- Title
-
Zebrafish mdk2, a novel secreted midkine, participates in posterior neurogenesis
- Authors
- Winkler, C. and Moon, R.T.
- Source
- Full text @ Dev. Biol.
|
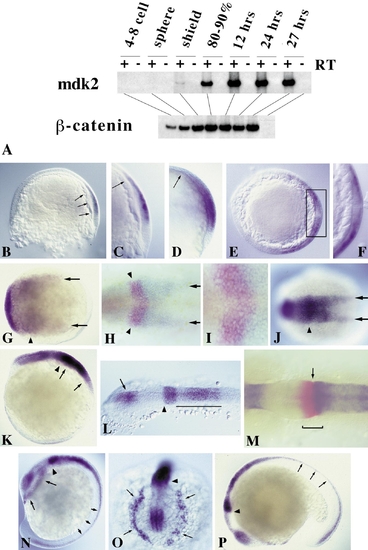
Expression of mdk2 during zebrafish embryogenesis. (A) RT-PCR of different embryonic stages. β-catenin was used as loading control. (B–N) RNA whole-mount in situ hybridization using full-length mdk2 as probe. (B) 60% epiboly (lateral view, dorsal to the right). Onset of mdk2 expression in the epiblast in a region overlaying the involuting hypoblast (arrows). (C) Close-up of shield region as in B. Arrow indicates anterior border of mdk2 expression overlaying the anterior edge of involuting mesendoderm. (D) 70% epiboly. Expression throughout the presumptive neural plate. Arrow indicates leading edge of axial hypoblast. (E) Optical section through embryo at 70% epiboly at the level of shield region (dorsal to the right). Dorsoventral gradient of mdk2 expression in the epiblast. (F) Higher magnification of shield region (as boxed in E). mdk2 transcripts are excluded from the involuting mesendoderm. (G) 80% epiboly, dorsal view, anterior to the left. Regionalization of mdk2 expression in the early neural plate. Arrowhead indicates elevated expression at the prospective mid–hindbrain boundary (MHB). Arrows demarcate expression at lateral edges of the neural plate. (H) 90% epiboly, dorsal view. Double labeling with eng2 (in red) demarcating future MHB (arrowheads). Arrows indicate mdk2 expression at the edges of the posterior neural plate. (I) Higher magnification of MHB region as in H showing overlapping expression of mdk2 (blue) and eng2 (red). (J) Bud stage, dorsal view. Arrowhead indicates future MHB, arrows denote expression at the edges of the forming neural keel. (K) Lateral view of embryo in J. Arrows mark the developing hindbrain exhibiting a gradient of mdk2 expression with its maximum at the anterior end. Arrowhead indicates strong mdk2 expression at the MHB. (L) 12-h embryo, dorsal view of head region. mdk2 is expressed in the diencephalon (arrow), at the MHB (arrowhead), and in a gradient in the hindbrain, but excluded from the eye fields and midbrain. (M) 12-h embryo, dorsal view of MHB region. Double labeling with eng2 (red). eng2 is expressed across the MHB (bracket), whereas mdk2 marks the anteriormost edge of the metencephalon (arrow). (N) 14-h embryo, lateral view. mdk2 expression at the MHB (arrowhead), in the dorsal neural tube (small arrows), and in dissociating prechordal plate cells underneath the forebrain (large arrows). (O) Frontal view of head region as in N. Arrows indicate dissociating prechordal plate. (P) Latemdk2 expression in fore- and dorsal midbrain, MHB (arrowhead), hindbrain, and dorsal neural tube (arrows). EXPRESSION / LABELING:
|
|
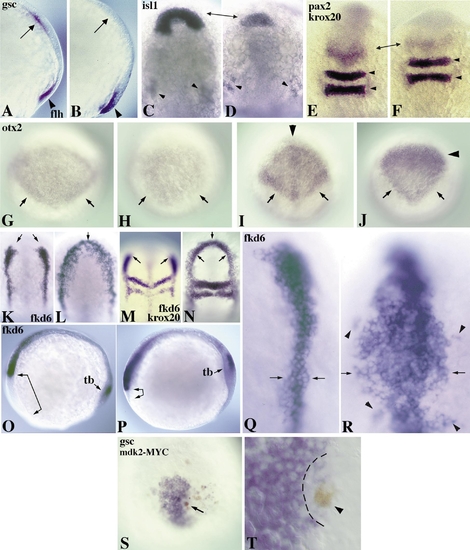
Ectopic expression of mdk2 promotes posterior cell fates and blocks anterior development. (A, B) 80% epiboly embryos, lateral view. Expression of flh (also known as Znot; arrowhead) is unaffected in uninjected (A) and mdk2-injected embryos (B), but gsc (arrow) is repressed in B. (C–F) 12-h embryos, dorsal view, anterior to the top. (C) isl1 expression (arrows) in an uninjected embryo. (D) Repression of isl1 expression in the polster region of a mdk2 injected embryo (arrows). Trigeminal ganglion cells appear normal (arrowheads in C and D). (E) pax2 and krox20 expression in an uninjected embryo. (F) Reduction of pax2 expression in the MHB (arrows) in mdk2-injected embryo. Expression of krox20 in rhombomeres 3 and 5 (arrowheads) is unaffected. (G, H) 80% epiboly embryos, dorsal view, anterior to the top. (G) otx2 expression in the anterior neural plate