- Title
-
Developmental beta-cell death orchestrates the islet's inflammatory milieu by regulating immune system crosstalk
- Authors
- Akhtar, M.N., Hnatiuk, A., Delgadillo-Silva, L., Geravandi, S., Sameith, K., Reinhardt, S., Bernhardt, K., Singh, S.P., Maedler, K., Brusch, L., Ninov, N.
- Source
- Full text @ EMBO J.
|
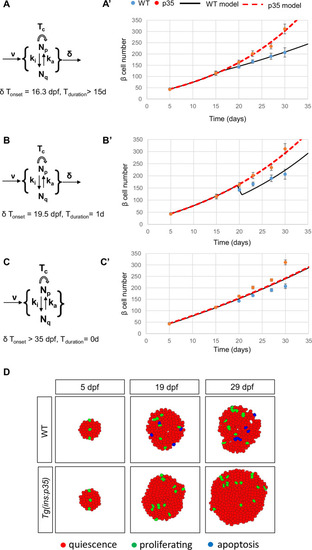
Genetic overexpression of ( |
|
Mathematical model. ( |
|
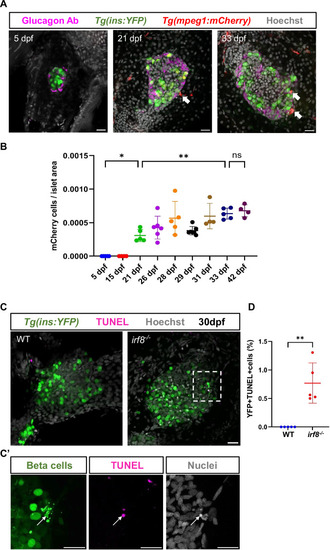
Macrophage colonization coincides with the onset of developmental beta-cell apoptosis, and their depletion reveals developmental beta-cell apoptosis. ( |
|
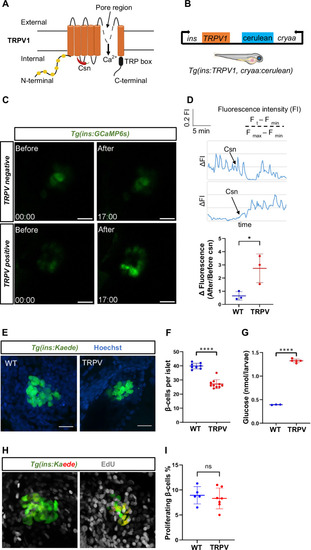
Generation of a genetic model of beta-cell excitotoxicity in zebrafish. ( |
|
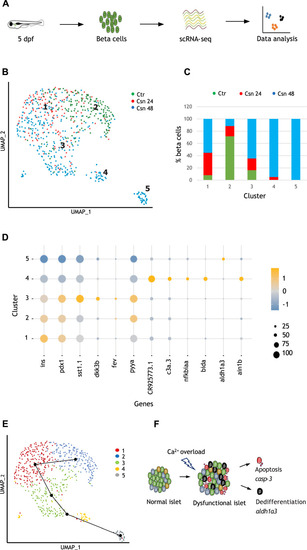
Beta cells experiencing Ca2+ excitotoxicity show transcriptional trajectories toward cell death or dedifferentiation. ( |
|
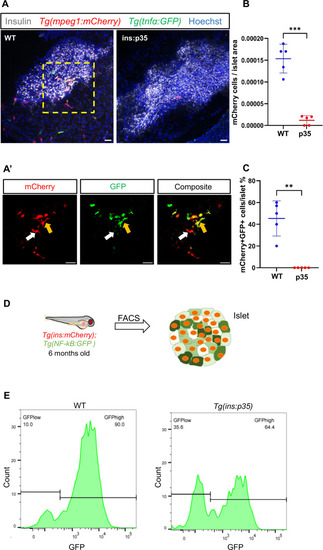
Beta-cell death regulates the islet’s immune component and inflammation. ( |
|
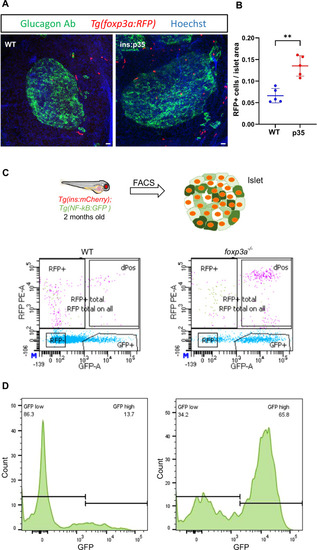
Zebrafish Tregs populate the islets of p35-expressing beta cells and regulate the levels of islet inflammation. ( |