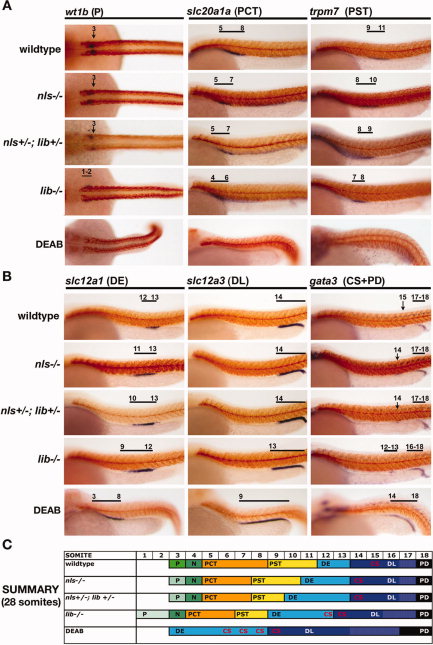
Fig. 5
Fig. 5 Genetic and chemical models of retinoic acid (RA) biosynthesis deficiency exhibit a similar nephron segment phenotype of reduced proximal fates and expanded distal fates, and lib is the most severely affected aldh1a2 mutant. Whole-mount in situ hybridization analysis for nephron segment markers (purple) and mhc (red) at the 28 somite stage in aldh1a2 mutants and diaminobenzaldehyde (DEAB) -treated wild-type embryos. Embryos are shown in lateral views with anterior to the left, with the exception of dorsal views to show podocytes. Black brackets and arrows indicated expression domains, and numbers correspond to somite position. A,B: Proximal fates of podocytes, the proximal convoluted tubule (PCT) and proximal straight tubule (PST), were all reduced in the absence of RA biosynthesis (A), whereas distal fates of the distal early (DE), distal late (DL), corpuscle of Stannius (CS), and pronephric duct (PD) were expanded (B). C: Summary of nephron segmentation with respect to embryo somite number. Each color represents a different epithelial population: podocytes (green), PCT (orange), PST (yellow), DE (light blue), CS (red lettering indicates location among nephron tubule cells), DL (dark blue), and PD (black), and the overlap between DL and PD-expressed genes is indicated in white. The nephron segment phenotypes among RA-deficient zebrafish embryos showed a graded severity such that: nls homozygote < nls/lib compound heterozygotes < lib homozygotes < DEAB-treated wild-type embryos.