|
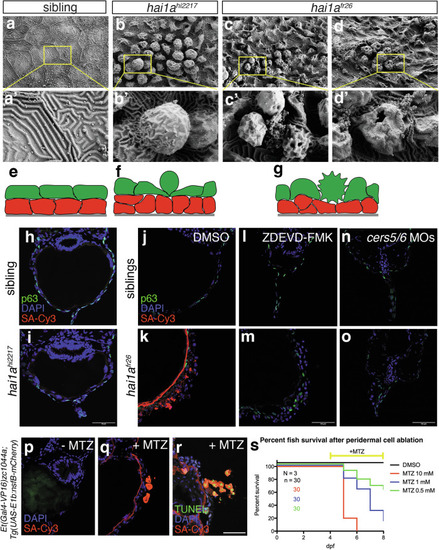
Embryo death is caused by apoptosis-induced loss of epidermal barrier. a–d Scanning electron microscopy images of the epidermal surface of sibling control (a), hai1ahi2217 hypomorphs (b), and hai1afr26 amorph mutants (c, d). e–g Cartoons of the effects of hai1a mutation on epidermal morphology (basal keratinocytes in red, peridermal cells in green, underlying basement membrane in grey). h, i Biotin epidermal barrier assay on transverse cryosections in 4 dpf sibling control (h) and hai1ahi2217 (i) mutants. Basal keratinocytes are labelled with p63 (green), nuclei using DAPI (blue), and biotin using streptavidin-Cy3 (SA-Cy3, red). Scale bar = 50 μm. j, k Biotin epidermal barrier assay on transverse cryosections in 4 dpf sibling control (j) and hai1afr26 (k) mutants. Basal keratinocytes are labelled with p63 (green), nuclei using DAPI (blue), and biotin using streptavidin-Cy3 (SA-Cy3, red). Scale bar = 50 μm. l–o Biotin epidermal barrier assay on transverse cryosections in 4 dpf sibling control (l, n) and hai1afr26 (m, o) mutants, treated with caspase inhibitor ZDEVD-FMK (l, m) or injected with cers5 and cers6 MOs (n, o). Basal keratinocytes are labelled with p63 (green), nuclei using DAPI (blue), and biotin using streptavidin-Cy3 (SA-Cy3, red). Scale bar = 50 μm. p–r Biotin epidermal barrier assay upon peridermal cell ablation, without (p) or with (q) the prodrug metronidazole (MTZ), and using TUNEL to label dying cells (r). Nuclei are labelled using DAPI (blue), biotin using streptavidin-Cy3 (SA-Cy3, red), and dying cells using TUNEL-fluorescein (green). Scale bar = 50 μm. s Survival curves of control (DMSO) or MTZ-treated fish at different concentrations, with treatments starting from 4 dpf.
|