- Title
-
Overexpression of a slit homologue impairs convergent extension of the mesoderm and causes cyclopia in embryonic zebrafish
- Authors
- Yeo, S.Y., Little, M.H., Yamada, T., Miyashita, T., Halloran, M.C., Kuwada, J.Y., Huh, T.L., and Okamoto, H.
- Source
- Full text @ Dev. Biol.
|
Expression patterns of zebrafish slit2 mRNA. (A) Expression of slit2 mRNA at the midblastula stage (4 h). (B) Expression of slit2 mRNA at 70% epiboly. Lateral view. (C, D) Expression of slit2 mRNA at 80% epiboly. Expression in the axial mesoderm (am, white arrows) is indicated. C, lateral view; D, dorsal view. (E, F) Expression of slit2 mRNA in 10-h embryos. E, dorsal view; F, lateral view. slit2 mRNA is expressed in the anterior margin of the neural plate (arrowheads), the axial mesoderm (white arrow). (G, H) Expression of slit2 mRNA in 16-h embryos. G, lateral view; H, a close-up view of the somites in a parasagittal section. slit2 mRNA is expressed in the anterior margin of the forebrain (arrowhead), floor plate cells (fp and thin arrow), the hypochord (hc), and the anterior margin of the somites (as). (I) Expression of slit2 mRNA in 18-h embryo. (J–M) Expression of slit2 mRNA in the head region of 48-h embryos. J and K, lateral views; M, dorsal view; L, a sagittal section. In K, the axons in the initial scaffold of the brain are immunohistochemically stained with an antibody for acetylated ;;alpha;-tubulin (brown signals). slit2 mRNA is expressed in the floor plate cells (thin arrows) and their anterior equivalents in the ventral forebrain (thick arrows), and in a small number of cells (asterisks) dorsally adjacent to the supraoptic tract (SOT). tc, tectum; hb, hindbrain; hy, hypothalamus; n, notochord; AC, anterior commissure; DVDT, dorsoventral diencephalic tract; POC, postoptic commissure. Bars, 200 ;;mu;m (A–D, E and F, G and I, J–M), 50 ;;mu;m (H). |
|
Expression patterns of zebrafish slit3 mRNA. (A) Expression of slit3 mRNA at 70% epiboly in the axial mesoderm (am, white arrow). Dorsal view. (B, C) Expression of slit3 mRNA in 10-h embryos. Dorsal views of the rostral (B) and caudal (C) parts of the embryo. slit3 mRNA is expressed in the polster (p) as well as in axial mesoderm cells (white arrow). (D) Expression of slit3 mRNA in 26-h embryo. slit3 mRNA is expressed in the motor neurons in the ventral spinal cord (arrows). The lower inset shows the close-up view of the motor neurons. (E–G) Expression of slit3 mRNA in the head region of 48-h embryos. E, lateral view; F, a sagittal section; G, dorsal view. slit3 mRNA is expressed in the cranial motor neurons (arrowheads) and the floor plate cells (fp) in the hindbrain. (H) Expression of Islet-1 mRNA in the head region of 48-h embryos. Islet-1 mRNA is also expressed in the cranial motor neurons (arrowheads). III, IV, Va, Vp, VII/VIII, X, oculomotor, trochlear, anterior trigeminal, posterior trigeminal, facial/octavolateralis, and vagus motor neurons, respectively. (I–L) Expression of