- Title
-
Development without germ cells: The role of the germ line in zebrafish sex differentiation
- Authors
- Slanchev, K., Stebler, J., de la Cueva-Mendez, G., and Raz, E.
- Source
- Full text @ Proc. Natl. Acad. Sci. USA
|
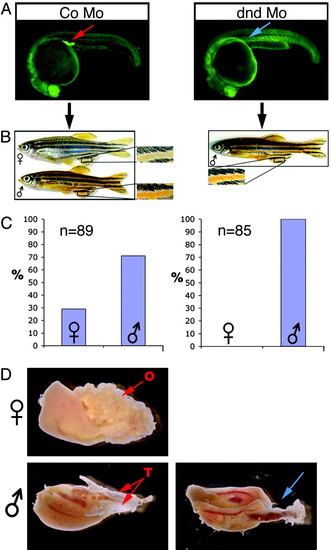
dead end MO-injected embryos develop into sterile adult male fish. (A) Fluorescent images of 24-hpf embryos injected with 200 pg of control antisense MO or dnd MO and GFP-nos1 3′-UTR to visualize the PGCs. The red arrow indicates the germ cells in control embryos, and the blue arrow indicates the region where the PGC are normally found. (B) Adult AB fish (6 months old) developed from control (Left) or dnd MO-injected (Right) embryos. Insets are magnifications of the male and female fins showing the typical sexually dimorphic pigmentation. (C) A quantitative analysis of the female/male ratio. (D) Dissected gonads from 180-day-old control and dnd MO-treated fish. Red arrows indicate ovary (O) or testis (T) in the control fish. The blue arrow indicates the region of the gonad in a phenotypically appearing male fish developed from a dnd MO-injected embryo. |
|
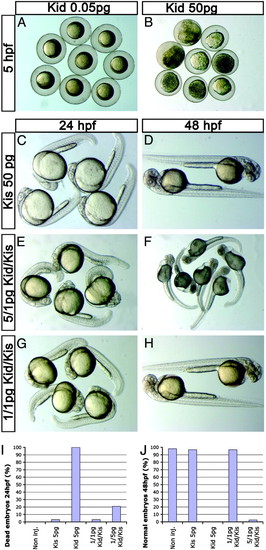
The prokaryotic toxin Kid and antitoxin Kis are functional in zebrafish. (A and B) High concentrations of Kid result in rapid embryonic lethality in embryos injected with kid-nos1 3′-UTR RNA. (C and D) Embryonic development is not affected by high levels of somatically expressed antidote kis-glo UTR RNA. The deleterious effect of 5 pg of Kid on somatic cell development is counteracted efficiently by 1 pg of kis-glo UTR for up to 24 hpf (E), but not at 48 hpf (F). (G and H) A ratio of 1:1 pg of kid-nos1 3′UTR/kis-glo UTR mRNAs allows proper somatic development during the first 2 days of development. (I and J) Quantitative representation of Kid-induced somatic phenotypes during the first 2 days of development. |
|
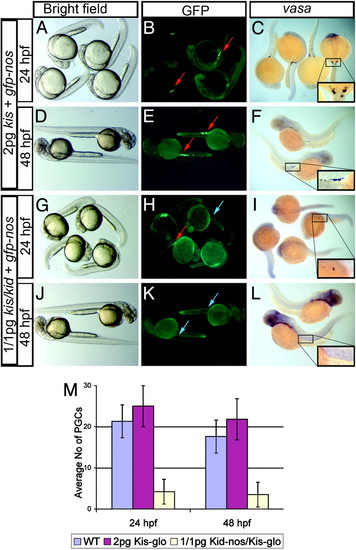
