- Title
-
Characterization of Tetratricopeptide Repeat-Containing Proteins Critical for Cilia Formation and Function
- Authors
- Xu, Y., Cao, J., Huang, S., Feng, D., Zhang, W., Zhu, X., Yan, X.
- Source
- Full text @ PLoS One
|
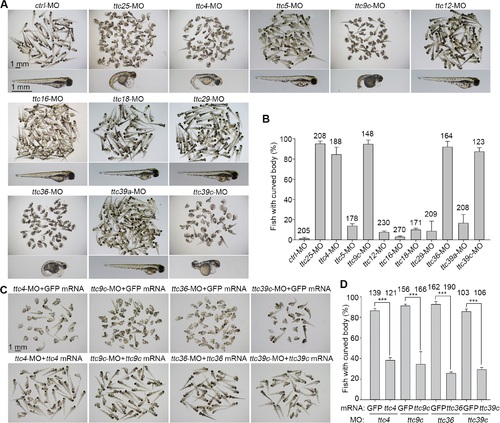
Screen for cilia-related TTC genes in zebrafish. (A) The morphologies of zebrafish morphants at 72 hpf. Note that the morphants of ttc4, -9c, -25, -36, and -39c displayed severe body curving. (B) Quantification results for the curved body phenotype. Three independent experiments were performed. (C) and (D) Rescue experiments for the ttc morphants. The indicated MO was coinjected with either the corresponding MO-resistant mRNA or GFP mRNA into zebrafish embryos. The body curvature phenotype was assayed at 72 hpf. The numbers of embryos counted are listed over the histograms. Statistical results are from three independent experiments. Student?s t-test, ***P < 0.001. Error bars represent s.d. PHENOTYPE:
|
|
Additional phenotypes of the zebrafish morphants of ttc4, -9c, -25, -36, and -39c. (A) Typical otolith morphologies at 72 hpf. The otic vesicle normally contains two otoliths as shown. Otoliths that show differences in number, size and/or position were considered as abnormal. (B) Quantification results for abnormal otoliths in the indicated morphants. (C) and (D) Hydrocephalus formation (arrows in the bright-field images and asterisks in the histochemical sections) in the indicated morphants at 72 hpf. (E) and (F) The ttc25 and ttc4 morphants tended to develop pronephric cysts (arrows in the bright-field images and asterisks in the histochemical sections) at 72 hpf. (G) and (H) Left-right asymmetry patterning was disturbed in the indicated ttc morphants at 30 hpf. The cmlc2 probe was used to label the heart tube (arrows) in whole-mount in situ hybridization. All the quantification results were based on three independent experiments. Student?s t-test, ***P < 0.001. Error bars represent s.d. PHENOTYPE:
|
|
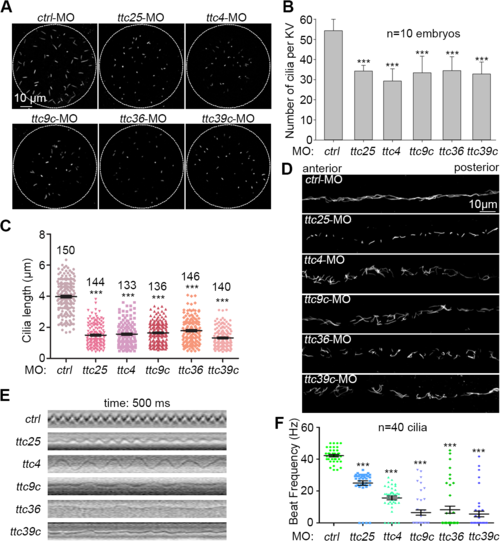
Examinations on cilia in the Kupffer?s vesicle and pronephric duct. (A-C) Both cilia number and cilia length in the Kupffer?s vesicle (KV) at the 7-somite stage were reduced in the indicated ttc morphants. Cilia were labeled using antibody against acetylated tubulin. The quantification results were based on three independent experiments. Student?s t-test, ***P < 0.001. Error bars represent s.d. (D) Typical morphology of cilia in pronephric ducts at 24 hpf. (E) and (F) Cilia motility in the pronephric duct at 60 hpf. The kymographs showed trajectories of the cilia marked with red line in S1, S2, S3, S4, S5 and S6 Videos. Cilia beat frequencies (CBFs) were measured at 60 hpf (40 cilia from 4 morphants for each gene). Student?s t-test, ***P < 0.001. Error bars represent s.e.m. PHENOTYPE:
|
|
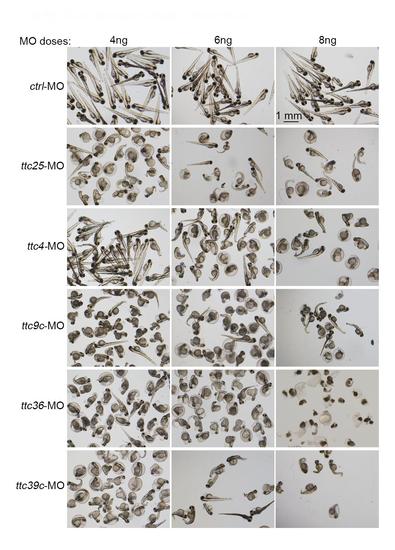
Dose-dependent effects of the ttc MOs. One-cell-stage zebrafish embryos were injected with the indicated dose of the MOs. The images were taken at 72 hpf. |
|
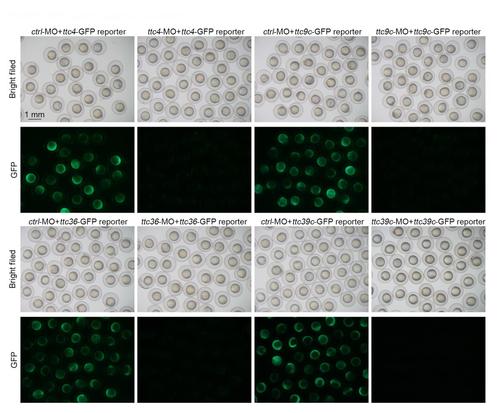
GFP reporter assays for MO efficiency. The indicated ttc-GFP reporter mRNAs were coinjected with the corresponding MOs or ctrl-MO into zebrafish embryos. The brightfield and fluorescent images were taken at 12 hpf. The expression of GFP was suppressed by the ttc MOs but not ctrl-MO. |