- Title
-
Patterning the cone mosaic array in zebrafish retina requires specification of ultraviolet-sensitive cones
- Authors
- Raymond, P.A., Colvin, S.M., Jabeen, Z., Nagashima, M., Barthel, L.K., Hadidjojo, J., Popova, L., Pejaver, V.R., and Lubensky, D.K.
- Source
- Full text @ PLoS One
|
Rod photoreceptors develop rapidly and UV cones are missing in tbx2b mutants. A) Isolated, larval rh1:EGFP eye at 3 days post-fertilization (dpf) viewed from the scleral aspect. Rod photoreceptors are green and immunostaining for the apical junctional marker, Zonula Occludens-1 (ZO-1) is in white. Dorsal is up; the optic nerve appears as a small white ring ventral to the center. B) Mutant tbx2b; rh1:EGFP larval eye at 3 dpf viewed from the scleral side. Note increased number of rods, especially in the ventral retina. C, D) Higher magnifications of central retina in wild-type and mutant eyes, respectively. E) Isolated, larval sws1:EGFP eye and F) tbx2b; sws1:EGFP eye at 4 dpf, viewed from the scleral aspect. Cones expressing the UV opsin reporter are pseudocolored magenta. The lens, outlined with a dashed white line, shows background fluorescence in F, due to the longer exposure time required to capture immunofluorescence of the few scattered UV cones. Scale bars: = 50 μm (A,B); 10 μm (C,D); 50 μm (E,F). EXPRESSION / LABELING:
PHENOTYPE:
|
|
The rod phenotype in tbx2b mutant embryos is partially compensated in adults. Rods and cones were counted in wild-type (wt) and tbx2b mutant (mut) embryos at 3 days post-fertilization (dpf) and in adult retinas and plotted as planimetric density (#/103 µm2). Photoreceptor profiles were identified by ZO-1 immunostaining and the rod opsin transgenic reporter was used to distinguish rods. Means ± standard deviation are plotted for n = 11 samples from 3 retinas (wt embryo), n = 8 samples from 4 retinas (mut embryo), n = 12 samples from 3 retinas (wt and mutant adult). *** p<0.001; ** p<0.01. PHENOTYPE:
|
|
Variable number and distribution of UV cones in adult tbx2b mutants. A, B) Flat-mounted retinas from sws1:EGFP wild-type fish; the optic disc is indicated by an asterisk; dorsal is up. C–H) Six examples of tbx2b; sws1:EGFP mutant retinas. Residual UV cones are typically at or near the dorsal margin (curved white arrows) and immediately dorsal to the optic disc (arrowhead). Scale bar: 500 μm (A–H). |
|
UV cones in tbx2b mutants are dysmorphic. A) Retinal cryosection from a sws1:EGFP adult zebrafish; UV cones are magenta, red-green double cones are immunostained with the specific marker, zpr1 (yellow), and nuclei are stained with the fluorescent dye, Hoechst (grey). The central panel is an overlay of the epifluorescent channel (UV cones) and transmitted light (differential interference contrast). Nuclei of red-green double cones (arrows), and blue cones (not indicated), are radially elongated and displaced apical to the outer limiting membrane (dashed line), whereas nuclei of UV cones are triangular in shape (circles) and positioned basal to the outer limiting membrane. B, C) In tbx2b; sws1:EGFP mutants, the morphology of red-green double cones (zpr1, yellow) and their nuclear position (white arrows) is normal, whereas the nuclei of UV cones (circles) are displaced apically beyond the outer limiting membrane (dashed line). One UV cone (black arrow) is collapsed. D, E) 3D volume renderings of UV cones near the retinal margin in wild-type, sws1:EGFP fish. Like all photoreceptors UV cones have a single, bulbous axonal terminal (cone pedicle, arrow). D) Many UV cones in tbx2b; sws1:EGFP mutant adults have a bifurcated axon and two pedicles (arrows). Scale bar: 20 μm (A,B,C). |
|
The regular cone mosaic lattice is disrupted in tbx2b mutants. A, C) Cone mosaic lattice in the dorsal retina (A) and ventral retina (C) visualized in retinal flat-mounts of adult, wild-type, double transgenic (UV and blue cone reporter) fish. Cell profiles are stained with anti-ZO-1 (white). Rows of UV cones (magenta) and blue cones (blue) alternate with red-green double cones. Smaller, rounded ZO-1 profiles represent rods and irregular profiles are Müller glial processes. The inset shows a cartoon of the cone pattern; rods are represented by black dots. Rarely, cones are missing from the lattice (indicated by blue star in A and magenta star in