of an uninjected embryo. Note pronounced expression at the caudal borders (arrows). (H) Reduction of otx2 expression in a mdk2-injected embryo. Border expression is lacking (arrows). (I–R) 12-h embryos, dorsal view (except O and P, lateral view). (I) Diamond-shaped expression of otx2 in the converging anterior neural plate with pronounced caudal borders (arrows) in an uninjected embryo. (J) Ectopic mdk2 expression results in a changed shape of otx2 expression with repression in the anterior domain (anterior to the arrowhead) and a reduction at the posterior edges (arrows). (K) fkd6 expression in premigratory neural crest cells of an uninjected embryo. (L) Expansion of fkd6-positive cells by ectopic expression of mdk2. This leads to a complete anterior fusion of the lateral fkd6 domains (arrows) and a substantial increase in the number of expressing cells. (M) Double labeling of fkd6 with krox20 in an uninjected embryo. (N) Anterior fusion of fkd6 domains (arrows). krox20 expression shows that anteroposterior register is not significantly changed. Lateral views (O, P) show mdk2-induced repression of anterior head structures (arrow-brackets) in mdk2-injected (P) versus uninjected (O) embryos. tb, tail bud. (Q, R) Higher magnification dorsal view of fkd6 expression in neural crest in uninjected (Q) and mdk2-injected (R) embryo. Arrows indicate the width of neural crest precursor populations, arrowheads show isolated population of crest cells. (S, T) 80% epiboly embryos, dorsal view. (S) Local repression of gsc expression in a DNA-injected embryo around single mdk2-MYC-expressing cell in the hypoblast (brown staining, arrow). (T) Higher magnification of a different embryo with repression of gsc in cells surrounding a mdk2-MYC-positive cell (brown, arrowhead). All embryos were injected with 200 pg RNA encoding mdk2 into 1 cell of the 2- to 4-cell stage embryo, except in S and T in which 20 pg DNA encoding MYC-tagged mdk2 was injected into 1 cell of the 8- to 16-cell stage embryo. |
|
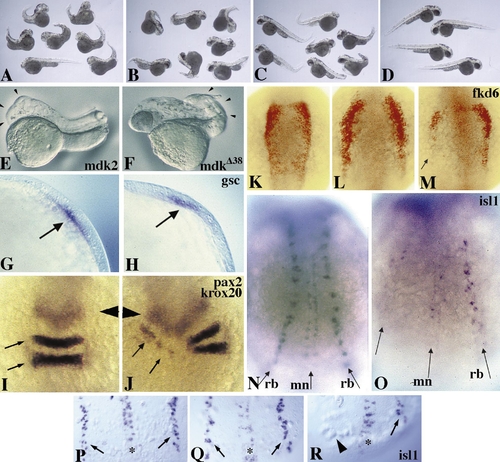
Repression of posterior neural fates by the dominant interfering mdk2 variant mdkΔ38. (A to D) Coexpression of mdkΔ38 rescues head deficiencies induced by ectopic mdk2 expression. (A) Embryos exhibiting a strong posteriorized phenotype at 48 h after injection with 110 pg mdk2 and 330 pg prolactin RNAs. Note that ectopic mdk2 leads to specific repression of head and eye structures and to some curved tails. (B) Partial rescue of head deficiencies by co-injection of 110 pg mdk2, 220 pg mkΔ38, and 110 pg prolactin RNAs. (C) Greater rescue of head deficiencies by co-injection of 110 pg mdk2 and 330 pg mkΔ38. The majority of head structures appear indistinguishable from those of uninjected control embryos (D). The posterior defects observed are likely due to mdkΔ38 interfering with endogenous mdk2 function. (E, F) 24-h embryos injected with 200 pg wild-type mdk2 (E) and mdkΔ38 (F). Note anterior truncation resulting from gain of function (E, arrowheads) and posterior deficiencies resulting from loss of function (F, arrowheads). (G) Lateral view of gsc expression in an uninjected embryo at 80% epiboly. (H) gsc expression (arrow) is unaffected in mdkΔ38-injected embryos. (I–O) 12-h embryos, dorsal view, anterior to the top. (I) pax2 (arrowhead) and krox20 (arrows) expression in an uninjected embryo. (J) Repression of krox20 expression in rhombomeres 3 and 5 of an embryo unilaterally injected with mdkΔ38. Expression of pax2 at MHB appears normal compared to I. (K) fkd6 staining (red) in an uninjected embryo. (L) Expansion of the fkd6-positive neural crest domain in mdk2-injected embryo. (M) Repression of fkd6 by mdkΔ38 in one half of a unilaterally injected embryo (arrow). (N–R) Ectopic mdkΔ38 expression blocks formation of posterior moto- and sensory neurons. (N) isl1 expression in Rohon–Beard sensory neurons (rb) and motoneurons (mn) of an uninjected embryo at 12 hpf. (O) Unilateral lack of both neural fates in an embryo unilaterally injected with mdkΔ38. (P–R) Dorsal view of 10-h embryos. (P) isl1 expression in an uninjected embryo. (Q) Primary neurons appear unaffected in a mdk2-injected embryo. (R) Note unilateral absence of sensory neurons (arrowhead) in mdkΔ38-injected embryo. |
