|
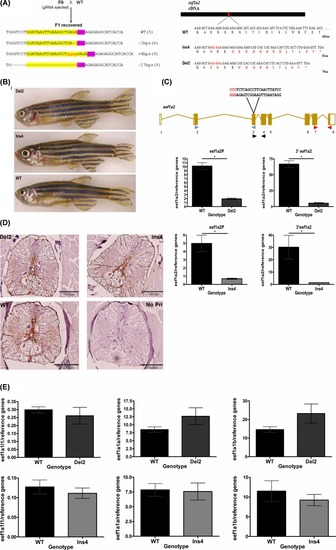
Generation and characterisation of Eef1a2-null zebrafish lines using CRISPR/Cas9(A) Schematic showing outcross mating of founder (F0) fish and wild-type fish showing recovered F1 sequences (number of F1 fish for each allele are indicated in brackets, target sequences (yellow highlight) and PAM site (purple) with red showing inserted bases) and the predicted effect of Ins4 and Del2 mutant allele with aberrant residues shown in red (right). (B) No overt difference in homozygous Del2 (6 months) and homozygous Ins4 (8 months) adult fish from wild-type (6 months) adult fish. (C) The position of the three different primer sets; eef1a2S (blue triangle), eef1a2P (black triangle) and 3′eef1a2 (red triangle) is illustrated in relation to the gRNA target site (PAM site sequence in red). Expression levels of eef1a2 in homozygote Del2 (top panel) and homozygote Ins4 (bottom panel) fish using eef1a2P and 3′eef1a2 primer sets. Results were normalised to ATPsynth, NADH and 16S. (means ± S.E.M; n=3), *P<0.05. (D) Anti-GFAP antibody stained transverse sections of spinal cords of homozygous Del2 and Ins4 adult fish showed no sign of neurodegeneration. Negative control of a no primary (No Pri) was included which showed no staining. Scale bar = 100 µm. (E) Expression levels of eef1a1l1 (left), eef1a1a (middle) and eef1a1b (right) mRNA in homozygous Del2 (top panel) and Ins4 (bottom panel) adult brain. Data were normalised to ATPsynth, NADH and 16S and were presented as means ± S.E.M.; n=3.
|