- Title
-
The roles of SMYD4 in epigenetic regulation of cardiac development in zebrafish
- Authors
- Xiao, D., Wang, H., Hao, L., Guo, X., Ma, X., Qian, Y., Chen, H., Ma, J., Zhang, J., Sheng, W., Shou, W., Huang, G., Ma, D.
- Source
- Full text @ PLoS Genet.
|
Quantitative RT-PCR and whole-mount in situ hybridization and qRT-PCR analyses of smyd4 expression in zebrafish early embryogenesis. (A) The overall expression levels of smyd4 at 0.2, 2, 4, 8, 10, 24, 48 and 72 hpf were analyzed using qRT-PCR. smyd4 displayed its highest expression levels during the blastula stage (4 hpf) and was significantly decreased at 48 hpf. At 72 hpf, there was a quick reactivation of smyd4 expression. (B-C) During the mid-gastrula stage (8 hpf), smyd4 was ubiquitously expressed in the mid-gastrula embryo (8 hpf). (D, E) smyd4 was expressed ubiquitously at 10 hpf, with the highest expression levels found at the polster. (F, G) smyd4 was abundant in the heart and blood vessel at 24 hpf. (H-M) Lateral and frontal view of smyd4 expression at 48 hpf. smyd4 became more enriched in the developing hearts at 48 hpf, and the ventricle had a higher expression level than that in the atrium. A smyd4 sense probe was used as the negative control for the in situ hybridization analysis. EXPRESSION / LABELING:
|
|
The generation of smyd4–deficient zebrafish using the CRISPR/Cas9 technology. (A) Schematic diagram of the smyd4 gene structure and the sgRNA3 sequence for the exon 6 targeting site. The sgRNAs were injected into the embryos with Cas9 mRNA at the single-cell stage; (B) The targeting site of sgRNA3 was well-conserved from zebrafish to humans; (C) Sanger sequencing confirmed the insertion mutation (c.1629_1630inGAATAATACTG; p.Leu544Glufs*1); (D) A schematic diagram of the insertion mutation. The smyd4 protein contains four functional domains: two TPR domains (green), one MYND domain (yellow), and one SET domain (purple). The insertion mutation created a truncated mutant protein that lacks the entire C-terminus, which contained the key functional domain TPR2; (E) qRT-PCR confirmation that smyd4 expression is significantly decreased in MZsmyd4L544Efs*1 embryos; (F) Whole-mount in situ hybridization analysis confirmed the loss of smyd4 expression in MZsmyd4L544Efs*1 embryos at 72 hpf. |
|
Phenotypical analyses of MZsmyd4L544Efs*1 embryos. (A) Representative images of the MZsmyd4L544Efs*1 and control embryos at 48 hpf, exhibiting congested blood flow in the ventral vein; (B) Representative images of the MZsmyd4L544Efs*1 and control embryos at 96 hpf, displaying severe pericardial edema; (C) Representative images of the MZsmyd4L544Efs*1 and control hearts at 72 hpf, demonstrating abnormal cardiac morphology and left-right looping defects; (D) Significantly more MZsmyd4L544Efs*1 embryos had abnormal cardiac development than control fish Tg (cmcl2:GFP) (61% ± 4.0% mutants vs. 14% ± 4.6% controls, n = 100, p < 0.01**); (E) Comparison of left-right patterning defects in the MZsmyd4L544Efs*1 (n = 100) and control (n = 100) embryos (green: normal L-loop; blue: straight; dark blue: D-loop). Only 10% of the control embryos showed an abnormal “straight” cardiac left-right asymmetry pattern. However, 48% of MZsmyd4L544Efs*1 embryos exhibited a “straight” left-right asymmetry pattern, and 11% showed a “D-loop” left-right asymmetry pattern; (F) Representative confocal images of the ventricular wall and 3D re-construction images of the MZsmyd4L544Efs*1 and control hearts at 96 hpf. The confocal scanned images demonstrated hypoplastic wall with significant reduction of trabeculation in MZsmyd4L544Efs*1 ventricle when compared to the wild-type control ventricle. The 3D-reconstruction of the hearts confirmed left-right looping defects in MZsmyd4L544Efs*1 hearts (For a better view of 3D-structures, see supplemental material S1 and S2 Movies). (G) Histological sections and H&E staining images of the MZsmyd4L544Efs*1 and control hearts at 96 hpf, demonstrating abnormal ventricular walls and reduced trabeculation; (H) The quantitative analysis of the number of cardiomyocytes in the ventricle displayed that MZsmyd4 L544Efs*1 had significantly reduced cardiomyocytes (p<0.01, **). |
