- Title
-
Metabolic Regulation of Cellular Plasticity in the Pancreas
- Authors
- Ninov, N., Hesselson, D., Gut, P., Zhou, A., Fidelin, K., and Stainier, D.Y.
- Source
- Full text @ Curr. Biol.
|
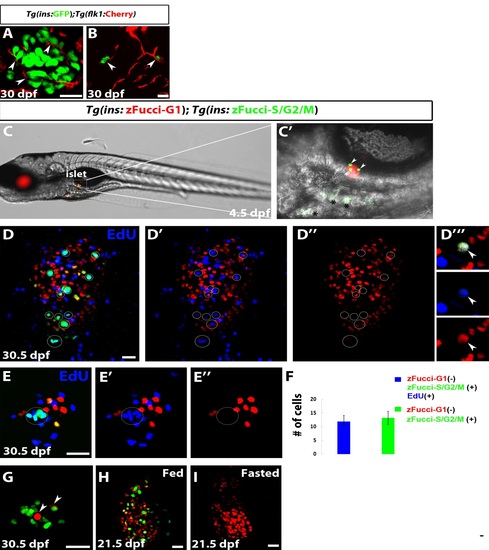
β Cells Transition from Quiescence to Proliferation in Response to Nutrients (A and B) Wild-type (WT) animals imaged at 5 (A) and 21 (B) dpf showing the dramatic growth that takes place in the feeding animal. (C?E) Tg(TP1:H2BmCherry);Tg(ins:GFP) larvae were examined with confocal imaging. Tg(TP1:H2BmCherry) drives the expression of H2BmCherry in NRCs in the IPD, and Tg(ins:GFP) labels β cells. Arrows point to the principal islet (PI). Arrowheads point to secondary islets (SI). (C and C′) The larva was imaged live at 5 (C) and at 7 (C′) dpf. At 7 dpf, two new β cells (arrowheads) had formed posterior to the PI. These cells derived from NRCs, given that they were also Tg(Tp1bglob:H2BmCherry)+. The insets show single planes with separated channels. At 7 dpf, 0.45 (�0.6, n = 21 larvae) β cells containing SIs per larva were present posterior to the PI. (D) At 15 dpf, the number β cells containing SIs per larva was 1.2 (�1.3, n = 22 larvae). (E) At 30 dpf, there was a dramatic increase in the number of β cells, which clustered in multiple SIs (11.8 � 5.7 SIs per animal, n = 6 animals). The pancreas is outlined by a dashed line. (F) Quantification of the PI?s area at 5.5, 16.5 and 27.5 dpf, showing a dramatic increase in its size between 16.5 and 27.5 dpf (n = 6 animals at 5.5 and 16.5 dpf; 5 animals at 27.5 dpf). (G) Quantification of the number of Tg(ins:GFP)+ SIs at 7, 15, and 30 dpf showing a dramatic increase between 15?30 dpf (n = 22 animals at 7 and 15 dpf; 6 animals at 30 dpf). (H) Live imaging of a Tg(ins:zFucci-G1);Tg(ins:zFucci-S/G2/M) larva at 4.5 dpf. A projection of the stack is shown. At t = 0 min, two Tg(ins:zFucci-S/G2/M)+ β cells (arrowheads) were present in the PI. Both cells (1 and 2) divided during the course of the imaging, resulting in the downregulation and the subsequent loss of Tg(ins:zFucci-S/G2/M) expression in their daughter cells (1a and 1b and 2a and 2b, respectively). (I?L) Tg(ins:zFucci-G1);Tg(ins:zFucci-S/G2/M) animals were used to examine the effects of nutrients on β cell proliferation; the images show single confocal planes through the PI. (I?J) Animals from the same clutch were fixed at 1.5 (I) or 10 hr (J) after feeding (AF) starting at 27 dpf. Several β cells (12.2 � 4.3 β cells, n = 9 animals) were proliferating 1.5 hr AF. (J) The number of proliferating β cells increased at 10 hr AF (39 � 23 β cells, n = 11 animals, ***p < 0.005). (K) The animals were fed regularly and examined at 28.5 dpf (12 hr AF). Numerous β cells in the PI were proliferating (21.4 � 4 β cells, n = 7 animals). (L) Animals from the same clutch as in (K) were deprived of food for 28 hr and examined at 28.5 dpf. The number of proliferating β cells in the PI was dramatically reduced (2.5 � 2.2 β cells, n = 14 animals, ****p < 0.0001). (M) Quantification of the average number of proliferating β cells in the PI for the experiments shown in (I)?(L). (A), (B), and (H) are lateral views, anterior to the left and dorsal to the top; (C) and (D) are lateral views, anterior to the top and dorsal to the left; and (E) and (I)?(L) are dorsal views, anterior to the top. Error bars represent SEM, and scale bars represent 20 μm. See also Figure S1. EXPRESSION / LABELING:
PHENOTYPE:
|
|
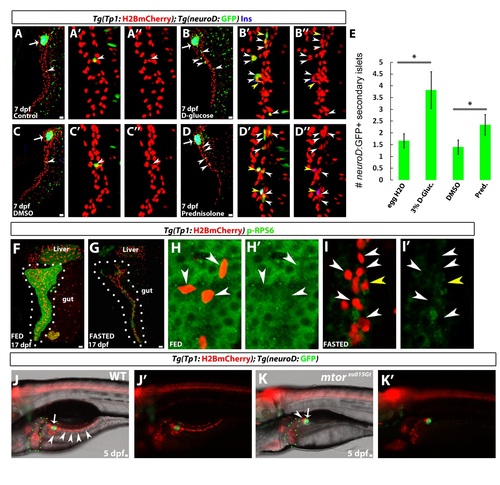
