- Title
-
Key Developmental Regulators Suggest Multiple Origins of Pancreatic Beta Cell Regeneration
- Authors
- Wang, H., Wei, X., Shi, W., He, J., Luo, L.
- Source
- Full text @ Zebrafish
|
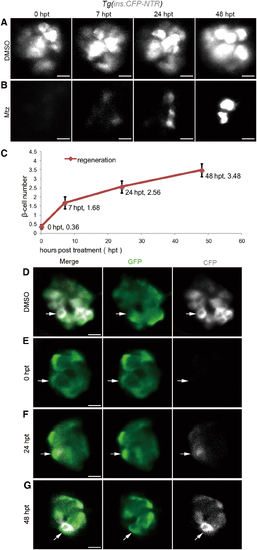
β cell regeneration after a near-total ablation. (A, B) Representative 3D projections of β cells in the primary islet of Tg(ins:CFP-NTR) larvae from 0 to 48 hpt. The larvae treated with DMSO in the control group showed steady expression of CFP in β cells of the primary islet (A, n = 30/30; scale bar, 10 μm). The larvae treated with Mtz showed 0 β cell at 0 hpt. Nascent β cells emerged as regeneration proceeded (B, n = 32/37; scale bar, 10 μm). (C) Quantification of nascent β cells at different time points post-treatment. The mean values of β cell number are indicated (n = 25 for each time point. Data are expressed as mean ± s.e.m.). (D–G) Time series of merged and single-channel confocal planes of primary islets treated with DMSO or Mtz. Tg(ins:CFP-NTR) was used to label β cells blue with CFP and perform ablation. Tg(neurod:EGFP) marked neurod+ cells with GFP. All β cells were neurod-positive in both the DMSO-treated control group and the Mtz-treated group. A neurod+ins- cell at 0 hpt showed positive CFP signal in the following 48 h (white arrows) indicating its commitment to β cell fate (D, n = 23/23; E, n = 17/19; scale bar, 10 μm). 3D, three-dimensional; CFP, cyan fluorescent protein; DMSO, dimethyl sulfoxide; GFP, green fluorescent protein; Mtz, metronidazole; NTR, nitroreductase; s.e.m., standard error of the mean. Color images are available online. |
|
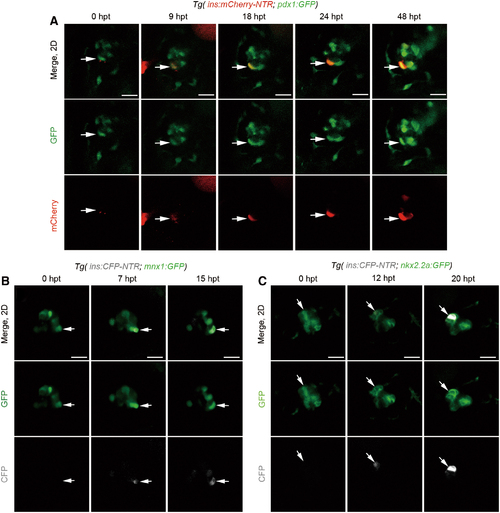
Nascent β cells emerged from pre-existing pdx1+, mnx1+, or nkx2.2a+ cells. (A) A set of real-time imaging of a regenerating β cell (white arrows) in the primary islet after Mtz treatment using the Tg(ins:mCherry-NTR; pdx1:GFP) transgenic line (n = 19/23). Merged and single-channel confocal planes showed a pdx1+ins- cell (0 hpt, white arrow) committed to the β cell fate in the next 48 h. β cell maturation was noted by the onset of mCherry fluorescence indicating insulin expression since 9 hpt. mCherry expression was getting steadily stronger from 18 to 48 hpt, indicating the maintenance of β cell identity. (B, C) Time course of a regenerating β cell (white arrows) in the primary islet after Mtz treatment using the Tg(mnx1:GFP; ins:CFP-NTR) or Tg(nkx2.2a:GFP; ins:CFP-NTR), respectively. A GFP+CFP− cell (white arrow) showed commitment to the β cell fate within several hours after removal of Mtz (B, n = 21/25; C, n = 18/26; scale bar, 20 μm). Color images are available online. |
|
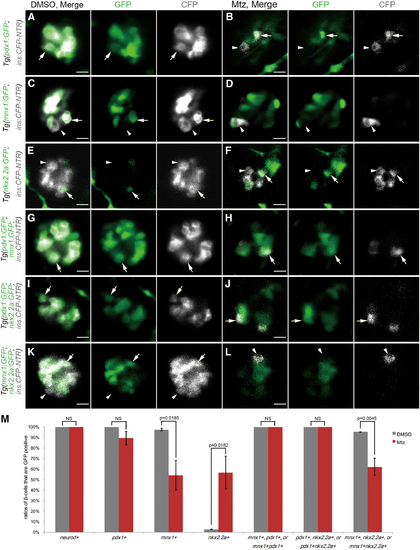