|
mdk2 activity affects emx1 expression pattern in the anterior forebrain and hoxC10 expression in the posterior CNS. (A–D) Lateral views of emx1 expression (arrow) in the telencephalon and hoxc10 expression (arrowhead) in the tail bud of a control embryo (A) and embryos injected with 150 pg RNA encoding mdk2 (B, C) and with 500 pg RNA encoding dominant-negative mdkΔ38 (D). Note reduction (B) or complete repression (C) of emx1 expression (arrows) and expansion of hoxC10 (C, arrowhead) in mdk2-injected embryos. On the other hand, emx1 expression (arrow) appears caudally expanded in mdkΔ38-injected embryos (D). (E–L) Higher magnification of head regions (E–H lateral views, I–L dorsal views, anterior to the left). The emx1 expression domain is shortened (F, J) and dislocates ventrally (F) or is completely missing (G, K) in mdk2-injected embryos compared to control embryos (E, I), while it extends caudally in mdkΔ38-injected embryos (H, L). (M–P) Dorsal views of hoxC10 expression in the tail bud. hoxC10 is upregulated in mdk2-injected embryos (O, compare to control embryo in M), but is not significantly affected in mdkΔ38-injected embryos (P). Note that the mdkΔ38-injected embryo in P is at a slightly earlier stage (12 hpf compared to 14 hpf for control and mdk2-injected embryos), which accounts for the apparently weaker hoxC10 staining. EXPRESSION / LABELING:
|
|
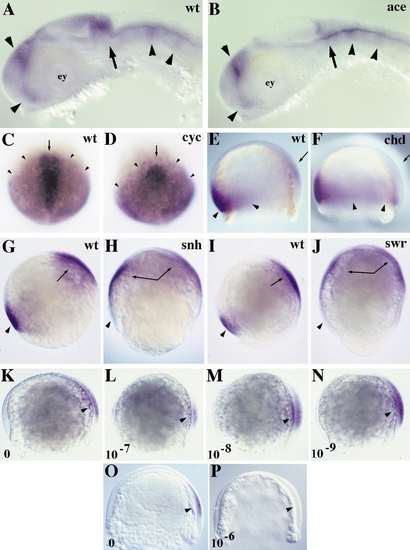
mdk2 expression is independent of FGF8 and nodal-related1 signaling, inhibited by BMP signaling, and upregulated by low doses of exogenously applied RA. (A, B) Lateral views of mdk2 expression in 24-hpf wild-type (wt, A) and acerebellar (ace, B) mutant embryos. mdk2 expression at the MHB (arrow) is absent due to lack of cerebellar structure. Otherwise expression appears unaffected (arrowheads). ey, eye. (C, D) Dorsal views of mdk2 (arrowhead) and gsc (arrow) expression in wt (C) and cyclops (cyc, D) mutant embryos at 90% epiboly. gsc is repressed in cyc embryos, but mdk2 expression appears unaffected. (E–P) Lateral views, dorsal to right. (E, F) Expression of eve1 (arrowhead) and mdk2 (arrow) in wt and chordino (chd) mutant embryos at 70% epiboly. eve1 expression is expanded in chd embryos, but mdk2 is repressed (arrow in F compared to E). (G–J) Expression of eve1 (arrowhead) and mdk2 (arrow) in wt (G, I), snailhouse (snh, H), and swirl (swr, J) mutant embryos at 80% epiboly. While eve1 is repressed in snh and swr, mdk2 expression is ventrally expanded. (K–P) Expression levels of mdk2 (arrowhead) are dependent on the dose of exogenously applied RA. (K–N) Dose response of mdk2 expression in 70% epiboly embryos. RA applied at 10-7 M reduces mdk2 expression (L), whereas lower doses (M, N) increase expression when compared to untreated embryos (K). (O) Expression of mdk2 in an untreated embryo at 70% epiboly. (P) Complete repression of mdk2 expression by treatment with RA at a dose of 10-6 M. |
Reprinted from Developmental Biology, 229(1), Winkler, C. and Moon, R.T., Zebrafish mdk2, a novel secreted midkine, participates in posterior neurogenesis, 102-118, Copyright (2001) with permission from Elsevier. Full text @ Dev. Biol.