Nutrients Regulate β Cell Differentiation (A) Tg(TP1:CreERT2);Tg(ubi3C:loxp-GFP-loxp:mCherry) larvae were treated with 4-OHT at 14 dpf for 16 hr, fixed at 17 dpf, and stained for 2F11 to mark IPD cells. 4-OHT treatment resulted in the mosaic labeling of individual IPD cells (mCherry+;GFP cells) (arrowheads). (B) Experimental setup for the lineage tracing of β cells from IPD cells. Tg(insulin:loxP:mCherry-STOP:loxP:H2BGFP)+ β cells that originated from IPD cells with Tg(TP1:CreERT2) activity exhibit H2BGFP expression instead of mCherry expression. In a single-progenitor scenario (1), each SI would be composed of H2BGFP+ or mCherry+ cells, whereas, in a multiple-progenitor scenario (2), SIs would be mosaic, containing both H2BGFP+ and mCherry+ cells. (C?F) Tg(TP1:CreERT2);Tg(insulin:loxP:mCherry-STOP:loxP:H2BGFP) larvae treated with limiting concentrations of 4-OHT at 16 dpf for 16 hr and analyzed at 40 dpf. (C) A mosaic SI composed of H2BGFP+ (arrowheads) and mCherry+ β cells. (D) Three individual H2BGFP+ β cells (arrowheads) located in the periphery of a SI, suggesting that they originated from three different IPD cells. (E) A PI showing two peripheral H2BGFP+ groups of β cells (arrowheads), indicating several cell cycles after differentiation. (F) A single H2BGFP+ β cell (arrowhead) in proximity to an SI. Of 20 animals treated with 4-OHT, 15 contained lineage-traced cells, whereas no lineage-traced cells were observed in vehicle-treated controls (n = 7 animals, 80 SIs and 7 PIs). (G) Tg(TP1:H2BmCherry);Tg(ins:GFP) animals were examined at 30 dpf. A projection of several planes shows an SI (arrow). The white arrowhead points to a single β cell in the periphery of the SI. The yellow arrowhead points to a single β cell outside the SI, extending a long cellular process toward it. Both of these β cells exhibit higher levels of Tg(TP1:H2BmCherry) fluorescence in comparison to the rest of the β cells in the SI, suggesting that they recently differentiated and did not undergo proliferation. (H and I) Tg(ins:Kaede) animals were maintained on a low-calorie diet until 20 dpf (H) or switched to a high-calorie diet (I) from 15 to 20 dpf (see Experimental Procedures). Although there is only a single Tg(ins:Kaede)+ SI (arrowhead) posterior to the PI (arrow) in (H), multiple SIs (arrowheads) formed in (I). (J) Quantification of the number of SIs posterior to the PI (n = 17 animals for each group). The high-calorie diet induced the formation of more SIs in comparison to the low-calorie diet (****p < 0.0001). Error bars represent SEM, and scale bars represent 20 μm. See also Figure S2. EXPRESSION / LABELING:
PHENOTYPE:
|
|
β Cell Ablation Triggers a Regenerative Response in the IPD via Notch Signaling (A?D) Confocal micrographs of WT and β-cell-ablated animals at the indicated stages. β cells are marked by insulin immunoreactivity (blue). Tg(TP1:H2BmCherry) expression labels pancreatic NRCs in the IPD, and Tg(neuroD:GFP) expression marks endocrine cells and their direct precursors. Arrows point to the PI, and arrowheads point to SIs. (A and B) At 6.5 dpf and in the absence of external nutrition, the β-cell-ablated animals (B) do not exhibit any visible phenotype in the IPD or in the differentiation of NRCs into endocrine cells in comparison to WT animals (A) (n = >30 animals each). (C and D) Confocal micrographs of WT and β-cell-ablated animals at 15.5 dpf under external nutrition. At this stage, in WT animals (C), the pancreatic NRCs have expanded in numbers, and their nuclei are clearly separated. In contrast, β-cell-ablated animals exhibit a dramatic clustering of the NRCs as well as an increase in the number of SIs composed of Tg(neuroD:GFP)+ cells. (E) Quantification of the number of SIs for WT (n = 24) and β-cell-ablated (n = 33) animals at 6.5 dpf. (F) Quantification of the number of SIs for WT (n = 12) and β-cell-ablated (n = 34) animals at 16.5 dpf. Overall, β-cell-ablated animals exhibit a significant increase in the number of SIs (p = 0.014) in comparison to WT animals. (G and H) Confocal micrographs of the pancreatic tails of WT (G) and β-cell-ablated animals (H) at 16.5 dpf. β cells are marked by insulin immunoreactivity (green). 