1), lesions coincide using either filter, chiefly in skeletal muscle of flanks. (C) PCR results visualized by capillary electrophoresis: P. neurophilia was detected in CNS and skeletal muscle of both fish (top, lanes 1?4). No template (lane 5, negative control) shows no product; positive control (lane 6) yielded band of identical size. P. hyphessobryconis, a Microsporidian known to infect muscle, was not detected in skeletal muscle of either fish (bottom, lanes 2 and 4). CNS, central nervous system; FITC, fluorescein isothiocyanate; PCR, polymerase chain reaction. |