slit3 mRNA in various parts of 48-h embryo. slit3 mRNA is expressed in the pectoral fin bud (I), the pharyngeal arches (m, h, b in J), spinal motor neurons, and the roof plate (arrows and white arrowhead in K, respectively) and the lens (l in L). I, lateral view; J, a parasagittal section; K, a cross section of the anterior spinal cord; L, a frontal section of the head. m, h, and b, the mandibular arch, the hyoid arch, and the posterior set of branchial arches, respectively; e, eye; ov, otic vesicle; s, somites; n, notochord. Bars, 200 μm (A–D, E–H, L), 100 μm (I–K). |
|
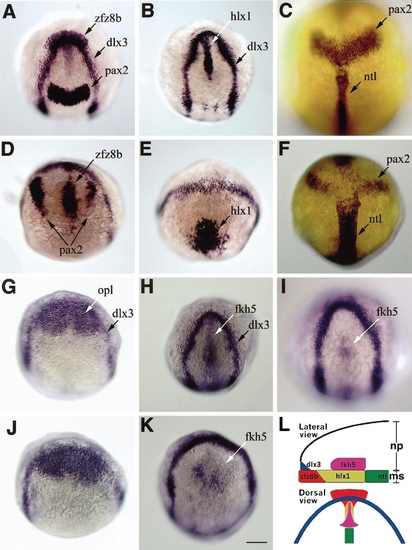
Overexpression of slit2 mRNA impairs migration of the prechordal mesodermal cells and the ventral diencephalic cells in the zebrafish embryo. (A–F) The retarded migration of the prechordal plate cells in the slit2-overexpressing embryos (D–F) in comparison to normal embryos (A–C) at 100% epiboly. The positions of the mesodermal cells were examined by comparing the expression patterns of the prechordal plate mesoderm markers, zfz8b (A, D) and ,em>hlx1 (B, E), and the chordal mesoderm marker ntl (C, F) relative to the neural plate markers dlx3 (A, B, D, E), and pax2 (A, C, D, F), which are expressed in the anterior margin of the neural plate and in the midbrain/hindbrain boundary (MHB), respectively. In the slit2-overexpressing embryos, the zfz8b- and hlx1-positive prechordal mesodermal cells are prominently displaced caudally and are improperly located between the anterior margin of the neural plate and MHB. The chordal mesoderm is wider and flatter in the slit2-overexpressing embryo than in the normal embryo. (G–K) The retarded migration of the ventral diencephalic cells both in the slit2-overexpressing and cyctz219 mutant embryos at 100% epiboly. The position of the fkh5-positive diencephalic cells relative to the dlx3-positive anterior margin of the neural plate is displaced caudally in the slit2-overexpressing embryo (K) in comparison to the normal embryo (H), as in the cyctz219 mutant (I). The opl-positive cells are normally distributed in the slit2-overexpressing embryo (J) as in the normal embryo (G). (L) Schematic of the normal expression patterns of the marker genes used in this figure. np, neural plate; ms, chordal and prechordal mesoderm. Bars, 200 μm. |
|
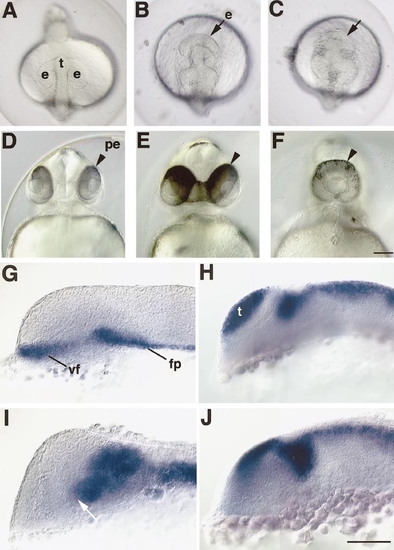
Overexpression of slit2 mRNA induces cyclopia in the zebrafish embryo. (A–F) Various degrees of cyclopia were observed in the slit2-overexpressing embryos at 16 h (B, C) and 48 h (E, F). A and D, normal embryos. Embryos are shown in dorsal views (A–C) and in frontal views (D–F). (G–J) Defect in the ventral forebrain of 16-h zebrafish embryo overexpressing slit2 mRNA. (G, H) Expression of shh (G) and opl (H) mRNA in normal embryos. (I, J) Expression of shh (I) and opl (J) mRNA in embryos overexpressing slit2 mRNA. In the slit2-overexpressing embryo, the anterior edge of the shh-positive domain in the ventral forebrain (vf) is caudally shifted as indicated by the arrow in (I). In contrast, opl mRNA is expressed indistinguishably in the dorsal forebrain of the normal embryo (H) and the embryo overexpressing slit2 mRNA (J). e and arrows, eyes; pe and arrowheads, the retinal pigment epithelium; t, telencephalon; fp, floor plate cells. Bars, 200 μm (A–F, G–J). |