|
Phenotypical analyses of MZsmyd4L544Efs*1 embryos. (A) The p-H3(S10) immunofluorescence staining showed decreased cell proliferative activity in MZsmyd4L544Efs*1 cardiomyocytes at 48 hpf. The number of p-H3 (S10) positive cells in the MZsmyd4L544Efs*1 ventricles was significantly decreased when compared to the control group (p<0.01, **); (B) The abnormal ventricular morphology of the adult MZsmyd4L544Efs*1 hearts compared to the morphology of control adult hearts; (C) Histological sections and H&E staining images of the adult MZsmyd4L544Efs*1 and control hearts, demonstrating significant thickening of the compact ventricular wall. (The red arrows showed that in the similar compact zone of the ventricle). The quantitative analysis displayed that the thickness of the ventricle compact zone was significantly increased in adult MZsmyd4 L544Efs*1 hearts compared to Tg(cmcl2:GFP) (p<0.05, *). PHENOTYPE:
|
|
SMYD4 interacts with HDAC1. (A) Immunofluorescence staining and representative confocal images of the sub-cellular localization of SMYD4 in HL-1 cells. SMYD4 was found in both the nucleus and cytoplasm. The experimental groups without primary anti-SMYD4 antibody incubation served as negative controls; (B) Co-IP/western blotting analysis to confirm the interaction between SMYD4 and HDAC1. SMYD4flag was overexpressed in HL-1 cells. The cell extracts were immunoprecipitated with an anti-FLAG affinity gel, followed by western blotting analysis; (C) The interaction between SMYD4 and HDAC1 was further confirmed by Co-IP/western blotting analysis in HEK293T cells; (D-E) Using a Co-IP/western blotting assay to map the domains in SMYD4 that are responsible for the interaction with HDAC1. The MYND domain of SMYD4 was found to be critical for this interaction; (F) Aberrant histone modifications in MZsmyd4L544Efs*1 embryos at 48 hpf. Specifically, H3K4me2 and H3K4me3 were significantly reduced, and H3K4me was increased, while H3K9me3 and H3K27me3 were not affected, suggesting that SMYD4 is a H3K4-specific methyltransferase. H3K4ac, H3K9ac, H3K14ac, and H3K27ac were significantly abolished in MZsmyd4L544Efs*1 mutants, suggesting the increased activity of HDAC1 in MZsmyd4L544Efs*1 mutants and confirming SMYD4 as an important negative regulator of HDAC1 function; (G) The semi-quantitative analysis of the histone modifications changes in MZsmyd4L544Efs*1 mutants. (p<0.05 *, p<0.01 **). PHENOTYPE:
|
|
SMYD4(G345D) contributes to cardiac abnormalities. (A) Representative cardiac morphology of wild-type embryos injected with either wild-type smyd4 or smyd4(G295D) mRNAs; (B) Representative cardiac morphology of MZsmyd4L544Efs*1 embryos injected with either wild-type smyd4 or smyd4(G295D); (C) Summary of the observations from (A) and (B). A total of 50 fish were screened. |
|
Heterozygous breeding scheme generated some abnormal cardiac situs ambiguus and hypoplastic hearts in homozygous and heterozygous mutants. PHENOTYPE:
|
|
Representative images for analyzing the number of cardiomyocytes in MZsmyd4L544Efs*1 mutant and control hearts. The confocal images of the largest section of the ventricle of MZsmyd4 L544Efs*1 and Tg(cmcl2:GFP) hearts at 96 hpf. PHENOTYPE:
|
|
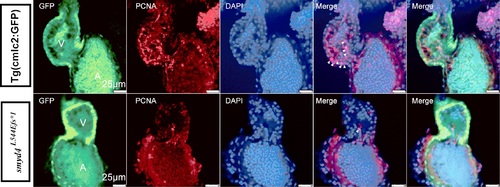
The PCNA immunofluorescence staining showed decreased cell proliferative activity in MZsmyd4L544Efs*1 cardiomyocytes at 48 hpf. PHENOTYPE:
|
|
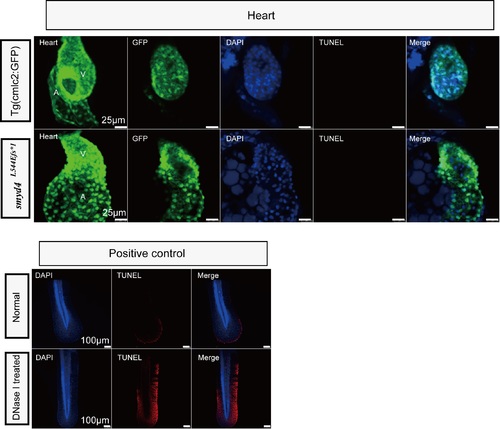
Cellular apoptosis was not seen in the MZsmyd4L544Efs*1 ventricles at 48 hpf. TUNEL assays showed that there is no apoptosis in neither control hearts and MZsmyd4L544Efs*1 hearts (Upper panel). The positive TUNEL signals (red fluorescence) in the tail fin and embryos at 48 hpf treated with DNase I were used as positive controls for the analysis (Lower panel). PHENOTYPE:
|
|
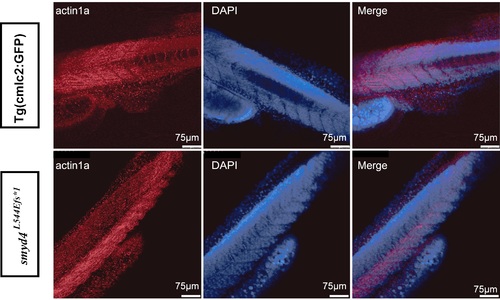
Confocal images of fluorescence immunostaining of actin1a, co-stained with DAPI. The skeletal muscle structures of MZsmyd4L544Efs*1 mutants appeared to be normal at 72 hpf. PHENOTYPE:
|