Variant key development regulators highly suggest different cellular origins of β cell regeneration. (A–L) Merged and single-channel confocal planes of primary islets after DMSO or Mtz treatment. Tg(ins:CFP-NTR) was used to label β cell blue with CFP and perform ablation. Single or double transgenic lines were combined to mark specific β cell development regulators green with GFP. White arrows indicated GFP-positive β cells. White arrowheads indicated GFP-negative β cells. Scale bar, 10 μm. Tg(pdx1:GFP) marked pdx1+ cells. β cells in the DMSO-treated control group were all pdx1+ (A, n = 35/35). In contrast, some β cells were not pdx1+ in the Mtz-treated group (B, n = 33/38). Tg(mnx1:GFP) marked mnx1+ cells. Not all β cells were mnx1+ in either the control or Mtz-treated groups (C, n = 22/22; D, n = 22/26). Tg(nkx2.2a:GFP) marked nkx2.2a+ cells. Not all β cells were nkx2.2a+ in either control or Mtz-treated groups (E, n = 21/21; F, n = 20/23). Tg(pdx1:GFP; mnx1:GFP) marked pdx1+, mnx1+, or pdx1+mnx1+ cells. All β cells were GFP positive in both the control and Mtz-treated groups (G, n = 34/34; H, n = 37/37). Tg(pdx1:GFP; nkx2.2a:GFP) marked pdx1+, nkx2.2a+, or pdx1+nkx2.2a+ cells. All β cells were GFP positive in both the control and Mtz-treated groups (I, n = 20/20; J, n = 23/25). Tg(mnx1:GFP, nkx2.2a:GFP) marked mnx1+, nkx2.2a+, or mnx1+nkx2.2a+ cells. Not all β cells were GFP positive in either the control or Mtz-treated groups (K, n = 21/21; L, n = 26/29). (M) Quantifications of ratios of β cells that were GFP positive in the primary islets after DMSO or Mtz treatment with listed transgenic lines (DMSO-treated control groups: mnx1+, n = 5; all the other groups, n = 3. Mtz-treated groups: neurod+, n = 4; pdx1+, n = 17; mnx1+, n = 8; nkx2.2+, n = 6; pdx1+, mnx1+, or pdx1+mnx1+, n = 18; pdx1+, nkx2.2a+, or pdx1+nkx2.2a+, n = 5; mnx1+, nkx2.2a+, or mnx1+nkx2.2a+, n = 8. Mean ± s.e.m. All tested by two-tailed Student's t-test.). Color images are available online. |
|
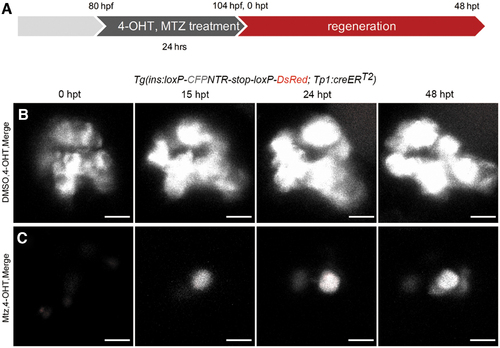
Pancreatic ductal cells did not contribute to nascent β cells. (A) The strategy for Cre/loxP-mediated lineage tracing of pancreatic ductal cells in β cell regeneration. 4-OHT was used in both the DMSO-treated control group and Mtz-treated group to label Notch-responsive pancreatic ductal cells. Mtz was used only in the Mtz-treated group, but not in the DMSO-treated control group. (B, C) 3D projections of islets in control and Mtz treatment groups from 0 to 48 hpt using Tg(Tp1:creERT2; ins:loxP-CFPNTR-stop-loxP-DsRed) transgenic lines. Notch-responsive pancreatic ductal cells should be labeled by DsRed after 4-OHT treatment. Data showed no β cell was positive for DsRed in control group (B, n = 19/19). Nascent β cells were not positive for DsRed, indicating a negative contribution of tp1+ pancreatic ductal cells to the nascent β cells (C, n = 18/23). Three tp1+ red fluorescent cells showed in the (C) 0 hpt were dying/dead β cells with tp1 activated during Mtz treatment. They disappeared thereafter possibly due to cell death. Scale bar, 10 μm. 4-OHT, 4-hydroxy-tamoxifen. Color images are available online. |