kid and kis allow effective ablation of the germ line without affecting embryonic morphology. (A and D) Control embryos injected with 2 pg of the antidote RNA developed without apparent somatic defects and are morphologically similar to embryos injected with 1 pg of kid-nos1 3′UTR plus 1 pg of kis-glo (G and J). Reduction in the number and lack of GFP-labeled (B, E, H, and K) or vasa RNA-labeled (C, F, I, and L) germ cells at 24 and 48 hpf. Red arrows indicate the germ cells, and blue arrows indicate the regions where PGCs are normally found in the fluorescent images (B, E, H, and K). The same regions are boxed and magnified in C, F, I, and L.(M) A quantitative representation of the number of germ cells after different treatment at 24 and 48 hpf, as determined by in situ hybridization using vasa antisense RNA probe. |
|
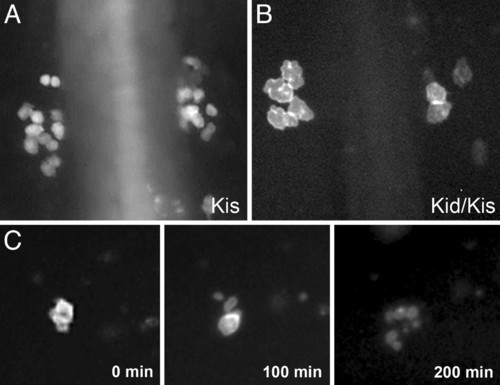
PGCs in kid-nos1 3′UTR treated embryos exhibit abnormal cell morphology culminating in cell death. Fluorescently labeled germ cells in kis-glo UTR (A) and kid-nos1 3′UTR/kis-glo UTR-injected (B) embryos at 12 hpf are shown. More PGCs are found in the control embryos and these appear smaller in size relative to kid-treated PGCs. (C) Frames from a time-lapse movie of a kid-nos1 3′-UTR-treated embryo showing a PGC undergoing cell death (see Movie 2). |
|
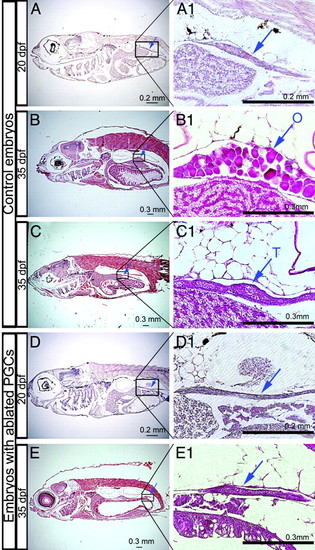
The formation of the zebrafish gonads does not depend on colonization by PGCs. (A–C) Hematoxylin–eosin-stained paraffin sections of 20- and 35-dpf fish derived from embryos injected with control MO. (A1–C1) Magnifications of the boxed regions of the gonad in A–C. Blue arrows indicate the gonads labeled as testis (T), ovary (O), or undifferentiated (left blank). (D and E) Sections of 20- and 35-dpf fish injected with 200 pg of dnd MO. Arrows indicate gonadal tissue, which is detected at the correct position (D1 and E1). |
|
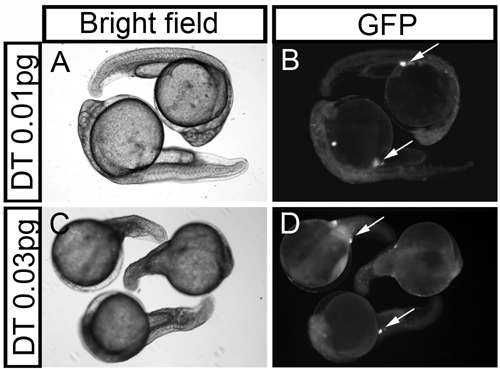
Diphtheria toxin (DT) A-chain-nos1 3′-UTR mRNA-injected embryos exhibit somatic malformations. (A and C) Bright field images of embryos injected with 0.01 and 0.03 pg of DT-nos1 3′-UTR mRNA show concentration-dependent increase of somatic defects. (B and D) Fluorescent images of the DT-injected embryos, demonstrating that some PGCs survive in the defective treated embryos. Arrows in B and D indicate the PGCs. |