|
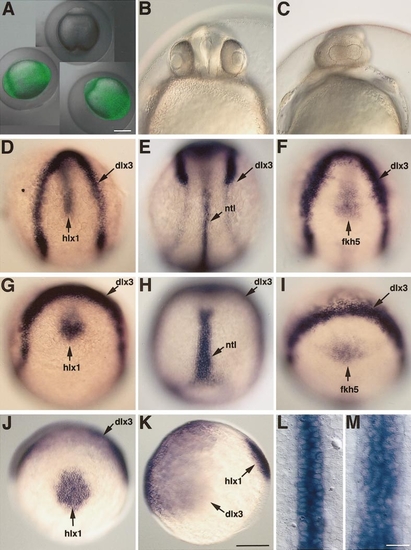
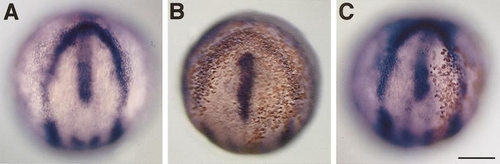
Overexpression of slit2 at the late gastrula stage suppressed the convergent extension of the mesoderm. (A) The transgenic embryos (green) overexpressing the Slit2/GFP fusion protein and a wild-type sibling (dark) at 12 h which had been heat-shocked at 100% epiboly (10 h). (B, C) The slit2-overexpressing transgenic embryo which had been heat shocked at 80% epiboly suffered from severe cyclopia at 28 h. Frontal views of the wild-type sibling (B) and the slit2-overexpressing transgenic embryo (C). (D–I) Heat-shock induction of Slit2 at 80% epiboly impaired the convergent extension movement of the mesoderm and the anterior migration of the diencephalic cells. dorsal views, anterior to the top. The positions of the axial cells were examined by comparing the expression of axial mesoderm markers, hlx1 (D, G) and ntl (E, H), and the ventral forebrain marker, fkh5 (F, I), relative to the marker for anterior neural plate margin, dlx3 (D–I). Compared to the wild-type siblings (D–F), the transgenic embryos (G–I) overexpressing Slit2 by heat shock at 80% epiboly showed impaired convergent extension of the mesoderm and defect in the anterior migration of the diencephalic cells. (J, K) Normal embryos at 80% epiboly for comparisons. (L, M) Close-up views of the notochord of the normal embryo shown in E and the heat-shocked transgenic embryo shown in H, respectively. Bars, 250 μm (A, B–K), 50 μm (L and M). |
|
Localized ectopic expression of Slit2 impairs the mesodermal movement. The embryos expressing slit2 and gfp mRNA in a mosaic pattern were fixed at 10 h and stained for hlx1 and dlx3 expression by in situ hybridization and for GFP by immunohistochemistry. Dorsal views, anterior to the top. (A) Control embryo. (B) The embryos (n 5 3) in which the GFP-positive cells were broadly distributed in the anterior region of the neural plate. The hlx1-positive cells of the prechordal mesoderm failed to reach the anterior margin of neural plate. (C) The embryos (n 5 17) in which the GFP-positive cells were incorporated on one side of the neural plate and the adjacent ectoderm. The hlx1-positive cells were bent toward the side which incorporated the GFP-positive cells. Bar, 250 μm. |
Reprinted from Developmental Biology, 230(1), Yeo, S.Y., Little, M.H., Yamada, T., Miyashita, T., Halloran, M.C., Kuwada, J.Y., Huh, T.L., and Okamoto, H., Overexpression of a slit homologue impairs convergent extension of the mesoderm and causes cyclopia in embryonic zebrafish, 1-17, Copyright (2001) with permission from Elsevier. Full text @ Dev. Biol.