2F11 immunoreactivity (blue) marks IPD cells. Whereas, in WT animals, the vast majority of Tg(TP1:H2BmCherry)+ NRCs exhibit 2F11 immunoreactivity and form part of the ductal network, in β-cell-ablated animals, a large proportion of the Tg(TP1:H2BmCherry)+ NRCs have lost 2F11 immunoreactivity as well as ductal organization. Some of these NRCs exhibit insulin immunoreactivity (white arrowheads), indicating an attempted β cell regeneration. Only a few of the Tg(TP1:H2BmCherry)+ cells maintain high 2F11 immunoreactivity (yellow arrowheads). (I?L) Tg(TP1:VenusPEST), which expresses a fluorescent protein with a short half-life under a Notch-responsive element (TP1), was used to determine the levels of Notch signaling in WT and β-cell-ablated animals at the indicated stages. Arrows point to the PI. (I and J) At 6.5 dpf, in both WT (I) and β-cell-ablated (J) animals, the IPD cells maintain active Notch signaling and are double positive for Tg(TP1:H2BmCherry) and Tg(TP1:VenusPEST) expression. Note that, in both cases, the IPD cells exhibit branching morphogenesis (yellow arrowheads). (K and L) At 15.5 dpf, in contrast to WT animals (K) where a majority of IPD cells maintain active Notch signaling, β-cell-ablated animals (L) exhibit a strong reduction in, and even a loss of, Notch signaling in the IPD, as indicated by the presence of numerous Tg(TP1:H2BmCherry)+ but Tg(TP1:VenusPEST) cells (white arrowheads). Only some cells with active Notch signaling are present (yellow arrowheads). All panels show lateral views, anterior to the top and dorsal to the left. Error bars represent SEM, and scale bars represent 20 μm. See also Figure S3. EXPRESSION / LABELING:
PHENOTYPE:
|
|
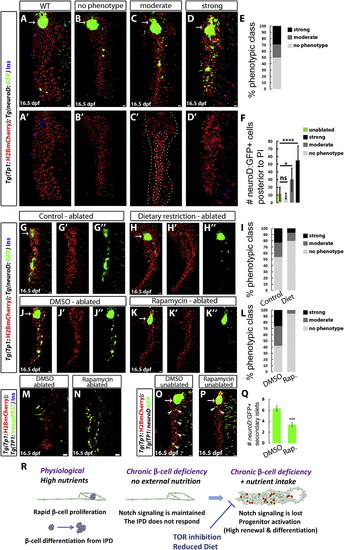
High Nutrient Intake and TOR Signaling Are Required for the Activation of the Regenerative Response of IPD Cells (A?D) WT (A) and β-cell-ablated (B?D) animals at 16.5 dpf. The phenotypes of β-cell-ablated animals fall into several classes based on the degree of IPD disorganization and the extent of endocrine differentiation. (B) No phenotype; lack of IPD cell clustering and no increase in ectopic Tg(neuroD:GFP)+ cells posterior to the PI in comparison to WT animals (quantified in F). (C) Moderate phenotype; only some Tg(TP1:H2BmCherry)+ cells exhibit clustering (outlined by the white/inner dashed line in C′), whereas the rest (outlined by the yellow/outer dashed line in C′) appear unaffected. In these animals, the number of Tg(neuroD:GFP)+ cells posterior to the PI is significantly increased in comparison to WT animals (quantified in F). (D) Strong phenotype; complete clustering of the pancreatic NRCs and dramatic increase in Tg(neuroD:GFP)+ cells (quantified in F). (E) Distribution of phenotypic classes (no phenotype, 17 animals; moderate phenotype, 7 animals; and strong phenotype, 10 animals). (F) Quantification of the number of Tg(neuroD:GFP)+ cells posterior to the PI for each phenotypic class (ns, not significant; p < 0.05, ****p < 0.0001). (G and H) β-cell-ablated animals reared under a normal feeding regimen (G) or a 24 hr feeding-fasting regimen (H) (6?16 dpf). (I) Distribution of phenotypic classes for control (n = 22) versus dietary restriction (n = 15). The restricted diet suppressed the strong and moderate IPD phenotypes. (J and K) β-cell-ablated animals treated with DMSO (J) or 50 nM rapamycin (K) (8?16 dpf). The DMSO-treated animals exhibit clustering of the pancreatic NRCs as well as numerous Tg(neuroD:GFP)+ cells in the pancreatic tail, whereas, in rapamycin-treated animals, the NRCs did not undergo clustering or endocrine differentiation. (L) Distribution of phenotypic classes for animals treated with DMSO (n = 31) versus rapamycin (n = 16). Rapamycin treatment strongly suppressed the phenotypes observed in β-cell-ablated animals. (M and N) Tg(TP1:H2BmCherry); Tg(TP1:VenusPEST) β-cell-ablated animals treated with DMSO (M) or 50 nM rapamycin (N) (6?16 dpf). Of the ten DMSO-treated animals, three exhibited a loss of Tg(TP1:VenusPEST) expression in the IPD, whereas, in the rapamycin-treated animals, Tg(TP1:VenusPEST) expression was maintained (n = 15 animals). (O and P) WT animals treated with DMSO (O) or 50 nM rapamycin (P) (6?16 dpf). The DMSO-treated animal exhibits five Tg(neuroD:GFP)+ SIs (arrowheads) in its pancreatic tail, whereas the rapamycin-treated one exhibits two SIs (arrowheads). (Q) Quantification of the number of SIs posterior to the PI for DMSO- and rapamycin-treated WT animals (n = 28 for DMSO; 27 for rapamycin). Rapamycin reduced the formation of new SIs (***p < 0.001). Error bars in (F) = SD, error bars in (Q) = SEM. (R) Under physiological conditions, high nutrient intake induces β cell proliferation as well as a progressive differentiation of new β cells from the IPD. When the differentiated β cells are ablated, the combination of β cell deficiency and nutrient intake leads to the downregulation of Notch signaling in the IPD. The IPD cells undergo differentiation toward the endocrine lineages as well as an increase in proliferation and loss of ductal characteristics. This process is nutrient dependent, given that it requires high nutrient intake and can be suppressed by a dietary restriction or TOR signaling downregulation. All panels show lateral views, anterior to the top, dorsal to the left. (M and N) show single planes through the IPD, and all other panels show projections of stacks. Scale bars represent 20 μm. See also Figure S4. |
|
Secondary Islets Are Vascularized; Development of Transgenic FUCCI Reporters for Dynamic Assessment of β Cell Proliferation, Related to Figure 1 (A and B) Tg(ins:GFP);Tg(flk1:ras-Cherry) expression labels β-cells and blood vessels, respectively. (A) Single plane through a large SI. Individual β-cells appear to be in direct contact with blood vessels (arrowheads) (30dpf). (B) Projection of several planes showing two small SIs (arrowheads) closely associated with blood vessels (30 dpf). |
|
Differentiation of β Cells from Notch Responsive Cells during Juvenile Stages, Related to Figure 2 (A and B) Tg(TP1:CreERT2);Tg(ubi3C:loxp-GFP-loxp:mCherry) larvae were treated with EtOH as vehicle (A) or 4-OHT (B) at 14dpf for 16h, fixed at 17dpf, and stained for 2F11 to mark the IPD cells. 4-OHT treatment resulted in the mosaic labeling of IPD cells (mCherry+; 2F11+ cells). |
|
β Cell Deficiency during the Stage of External Nutrition Remodels the IPD and Activates Endocrine Progenitors; This Process Can Also Be Stimulated by Notch Signaling Downregulation in WT Animals, Related to Figure 3 (A and B) Conditional ablation of β-cells is achieved by Tg(ins:Cre) mediated excision of the BFP cassette from the floxed ins:loxp:BFPloxp:DTA transgene. |
|
High Glucose Increases Endocrine Differentiation; TOR Is Required for the Expansion of the Progenitor Pool in the IPD, Related to Figure 4 (A?D) Tg(TP1:H2BmCherry);Tg(neuroD:GFP) animals were incubated in egg water (A), 3% D -glucose solution (B), DMSO (C) or 10 μM Prednisolone (Glucocorticoid) (D) from 3.5 to 7dpf. Whereas each control animal (A, C) exhibits only one Tg(neuroD:GFP)+ SI (arrowheads), the glucose- and prednisolone-treated animals have formed several SIs (arrowheads). The high magnification projections with separate channels show that all ectopic Tg(neuroD:GFP)+ cells are also Tg(TP1:H2BmCherry)+ and thus have recently 10 differentiated from NRCs. Yellow arrowheads point to Tg(neuroD:GFP)+ cells that also exhibit Insulin immunoreactivity. |