- Title
-
Essential role for the alpha 1 chain of type VIII collagen in Zebrafish notochord formation
- Authors
- Gansner, J.M., and Gitlin, J.D.
- Source
- Full text @ Dev. Dyn.
|
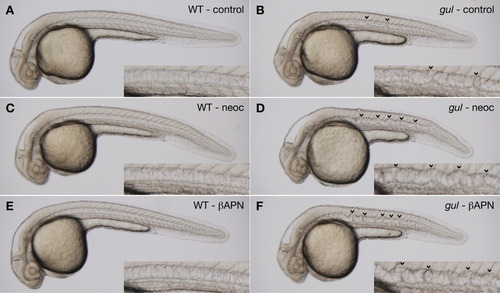
gulm208 mutants are sensitized to lysyl oxidase inhibition. A-F: Clutches from gulm208/+ intercrosses were incubated in vehicle (A,B), the copper chelator neocuproine (2 μM; C,D), or the lysyl oxidase inhibitor β-aminopropionitrile (1 mM; E,F). The notochord is normal in wild-type embryos treated with vehicle, neocuproine, or β-aminopropionitrile (A,C,E). Notochords of gulm208 mutants incubated in neocuproine and β-aminopropionitrile (D,F, arrowheads) are substantially more distorted than mutants incubated in vehicle (B, arrowheads). Insets show notochord at higher magnification. Embryos were treated with PTU to inhibit melanin pigmentation and photographed at 30 hours postfertilization (hpf). PHENOTYPE:
|
|
Electron microscopy reveals notochord abnormalities in gulm208 mutants. A-E: Transmission electron micrographs of truncal cross-sections from embryos at 24 hpf. A: Notochord sheath and vacuolated notochord cell of a wild-type embryo, with small areas of protein accumulation visible in the rough endoplasmic reticulum (arrow). B: Notochord sheath and vacuolated notochord cell of a gulm208 mutant with large circular aggregates of protein in the rough endoplasmic reticulum (arrow). C: Notochord and hypochord of a gulm208 mutant with large circular aggregrates of protein in the hypochord (red arrows) and notochord (yellow arrow). D: Notochord sheath of a wild-type embryo with inner (i), medial (m), and outer (o) layers indicated. Small areas of protein accumulation are visible in the rough endoplasmic reticulum (red arrow). E: Notochord sheath of a gulm208 mutant where the collagen fibrils in the medial (m) layer appear disorganized. A large circular aggregate of protein is visible in the rough endoplasmic reticulum (red arrow). Not, vacuolated notochord cell; Hyp, hypochord. PHENOTYPE:
|
|
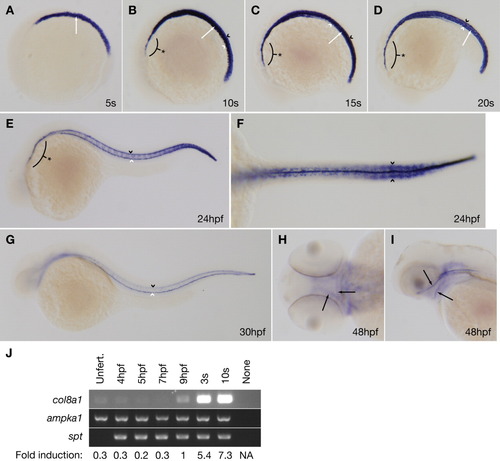
col8a1 expression is consistent with the gulm208 phenotype. A-I: Whole-mount in situ hybridization for col8a1 was performed on wild-type embryos at various developmental stages. A: Lateral view of a five-somite embryo with col8a1 expression in the vacuolated notochord cells (arrow). B-D: Lateral views of 10- (B), 15- (C), or 20- (D) somite embryos with col8a1 expression in the notochord (arrow), floorplate (black arrowhead), hypochord (white arrowhead), and prechordal plate region (asterisk). E: Lateral view of an embryo at 24 hours postfertilization (hpf) with col8a1 expression in the floorplate (black arrowhead), hypochord (white arrowhead), and prechordal plate region (asterix). F: Dorsal view of an embryo at 24 hpf with col8a1 expression in the caudal somites (arrowheads). G: Lateral view of an embryo at 30 hpf with col8a1 expression in the floorplate (black arrowhead) and hypochord (white arrowhead). H: Dorsal view of an embryo at 48 hpf with col8a1 expression in jaw cartilages (arrows). I: Lateral view of an embryo at 48 hpf with col8a1 expression in jaw cartilages (arrows). J: reverse transcriptase-polymerase chain reaction (RT-PCR) for col8a1 at the indicated developmental stages. As controls, maternally-expressed 5′-AMP-activated protein kinase catalytic subunit alpha-1 (ampka1) and zygotically expressed spadetail (spt) were amplified in parallel (Griffin et al.,[1998]; Wagner et al.,[2004]). The fold induction of col8a1 relative to ampka1 is noted. Unfert, unfertilized; NA, not applicable. |
|
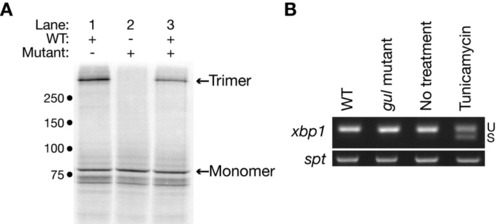
The Y628H substitution prevents Col8a1 trimerization. A: Constructs encoding wild-type (lane 1) or gulm208 mutant (lane 2) Col8a1 were transcribed and translated in the presence of radiolabeled methionine, and the products analyzed as described under the Experimental Procedures section. Col8a1 monomers and trimers are indicated. Cotranscription and translation of half the amounts of wild-type and mutant construct together does not prevent trimerization (lane 3). The approximate location of molecular weight markers (kDa) is noted. B: Reverse transcriptase-polymerase chain reaction (RT-PCR) to assess the splice status of xbp1 in wild-type embryos and gulm208 mutants at 24 hours postfertilization (hpf). Splicing of xbp1 is not observed in gulm208 mutants but occurs after incubation of wild-type embryos in tunicamycin. U, unspliced; S, spliced. Amplification of the gene spt was performed in parallel to confirm that equal amounts of cDNA were used for each condition. EXPRESSION / LABELING:
|
|
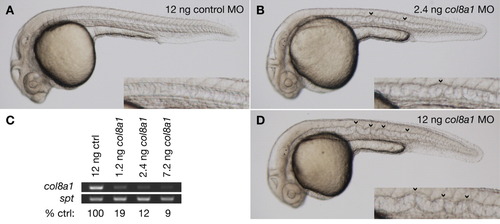
Morpholino knockdown of col8a1 recapitulates the gulm208 phenotype. A: Wild-type embryos injected with 12 ng of control morpholino exhibit normal morphology. B: Embryos injected with 2.4 ng of a splice morpholino targeting col8a1 develop notochord distortion (arrowheads) characteristic of gulm208 mutants. C: Reverse transcriptase-polymerase chain reaction (RT-PCR) at 24 hours postfertilization (hpf) demonstrating a dose-dependent reduction in col8a1 transcript after morpholino injection. D: Embryos injected with 12 ng of a splice morpholino targeting col8a1 develop notochord distortion (arrowheads) that is worse than in gulm208 mutants. Insets show notochord at higher magnification. All embryos were treated with PTU to inhibit melanin pigmentation and photographed at 30 hpf. |

Unillustrated author statements PHENOTYPE:
|