- Title
-
STW 5 Herbal Preparation Modulates Wnt3a and Claudin 1 Gene Expression in Zebrafish IBS-like Model
- Authors
- Piccione, M., Facchinello, N., Schrenk, S., Gasparella, M., Pathak, S., Ammar, R.M., Rabini, S., Dalla Valle, L., Di Liddo, R.
- Source
- Full text @ Pharmaceuticals (Basel)
|
Macroscopic features of gut explants from zebrafish under (A) resting conditions, (B) after 8 h of treatment with 0.5% dextran sodium sulfate (DSS), (C) after 16 h of treatment with 0.12 mg/Ml STW 5, and (D) after DSS + STW 5 treatment. (E–G) Tissue sections of zebrafish gut (bulb) stained with Alcian blue showing the distribution and density of goblet cells (blue) under resting conditions, DSS- or DSS+STW5-treatment. (H–J) Relative number of goblet cells in bulb, midgut, and hindgut explants. The quantification was performed using five different slides of each part of the intestine from three animals (n = 15 sections total for bulb, midgut, and hindgut). Data are expressed as the mean number of goblet cells ± standard deviation (SD). Statistical significance was calculated using the Wilcoxon test comparing DSS-treated or DSS+STW 5–treated samples with controls. * p ≤ 0.05; ** p ≤ 0.01. DSS+STW 5–treated samples vs. DSS (• p ≤ 0.05; •• p ≤ 0.01). |
|
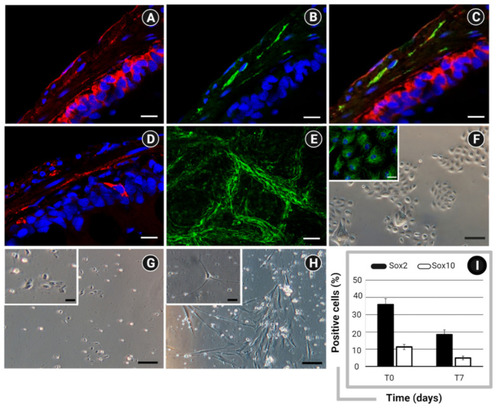
Immunofluorescent staining showing the expression of (A) Frizzled 9 (red) within the epithelial and neuromuscular compartments; (B) PAN neuronal marker (green) within the neuromuscular layer; and (C) both markers (Frizzled 9 and PAN neuronal marker) in colocalization (yellow); and (D) Wnt3a within the epithelium and neuromuscular layer. Nuclear counterstaining was performed using DAPI (blue). (E) Whole-mount immunofluorescent staining of zebrafish ENS network by detection of PAN neuronal marker (green) Bar: 50 µm. Optical microscopy of (F) primary intestinal epithelial cells, characterized by the expression of Pan-cytokeratin (left corner, green); ENS cultures at (G) 24 h and (H) 7 days from isolation. Bar: 100 µm (200 µm in left corner). (I) Flow cytometry analysis of Sox2 and Sox10 in ENS cells at the time of isolation (T0) and after culturing for 7 days (T7). Data are expressed as percentage (%) of positive cells ± standard deviation (SD). Created with BioRender.com, accessed on 31 October 2021. |
|
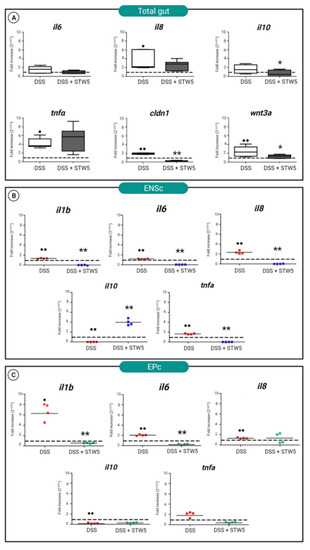
(A) Quantitative PCR analysis of il6, il8, il10, tnfα, wnt3a, and cldn1 genes in explanted zebrafish total guts. The tissues were explanted from animals treated for 8 h with DSS (controls) or incubated with STW 5 for 16 h after DSS stimulation. qRT-PCR of il6, il8, il10, and tnfα in (B) ENS cultures (ENSc), and (C) epithelial cells (EPc). Expression levels were determined as relative to the expression of the housekeeping gene ef1α, normalized to controls, and calculated using the ΔΔCt method based on the equation 2–ΔΔCt ± standard deviation. In the graphs, the expression level of controls was set to 1 and indicated with a dotted line. Statistical significance was calculated using the Wilcoxon test. Samples vs. controls (• p ≤ 0.05; •• p ≤ 0.01). Samples vs. DSS (* p ≤ 0.05; ** p ≤ 0.01). |
|
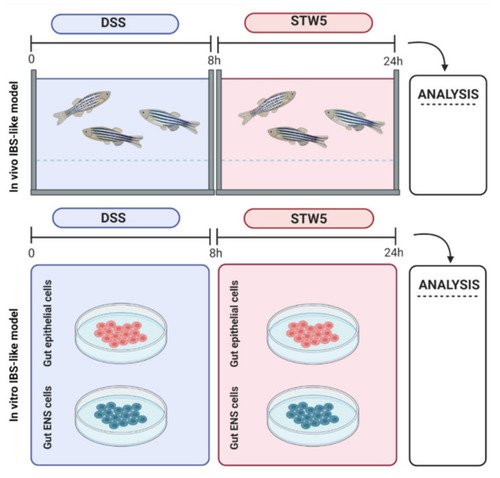
Experimental scheme to obtain in vivo and in vitro models of irritable bowel syndrome (IBS) (created with BioRender.com, accessed on 21 May 2021). (Top) In vivo treatment: IBS was induced in zebrafish with 0.5% dextran sodium sulfate (DSS) in the water. The water was exchanged after 8 h, a mixture of nine medicinal herbs (STW 5) was added to a concentration of 0.12 mg/mL, (dissolved in water), and the fish were kept in this for 16 h. The gut was explanted for examination. (Bottom) In vitro treatment: The intestine from wild-type zebrafish was dissected and digested to obtain cells from the epithelial compartment (EPc) and myenteric plexus (ENSc). After induction of IBS-like inflammation with 0.5% DSS for 8 h, EPc and ENSc were treated for 16 h with 0.12 mg/mL ethanol-free, lyophilized STW 5 resuspended in FBS-deprived culture medium. |