- Title
-
Interference with the Cannabinoid Receptor CB1R Results in Miswiring of GnRH3 and AgRP1 Axons in Zebrafish Embryos
- Authors
- Zuccarini, G., D'Atri, I., Cottone, E., Mackie, K., Shainer, I., Gothilf, Y., Provero, P., Bovolin, P., Merlo, G.R.
- Source
- Full text @ Int. J. Mol. Sci.
|
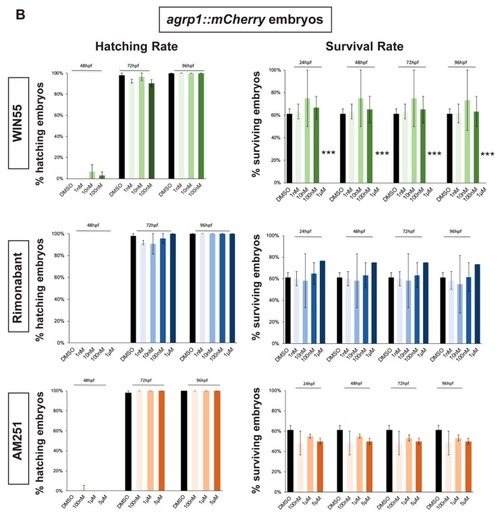
Survival and hatching rates in zebrafish embryos treated with CB1R ligands. ( |
|
Survival and hatching rates in zebrafish embryos treated with CB1R ligands. ( |
|
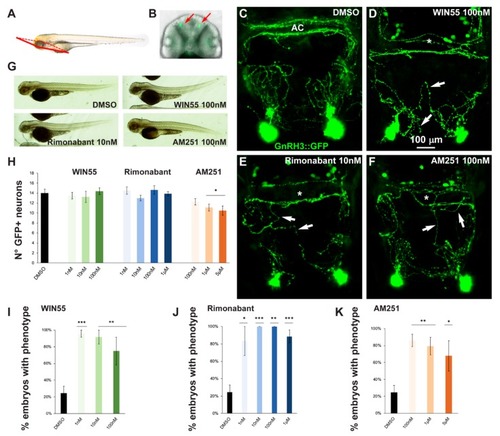
Pharmacological manipulation of CB1R on GnRH3 neurons in zebrafish embryos. ( EXPRESSION / LABELING:
PHENOTYPE:
|
|
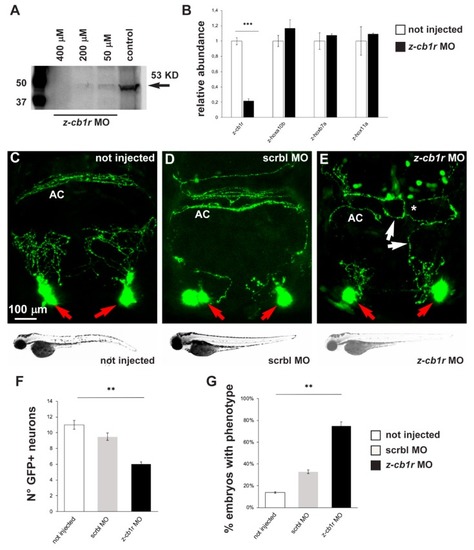
Effects of MO-mediated CB1R knockdown on GnRH3 neurons in zebrafish embryos. ( EXPRESSION / LABELING:
PHENOTYPE:
|
|
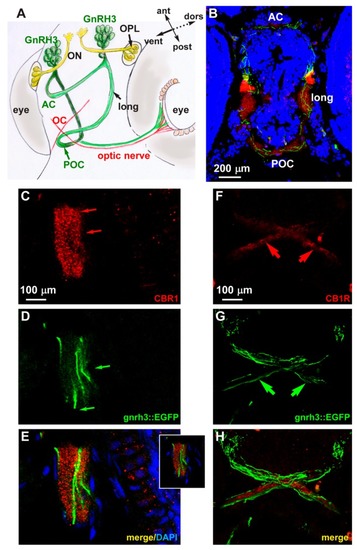
Expression of CB1R in developing zebrafish brain. ( EXPRESSION / LABELING:
|
|
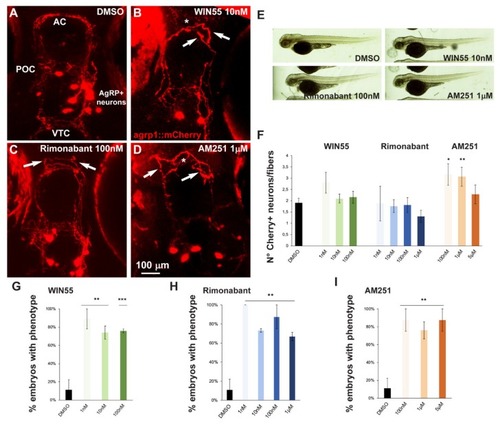
Effect of pharmacological manipulation of CB1R on AgRP1 neurons in zebrafish embryos. ( |
|
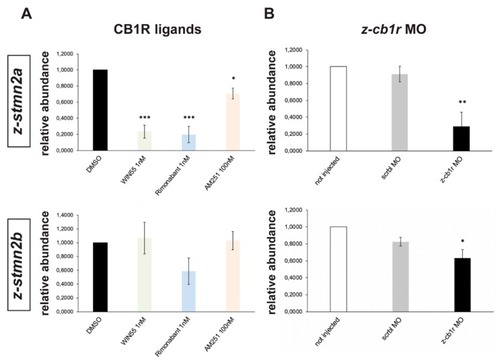
Expression of EXPRESSION / LABELING:
PHENOTYPE:
|