C). The yellow circle encloses a UV cone surrounded by rods. B) Retinal flat-mount near the dorsal retinal margin of a tbx2b mutant with the UV cone reporter. Immunolabeling with ZO-1 (white) reveals absence of a rectilinear order, independent of the presence of the unusually large UV cones (magenta). This fish did not carry the blue opsin reporter transgene. D) Retinal flat-mount from ventral retina in a tbx2b mutant double transgenic: blue cones (blue) and red-green double cones (white profiles); UV cones are absent from the ventral retina. The red box delimits a region used for cone cell counts. E) Retinal flat-mount from wild-type, rod reporter transgenic fish with rods (green) and cones (white profiles). F) Retinal flat-mount from tbx2b mutant adult with rod reporter. Scale bars: 20 μm (A,B,C,E,F) and 20 μm (D). |
|
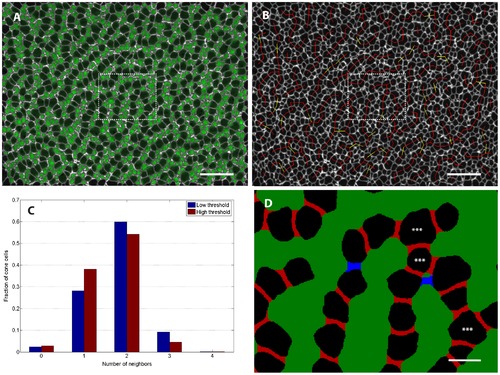
Infrequent three-fold coordination of mutant retina cone photoreceptors suggests strongly directional interaction. (See Methods section for details of image processing.) A) To identify rods, we superimposed on the ZO-1 label (white) the signal in the rod GFP reporter channel (green) from a single z-slice per cell chosen to coincide with the level of the OLM. B) Same retina as panel A. Adjacent cone photoreceptors in ventral-temporal retina of adult tbx2b mutant. Red lines join pairs of cones that are classified as adjacent at both low and high threshold (see Methods), while yellow lines indicate pairs that were identified as adjacent only with the less stringent threshold. (C) Histogram showing the fraction of cone photoreceptors with the specified number of identified neighbors in a sample of ventral-temporal retina. (See Table 1 for additional data.) D) Segmented image from the region outlined by the dashed box in panels A and B. Pixels between adjacent cells that were filled in by the morphological closing procedure are colored either red (high threshold) or blue (low threshold); at this magnification, 1 pixel corresponds to 0.1 μm. The two cells flanking each red region were classified as adjacent at the high threshold, but the blue region is too long (along the axis joining the centers of the two cells) and too narrow (along the orthogonal axis) to meet the adjacency criteria at the high threshold. The blue region meets the adjacency criteria at the low threshold. Stars indicate cone cells that are three-fold coordinated, i.e. have 3 adjacent cells. Scale bars are 20 μm for A and B, and 4 μm for D. EXPRESSION / LABELING:
|
|
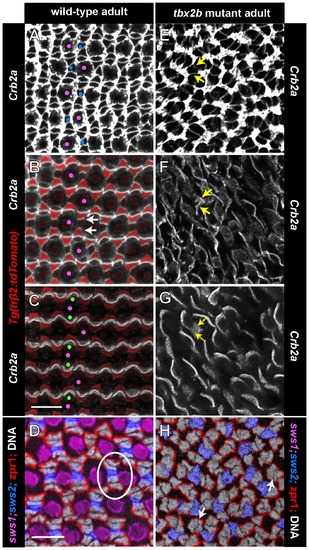
Planar polarized distribution of Crumbs2b-mediated adhesion organizes differentiating cones into linear columns, and column fragments are retained in tbx2b mutants. A, C, E) Confocal images of retinal flat-mounts immunostained for ZO-1 (white or cyan) and focused at the level of the OLM. B, D, F) Confocal images of retinal flat-mounts immunostained for Crb2b and focused at the level of the inner segments of cone photoreceptors. A) Retinal margin from a wild-type adult double transgenic (UV and blue cone reporters); proliferative germinal zone at the right edge. Cohorts of cones generated nearly synchronously at the germinal zone form straight columns; rows of blue cones and UV cones alternate with rows of red/green double cones (ZO-1 profiles). Expression of the blue cone reporter (blue) is initiated in differentiating cones (blue arrows) as the column emerges from the germinal zone. Cone profiles in the predicted position of UV cones (magenta arrows) emerge from the germinal zone prior to onset of the UV cone reporter. B) Retinal margin from a wild-type adult with the blue cone reporter; some UV cones, identified by their position in the mosaic lattice, are indicated by magenta dots. Crb2b immunostaining (yellow) is localized to the flattened boundaries between inner segments of five adjacent cones (green-red-blue-red-green) along a column, but not between columns; UV cones (magenta dots) are in the gaps separating the pentamers. Polarized Crb2b is in linear segments oriented orthogonal to the retinal margin in the earliest cone columns (arrows). C, D) Retinal margin from a tbx2b mutant double immunolabeled for ZO-1 (C) in white and Crb2b (D) in yellow; inset shows overlay of ZO-1 and Crb2b. Note that Crb2b is already polarized approximately orthogonal to the margin in the earliest cones. E) Dorsal retinal margin from a double transgenic (UV and blue cone reporters) tbx2b mutant adult with ZO-1 (white). F) Double immunolabeled tbx2b mutant. Left panel: ZO-1 (cyan) at the level of the OLM superimposed on Crb2b (yellow) at the level of the inner segments (3–6 μm apical to the OLM). Right panel: Level of Crb2b only. Dashes outline column fragments. Scale bars: 10 μm (A) and (B–F). EXPRESSION / LABELING:
|
|
Planar polarized distribution of Crumbs2a is retained at the level of cone inner segments in tbx2b mutants despite loss of pentameric organization of red, green, and blue cones. Panels A–C and E–G are confocal images of retinal flat-mounts immunostained for Crb2a (white) in a wild-type, transgenic (trβ2:tdTomato) fish (A–C) in which red cones express the tdTomato reporter (red), and a tbx2b mutant (E–G), which expressed the sws1:EGFP reporter, although no UV cones are present in the region imaged. Each panel is a projection of 3 to 12 optical sections selected from a complete, vertical z-stack of the Crb2a immunolabeling: panels A, E are at the level of the OLM; panels B, F are 2–4 μm apical to the OLM, at the level of the red, green, and blue cone inner segments; panels C, G are 6–7 µm apical to the OLM, at the level of the red and green inner segments. A) Selected UV and blue cones are identified with magenta and blue dots, respectively, based on their relative sizes and positions in the mosaic array at the OLM. B) At the level of inner segments, Crb2a is preferentially localized to interfaces between red/green double cones and between a blue cone and the two flanking red cones (arrows). The inner segments of UV cones (magenta dots) are short and do not extend this far apically. C) Only the longest cones (red/green double cones) extend to this level, and Crb2a remains polarized. E) Retinal flat-mount from a tbx2b mutant at the OLM. This mutant did not carry the sws2:mCherry transgene, so the blue cones cannot be distinguished from the red and green cones. F, G) Short, curved segments of Crb2a, which resemble red/green double cone interfaces in the wild-type extend to the most apical level (G), but other Crb2a segments (yellow arrows) are at the intermediate level (F) but not higher (G). These may represent blue cones. Panels D and H show tangential cryosections through the level of nuclei of red, green, and blue cones; nuclei are stained with Hoechst (grey); red and green cones are immunolabeled with the membrane-associated, specific antibody, zpr1 (red). Both the wild-type (D) and tbx2b mutant (H) fish carried the UV and blue cone transgenes, sws1:EGFP (magenta) and sws2:mCherry (blue), although this retinal region lacked UV cones in the mutant. A single pentameric unit is encircled by an oval (D) and white arrows point to the interface between red and green double cone pairs. Scale bars: 10 μm (A, B, C, E, F, G) and (D, H). EXPRESSION / LABELING:
|
|
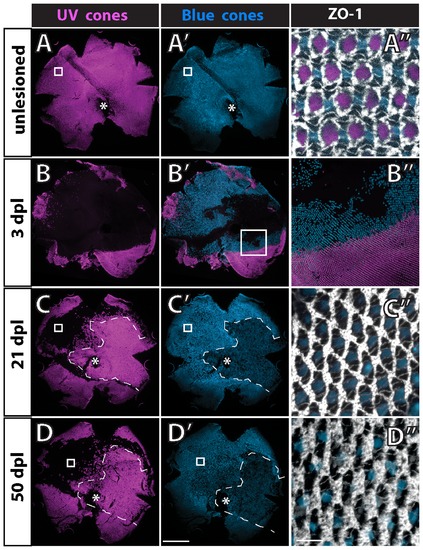
The mosaic pattern is not disrupted after selective ablation of UV cones in adult wild-type fish. Retinal flat-mounts from a double transgenic adult fish (UV and blue cone reporters) immunostained for ZO-1. Ventral is down; optic disc (*); boxed area in panels to the right. A) Control retina, UV cones; A2) blue cones; A3) ZO-1, UV and blue. B) Transgenic fish exposed to intense light, 3 days post-lesion (dpl), UV cones; B2) blue and UV cones; B3) boundary of ventral region in which no cones are ablated (lower right) and only UV cones but not blue cones are ablated (upper right). C) At 21 dpl, all cone types, including UV cones, were ablated and have regenerated within the central and temporal retina in the region enclosed by dashes; C2) blue cones have regenerated within the region enclosed by dashes; C3) in the boxed region where only UV cones were ablated they do not regenerate. D) At 50 dpl, UV cones (along with other cone types) have regenerated within the region enclosed by dashes; D2) blue cones have regenerated within the region enclosed by dashes; D3) rods continue to accumulate in the spaces previously occupied by UV cones in the region where they were selectively ablated and fail to regenerate. Scale bars: 500 μm (A, A2 through D, D2); 10 μm (A3 through D3). |